66 Salvage of Complex Problems
Summary
This chapter provides a series of extremely complex problems, both cosmetic and reconstructive, providing the authors’ analysis, solutions, and outcomes.
Key Topics
Latissimus flap salvage of failed expander reconstructions in radiated patients.
Salvage of radiated infected mastectomy site with deep inferior epigastric perforator (DIEP) flap in a high-risk patient.
Free Transverse Rectus Abdominis Myocutaneous (TRAM) flap salvage of massive full-thickness chest wall defect following resection of radiation-induced osteoradionecrosis of the chest wall.
Revision of capsular contracture and latissimus dorsi (LD) flap in the thin patient.
TRAM flap and LD flap salvage of failed bilateral expander reconstruction with irradiated tissue.
Aesthetic revision of LD flap reconstruction.
Aesthetic revision of a TRAM flap reconstruction.
Correction of left breast radiation deformity and asymmetry with animation deformity.
Salvage of multiple failed breast reconstructions with bilateral inferior gluteal artery perforator (IGAP) flaps.
Salvage of TRAM flap necrosis and breast deformity with latissimus flap.
LD flap after augmentation mammoplasty.
Augmentation of an autologous LD flap reconstruction with fat grafting.
Fat grafting to fill in a lower pole deficiency and infraclavicular hollow after an atrophied LD flap.
Fat grafting as a salvage procedure for correcting severe capsular contracture after implant reconstruction.
Key Teaching Points
For patients with thin skin and breast implants, it is appropriate to place a substantial vascularized layer of tissue between the breast implant and the intact skin. This can be either muscle or autologous fat graft.
When flap loss is lateral, the LD flap is a valuable salvage procedure to fill the defect.
When extensive skin and fat necrosis develops in any autologous reconstruction, it is preferable to perform early radical debridement and closure rather than allow second intention healing.
TRAM flaps in patients with subcostal incisions should be approached with extreme caution.
Fat grafting has assumed an important role in salvage of complex problem cases.
66.1 Introduction
The concept of this chapter has been to accumulate a series of complex cases encompassing a wide range of problems, which we confront in daily clinical practice. Many of the cases will address multiple problems such as inframammary crease asymmetry, volume deficits, hollowing, rippling, radiation complications, fat necrosis, or capsular contracture. Some of these problems are our own, but many have been referred for secondary revision. The aim of the chapter is to provide the reader with an insight into the thought processes accompanying the authors’ varying approaches to resolving these complex issues.
Observations
Well-planned and carefully executed surgical procedures usually result in excellent outcomes. Unfortunately, we do not live in a perfect world and even the best surgeons experience negative outcomes. It is also true to say that the more competent the surgeon, the more likely he or she is to be referred difficult cases to salvage. These patients are often high-risk cases with multiple comorbidities that make a successful outcome even more challenging to achieve. It was once said “a surgeon requires the eye of a hawk, the heart of a lion and the touch of a dove.” Nowhere is this more applicable than when facing complex case salvage.
Working with compromised tissue is familiar terrain to the reconstructive surgeon. Some problems, however, demand special ingenuity—a combination of techniques to produce the best result. In reoperative cases, tissues may be thinned, implants contracted and even extruded, and microcirculation impaired. Partial or total flap loss, correction of Poland’s syndrome deformity, and repair of a deteriorated previous reconstructive effort require surgical versatility to salvage these difficult situations. An LD flap may be needed to supplement thinning skin or prevent extrusion after augmentation or reconstruction. A TRAM/DIEP flap may be indicated for secondary reconstruction after an initial reconstruction with implants or after an LD flap has atrophied with time. Well-vascularized autologous tissue will be needed to cover an open chest wound resulting from radiation necrosis or to treat radiation fibrosis, and a TRAM flap or DIEP flap may be needed for bilateral reconstruction if a patient has a major deficit. The surgeon’s knowledge and experience provide the building blocks for solving these puzzling problems.
In this chapter, we present a series of patients with difficult reconstructive problems and complications following breast surgery. The format is a didactic approach: the clinical scenario is described, followed by an analysis of the problems associated with the case. An operative plan is then detailed, and postoperative photographs of the surgical correction are presented and evaluated, with a brief outcomes analysis.
