65 Surgical Treatments of Lymphedema
Summary
This chapter describes various surgical treatments of lymphedema including lymphaticovenular anastomosis, vascularized lymph node transfer, vascularized lymph vessel transfer, and suction-assisted lipectomy. The authors review their experiences with each of these procedures and extensively discuss their indications, contraindications, and relevant clinical and technical pearls. After reading this chapter, the readers are expected to gain a conceptual understanding of these procedures and sufficient technical knowledge to start building their lymphedema surgical expertise.
Key Teaching Points
Most secondary lymphedema in the first world is secondary to cancer therapy.
Indocyanine green (ICG) lymphography is replacing traditional lymphoscintigraphy as the diagnostic modality of choice.
Complex decongestive therapy (CDT) is the first-line treatment of choice.
Surgery should only be offered if CDT has failed.
Vascularized lymph node transfer (VLNT) and lymphaticovenular anastomoses (LVA) are the commonest lymphatic procedures available.
VLNT is used for more diffuse pattern disease on ICG lymphography.
LVA is used for linear pattern disease on ICG lymphography.
Liposuction and surgical excisional debulking remain useful adjuncts in some patients.
Debulking followed by lymphatic surgery is our preference.
65.1 Introduction
Lymphedema is a progressive disease that begins with benign-appearing edema of the affected body part. When left untreated, ongoing inflammatory fluid accumulation leads to bulky solid tissue deposition, and eventually causes severe functional compromise and potentially life-threatening infections. While the American Cancer Society estimates the incidence of arm lymphedema following axillary lymph node dissection for breast cancer to be 30%, the true incidence is unknown. Reported incidences range from 5 to 89%. This diagnostic ambiguity is due to the absence of standardized diagnostic criteria. Until recently, lymphedema had been considered an untreatable condition and treatments were palliative in nature, focusing on edema control and amelioration of symptoms. New reconstructive micro- and supermicrosurgical procedures aimed at restoring damaged lymphatic drainage have redefined treatment expectations. Successfully reconstructed patients can now anticipate significant reduction of symptoms, functional restoration, and even potential cure.
65.2 Pathogenesis
Lymphatic defect can be hereditary (primary) or acquired (secondary). Several inheritable mutations have been associated with primary lymphedema. Secondary lymphedema is most commonly caused by infection with filarial nematodes. In developed countries, however, secondary lymphedema is predominantly attributed to cancer treatment. Whether the lymphatic defect is related to congenital inadequacy or acquired injury, such as infection, surgery, and radiation, similar cascades of lymphatic derangements seem to occur. Initial lymphatic stasis leads to compensatory lymphatic hypertension and lymph vessel wall hypertrophy. As the disease progress, the compensatory system eventually depletes. The valve mechanism fails, and bidirectional flow ensues, causing dependent interstitial edema. This early stage of the disease is fluid predominant. As the condition progress, the impaired lymphatic clearance causes local accumulation of adipocytes, keratinocytes, and fibroblasts. With time, lipodystrophy, subcutaneous fibrosis, and thickened hyperkeratotic skin develop. This is the late, solid-predominant stage of lymphedema and is frequently described as “irreversible swelling.”
65.3 Patient Evaluation
65.3.1 History
History should focus on etiology, symptoms, functional impairment, progression of disease, prior treatments, and response to prior treatments. The etiology of secondary lymphedema is frequently obvious. A classic history of persistent limb edema following oncologic surgical extirpation and adjuvant radiation is suggestive of secondary disease. Primary lymphedema, on the other hand, lacks an inciting event. Occasionally, patients with primary lymphedema correlate disease onset to unrelated events such as an insect bite or bone fracture. Primary lymphedema is classified into congenital, praecox, and tarda, depending on the time of disease onset. Although duration of disease frequently correlates with disease severity, the correlation is not consistent. Patients can present with severe lymphatic injury despite recent onset of disease. Conversely, patients with decades of symptomatic disease may have only mild lymphatic injury.
Symptoms of lymphedema can be categorized as those directly related to the limb swelling or those arising as complications of chronic, poorly controlled lymphedema. The former includes heaviness, tightness, joint stiffness, weakness, numbness, and pain. The latter includes contour deformity from lipodystrophy, hyperkeratosis, skin ulceration, and cellulitis. Documenting the types, severities, and frequencies of symptoms establishes an initial baseline for patients and facilitates future longitudinal patient tracking.