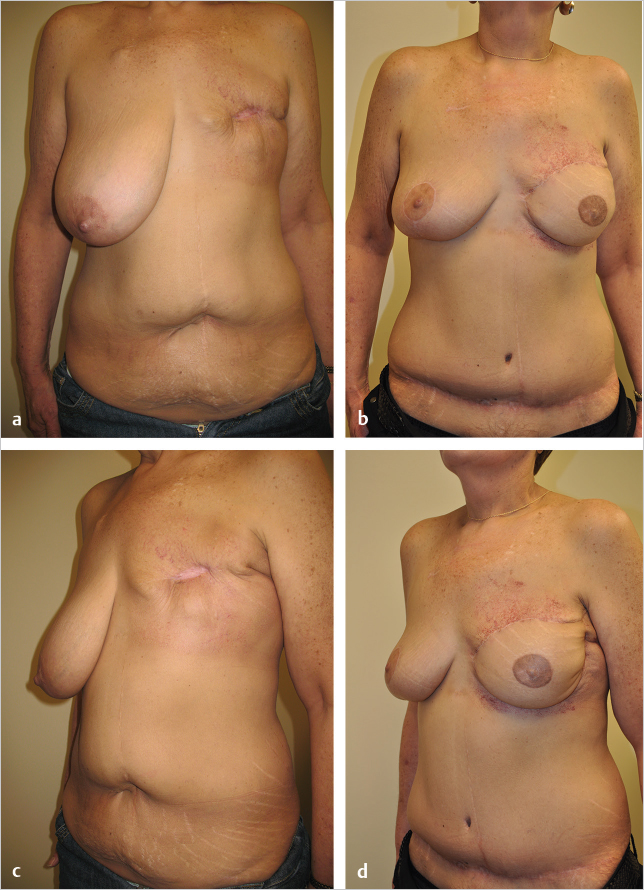
66.2 Case 1: Latissimus Flap Salvage of Failed Expander-Implant Reconstruction
66.2.1 History
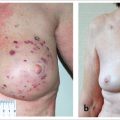
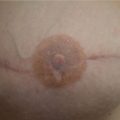
A 32-year-old woman presented to her surgeon for a prophylactic bilateral nipple sparing mastectomy as part of the management of BReast CAncer gene 1 (BRCA1) diagnosis. She was active and highly motivated. The breast surgeon performed bilateral nipple-sparing mastectomies and her plastic surgeon performed immediate subpectoral direct-to-implant insertion with lower pole acellular dermal matrix (ADM) coverage. Postoperatively, she developed infection, requiring explantation of the right implant with exchange for an expander, which was partially inflated. Subsequently, she developed signs of erythema with pain in both breasts and presented to the author for further management (▶Fig. 66.1)
66.2.2 Analysis
Probable recurrent infection now involving both breasts (right expander, left smooth gel implant).
Slim patient with BMI of 21.
Asymmetry.
Unsuitable abdominal volume. Well-shaped buttocks, but the patient refused gluteal flap harvest.
Thin attenuated skin with multiple areas of imminent erosion.
Surgical Plan
First stage: bilateral explantation.
Second stage: bilateral LD myocutaneous flaps with re-elevation of the skin and expander insertion, preserving the nipples, using LD flap skin islands to recover lower pole domain.
Third stage: expander implant exchange.
Fourth stage: staged excision of the inferior skin islands.
66.2.3 Procedure
First Stage
The patient was brought to surgery for explantation of all foreign material. The pockets were curetted, cultured, and lavaged with triple antibiotic solution and drains were inserted. She was allowed to heal for 2 months (▶Fig. 66.2).
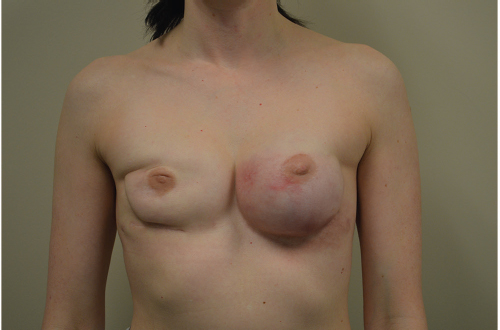
Second Stage
With the patient healed, she was returned to the OR for bilateral LD myocutaneous flaps with underlying expanders wrapped in ADM. The flaps were harvested in the prone position, passed into the axilla before donor site closure, and the patient was then turned supine for flap inset. The reconstruction was performed in the prepectoral plane. Expanders were inflated to 200 mL at operation and further expansion began at 2 weeks to a final volume of 400 mL. The skin islands were used to replace the thin, attenuated scarred skin of the most inferior aspect of both breasts.
Third Stage
Expander implant exchange for bilateral 375-mL round gel implants was performed (▶Fig. 66.3).

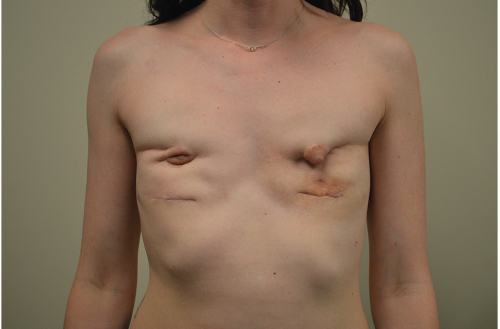
Fourth Stage
With the breast volume established and implants in place, two-staged excisions of her lower pole LD flap skin islands were performed, removing only the surface skin and leaving the fat and deep dermis for volume and padding over the lower poles of both implants (▶Fig. 66.4).

66.2.4 Outcome
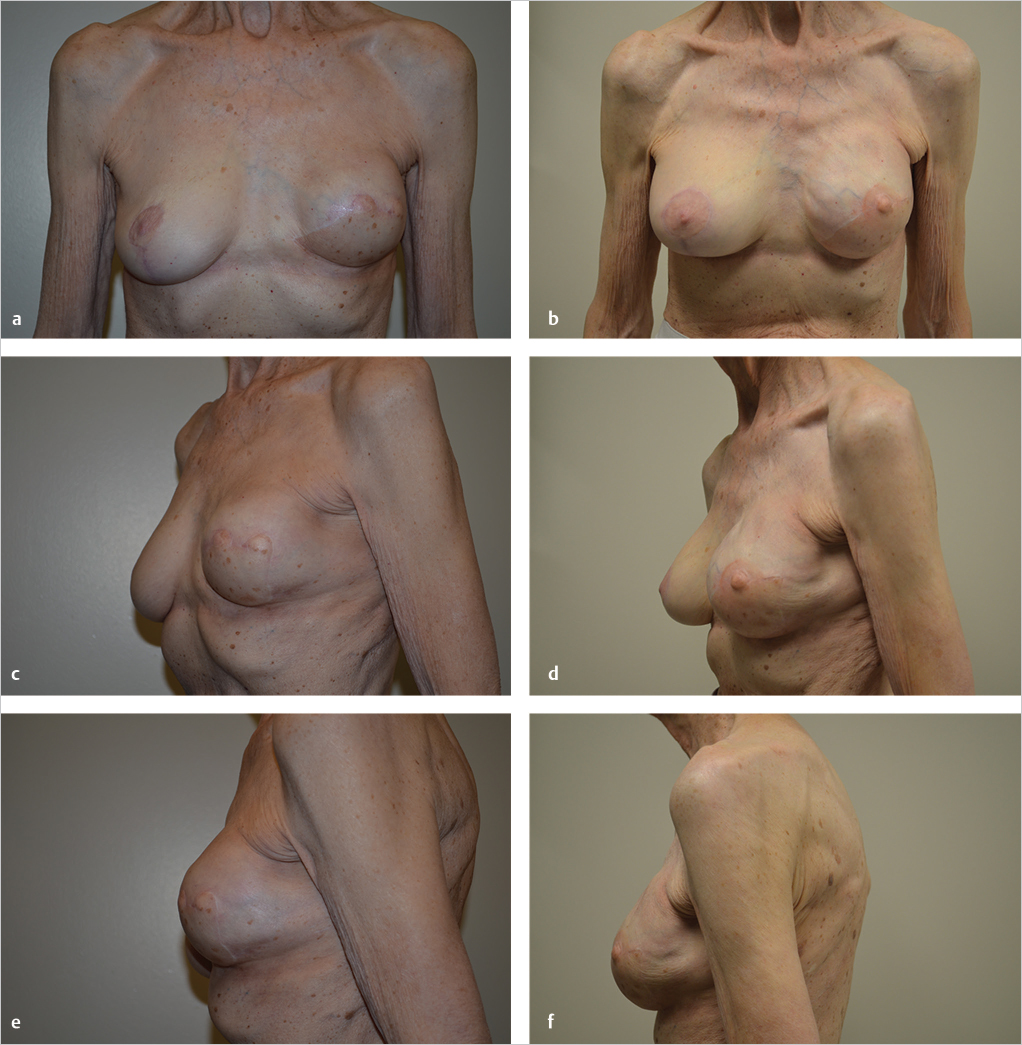
The patient is shown after explantation and then after her final stage of skin excision of her LD flap skin islands. The process took 12 months to complete. She was restored to an excellent shape with fuller breasts than she had had prior to mastectomy, with soft, grade l capsules. She has returned to full exercise including rowing and lifting weights despite bilateral latissimus flap harvest (▶Fig. 66.5).