Disease progression is an important factor in formulating a treatment plan. Is the patient’s disease stable after an initially deteriorating course? Or does the patient continue to worsen? Patients with rapidly worsening disease are preferentially considered for surgery before their surgical candidacy worsens or surgery becomes contraindicated.
Since surgery is not the first-line treatment for lymphedema, many patients presenting to the clinic have had prior nonsurgical, conservative treatments. The surgeon should identify all prior interventions, the patients’ responses to them, and why were they discontinued. Were conservative treatments terminated based on reaching successful or unsuccessful endpoints? It is not uncommon to find that patients stopped their lymphedema therapy simply because they reached the maximum number of visits per year covered by their insurance carriers, and not because they arrived at outcome-based treatment endpoints.
65.3.2 Physical Examination
Physical examination starts with a visual assessment of the affected limb. Is the entire limb swollen? Is the swelling limited to the more dependent part of the limb? Next, is the “swelling” fluid, solid, or a mixture of both? Early lymphedema characteristically demonstrates fluid-predominant disease, and therefore, would pit with digital pressure. Pitting edema can be easily demonstrated by applying thumb pressure for 5 seconds over bony regions such as the pretibial or malleolar areas. Late, solid-predominant disease, characterized by prominent lipodystrophy and fibrosis, will not pit.
Limb volume can be directly measured by one of the following:
Three-dimensional (3D) optoelectronic perometry.
Water displacement technique.
Indirectly deduced with multilevel circumference measurements.
While 3D perometry is the preferred volume measurement method, it is currently not Food and Drug Administration (FDA) approved in United States and thus not easily accessible to most patients. Water displacement, on the other hand, is inconvenient and impractical for a busy clinic environment. Thus, the most commonly used method for limb volume assessment is still circumference measurement. Among various circumference measurement systems, our preferred method is lymphedema indices. Lymphedema indices reduce the impact of intra- and inter-rater errors and weight fluctuation by squaring measurements from multiple levels and incorporating the body mass index. Regardless of the measurement method chosen, it is important to understand the limitations of volume-based measurement. All volume-based measurements are influenced by a multitude of variables including weather, time of day, diet, activity level, and pressure garment use.
65.3.3 Confirmatory Studies
Lymphedema is traditionally considered a clinical diagnosis and can be diagnosed merely with history and physical examination. Many conditions, such as extremity venous insufficiency and lipedema, can mimic the edema, skin changes, and the solid disease bulk of lymphedema. For this reason, we recommend obtaining a confirmatory study, especially when surgical treatment is being considered. A definitive diagnosis can be achieved with following imaging modalities.
Lymphoscintigraphy
Lymphoscintigraphy using technetium-99m–sulfur colloid has been the conventional gold standard in diagnosing extremity lymphedema. The study is performed by injecting the radiopharmaceutical at the digital web spaces of the extremity of interest. The radiopharmaceutical is then taken up by the lymphatic system, sequentially moving through lymphatic capillaries, precollectors, collectors, major lymphatic trunks, and lymph nodes. The rate of transit and the flow patterns are diagnostic of various lymphatic disorders. The disadvantages of lymphoscintigraphy include poor spatial resolution and obligatory radiation exposure.
Indocyanine Green Lymphography
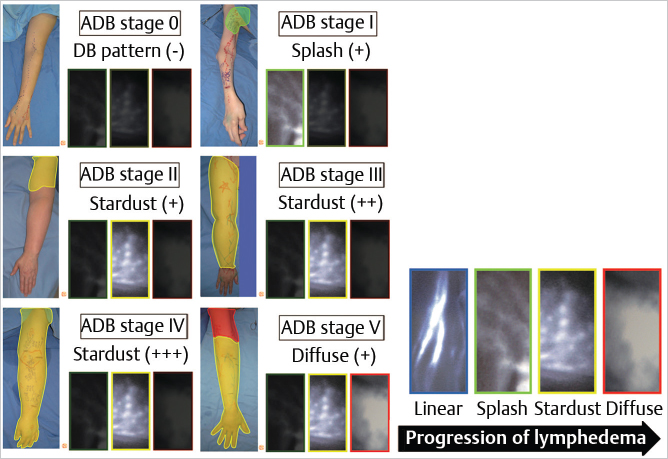