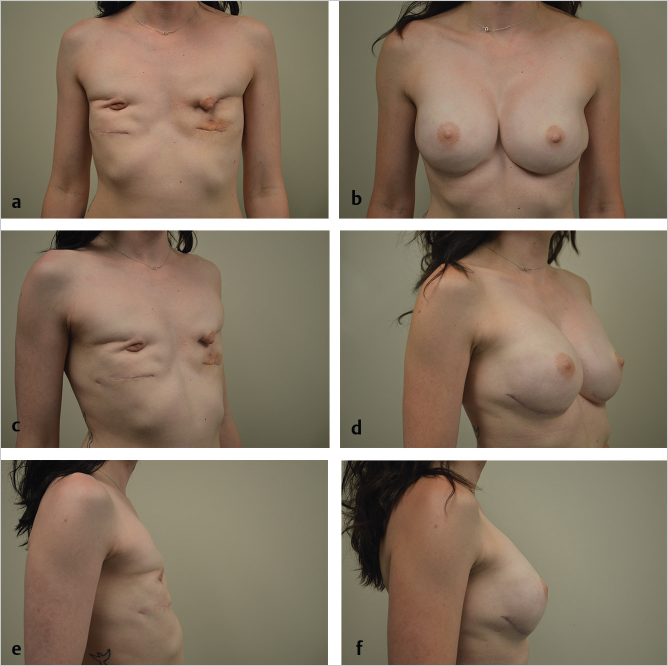
66.3 Case 2: Salvage of Radiated Infected Mastectomy Site with DIEP Flap in a High-Risk Patient
66.3.1 History
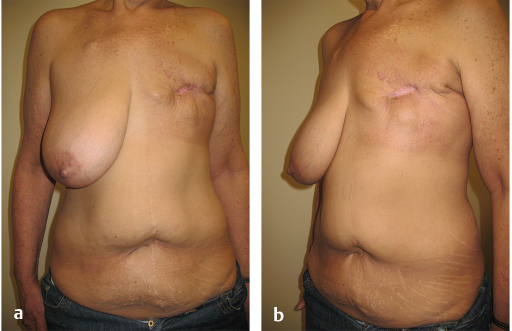
This 58-year-old woman with a 30-pack-year history of smoking developed left breast cancer. She had a left mastectomy with expander insertion and went on to get radiated. Shortly after completion of radiation, her expander became infected and had to be removed. She was colonized with methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococci (VRE) and developed a chronic draining chest wall wound, which took 9 months to heal by second intention after two failed attempts at closure by her plastic surgeon. She was referred to the author for salvage (▶Fig. 66.6).
66.3.2 Analysis
The patient was still smoking a pack a day.
Her LD flap donor site was surprisingly tight.
Radiated left chest wall.
Her chest wall was so tight that she required a large skin island plus volume.
She had an acceptable volume pannus but had a left paramedian incision from prior laparotomy.
Right breast was hypertrophied with grade 3 ptosis.
The patient was told that no surgery would be performed until she had stopped smoking. Given the paucity of her LD flap donor site, a muscle-sparing 2 (MS2) free TRAM flap was planned in order to capture the maximum number of perforators; it was felt that a DIEP flap would be riskier but not out of the question.
Surgical Plan
Stage 1:
Stop smoking altogether.
Preoperative multidetector computed tomographic angiography (MDCTA) or magnetic resonance angiography (MRA) of abdomen to evaluate vascular anatomy giver her prior laparotomy.
Stage 2:
Muscle sparing free TRAM flap based on right deep inferior epigastric artery and vein (DIEA/V) to left internal mammary artery and vein (IMA/V).
Stage 3:
Right breast reduction.
Left nipple–areolar reconstruction (NAR).
66.3.3 Procedure
Stage 1
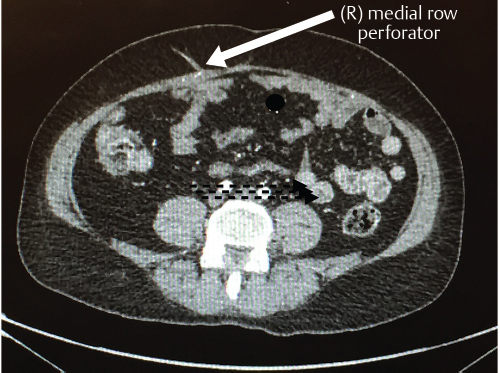
The patient stopped smoking over a 6-week period. Random cotinine testing confirmed her compliance. An MDCTA of the abdominal wall vascularity demonstrated several sizable perforators lying within the right medial rectus abdominis muscle (▶Fig. 66.7).
Stage 2
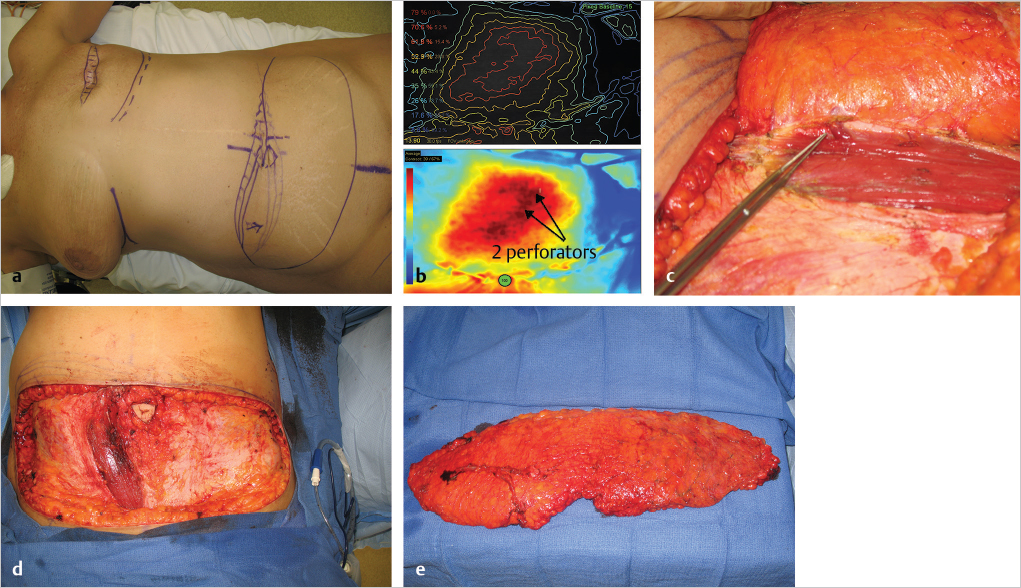
The patient was taken to surgery with a plan to perform an MS2-free TRAM flap, but the possibility of a DIEP flap was entertained based on the CTA results (▶Fig. 66.8a). A SPY angiogram with indocyanine green (ICG) was performed at the onset of surgery and two large perforator blooms could be seen arising from the medial row of the right rectus muscle; these were robust enough to commit to a DIEP flap instead of MS2-free TRAM (▶Fig. 66.8b,c). The flap was raised incorporating the cross midline tissue, but the tissue lateral to the left paramedian incision was discarded (▶Fig. 66.8d,e). The flap was anastomosed to the left IMA/V and the patient had an uncomplicated course apart from some minor skin necrosis of her donor site abdominal skin flap to the right of the midline. The DIEP flap healed without problems (▶Fig. 66.9).
Stage 3
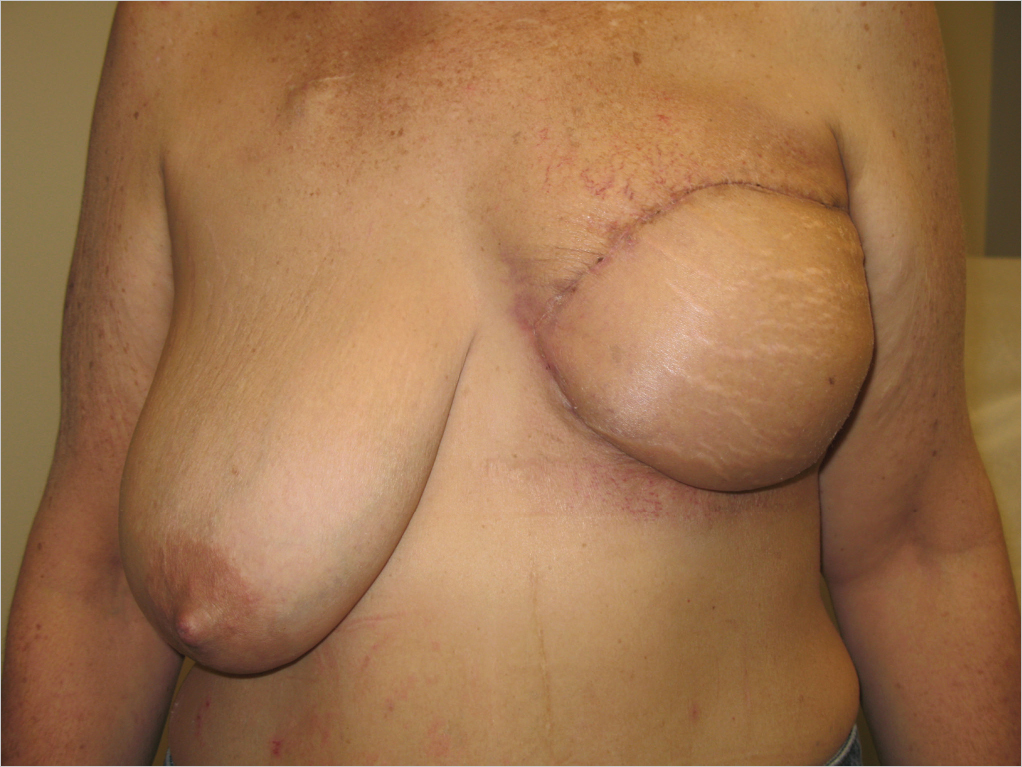
Six weeks later, she was taken back to surgery and a right vertical breast reduction and left nipple reconstruction was performed.
66.3.4 Outcome
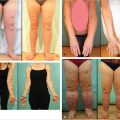
She is shown before and 1 year after surgery. She did well for 4 years but unfortunately succumbed to pancreatic cancer recently (▶Fig. 66.10).
66.4 Case 3: Free Tram Flap Salvage of Massive Full-Thickness Chest Wall Defect Following Resection of Radiation-Induced Osteoradionecrosis of the Chest Wall
66.4.1 History
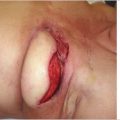
This elderly woman had undergone a Halsted radical mastectomy 30 years previously for left breast cancer. Over the years, she developed thinning of her skin with gradual but progressive ulceration and bleeding from the radiated anterior chest wall (▶Fig. 66.11).
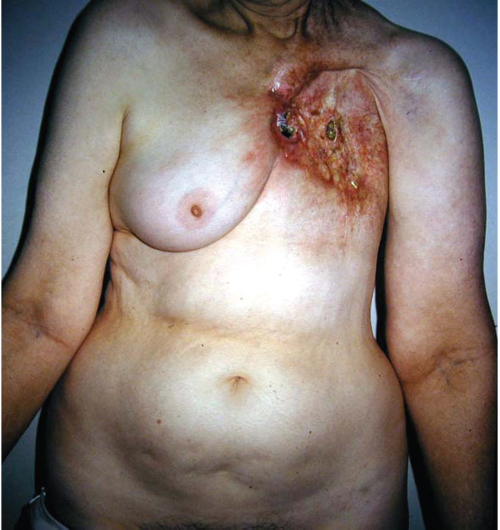
Eventually, she ulcerated through to the lung and was referred to us by the thoracic service. Dr. Bostwick had planned a bipedicled TRAM flap reconstruction, but the thoracic surgeons resected the entire sternum and both IMA/V vessels to their source. The author was called in to perform a free TRAM flap (▶Fig. 66.12).
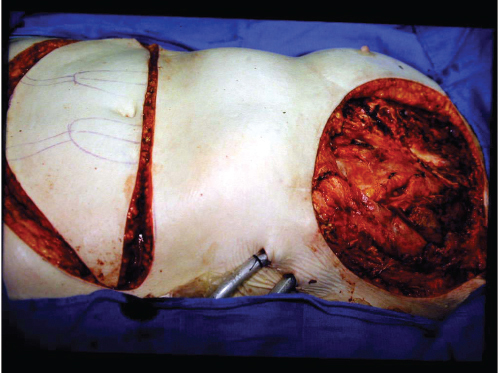
Assessment
Massive cross midline 30 × 25 cm defect.
Exposed heart and both lungs without pleura or pericardial covering.
Small stump of the left IMA arising from the great vessels.
Surgical Plan
Large free TRAM flap based on right DIEA/V.
Arterial anastomosis to left IMA stump.
Venous anastomosis end to side to innominate vein.
66.4.2 Procedure
The flap was raised with both pedicles in case perfusion from one side was inadequate (at that time, no perfusion assessment devices were available). The DIEA was suture end to end to the left IMA stump with 9–0 nylon. As the internal mammary veins (IMVs) had been totally resected, and end-to-side anastomosis of the IMV to innominate vein was performed using a Satinsky side clamp to the great vessels. Perfusion proved to be excellent to all zones of the flap in this otherwise healthy nonsmoker (▶Fig. 66.13).
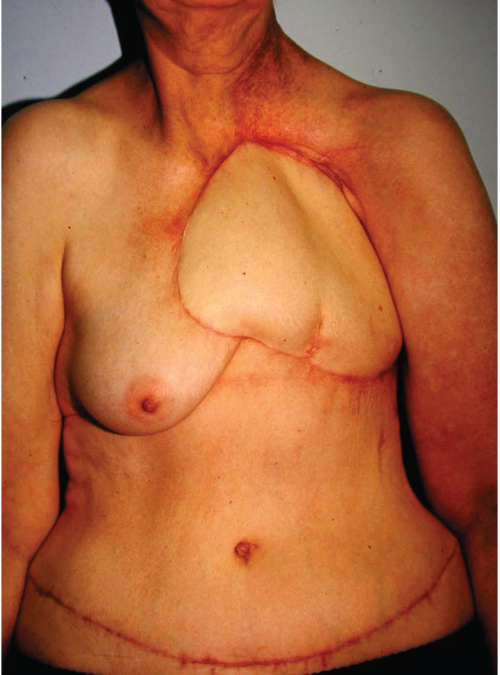
66.4.3 Outcome
The patient healed uneventfully and chest tubes were removed at 3 days postoperatively. She left hospital at postoperative day 7. As no methylmethacrylate was used to buttress the chest wall due to the stiffness of her radiated lungs and heart, she wore an acrylic chest shield when she drove a car. She was followed for 7 years until she moved elsewhere, but she remained solidly healed without respiratory problems during that time.
66.5 Case 4: Revision of Capsular Contracture and LD Flap in the Thin Patient
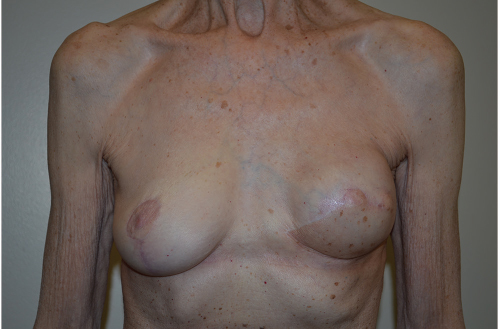
A 70-year-old woman presented having had right breast cancer 20 years ago. She had a left mastectomy. Ten years later, she underwent delayed left latissimus flap and implant reconstruction with contralateral augmentation. Her initial result was good with reasonable symmetry. She then went on a diet and exercise program and lost excessive amounts of weight to a BMI of 16 as well as developing painful grade 4 capsular contracture on the left side. Although she was not radiated, her left pectoral muscle was very tight and she had significant animation deformity (▶Fig. 66.14).
66.5.1 Analysis
Excessively thin with BMI of 16.
Asymmetry: left smaller than right.
Grade IV painful capsular contracture left breast.
Left breast to far lateral.
No cleavage.
Good quality left LD flap tissues.
No fat available for fat grafting.
Severe animation deformity.
Surgical Plan
Left breast prepectoral conversion with anterior ADM coverage.
Conversion to anatomic implant to improve profile.
Larger implants bilaterally.
The patient was appraised of the plan and warned that rippling would become an issue despite cohesive gel implant usage. She was not a candidate for fat grafting as her fat reserves were almost nonexistent. Her abdomen was too thin and tight to be used as an autologous donor site. She was informed that prepectoral conversion would resolve her pectoral muscle tightness and eliminate animation deformity.
66.5.2 Procedure
At operation, her left breast implant was removed and the pectoralis major muscle was returned to the chest wall, creating a prepectoral pocket for the new implant. A 16 × 20 cm sheet of AlloDerm (LifeCell, Branchburg, NJ) was sutured to the anterior surface of the pectoralis major creating a prepectoral pocket.
A full-height textured anatomic implant was inserted. On the right side, the old round implant was exchanged for a larger moderate-height anatomic implant.
66.5.3 Outcome
The patient is shown 3 years after her procedure with good shape retention and minor rippling of the medial upper edge of the left breast. She had a grade 1 capsule bilaterally and is delighted with her level of comfort, aesthetics, symmetry, and lack of animation deformity (▶Fig. 66.15).
66.6 Case 5: TRAM Flap and Latissimus Dorsi Flap Salvage of Failed Bilateral Expander Reconstruction with Irradiated Tissue
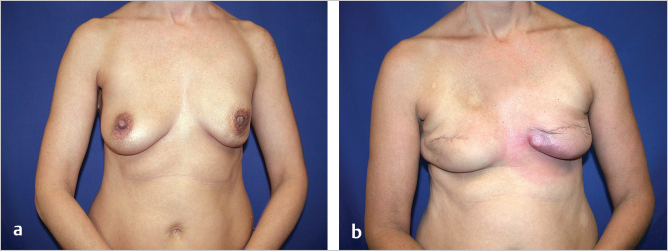
66.6.1 History
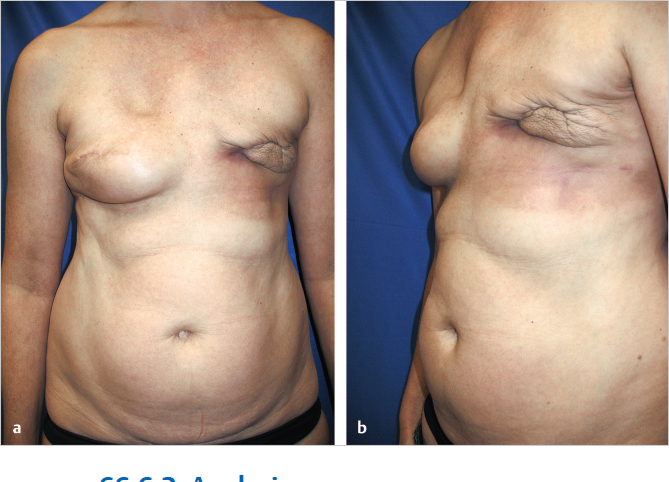
This 42-year-old woman developed cancer of the left breast that required a mastectomy and subsequent radiation therapy. Her reconstructive surgeon opted to place interim tissue expanders with a view to performing expander–implant exchange after radiation therapy, assuming the skin envelope was soft enough and her shape acceptable. No ADM was placed. After radiation, her left expander developed attenuated coverage with ultimate erosion (▶Fig. 66.16).
The expander was removed. The skin overlying her partially inflated right expander was also showing signs of thinning. She was referred for salvage (▶Fig. 66.17).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree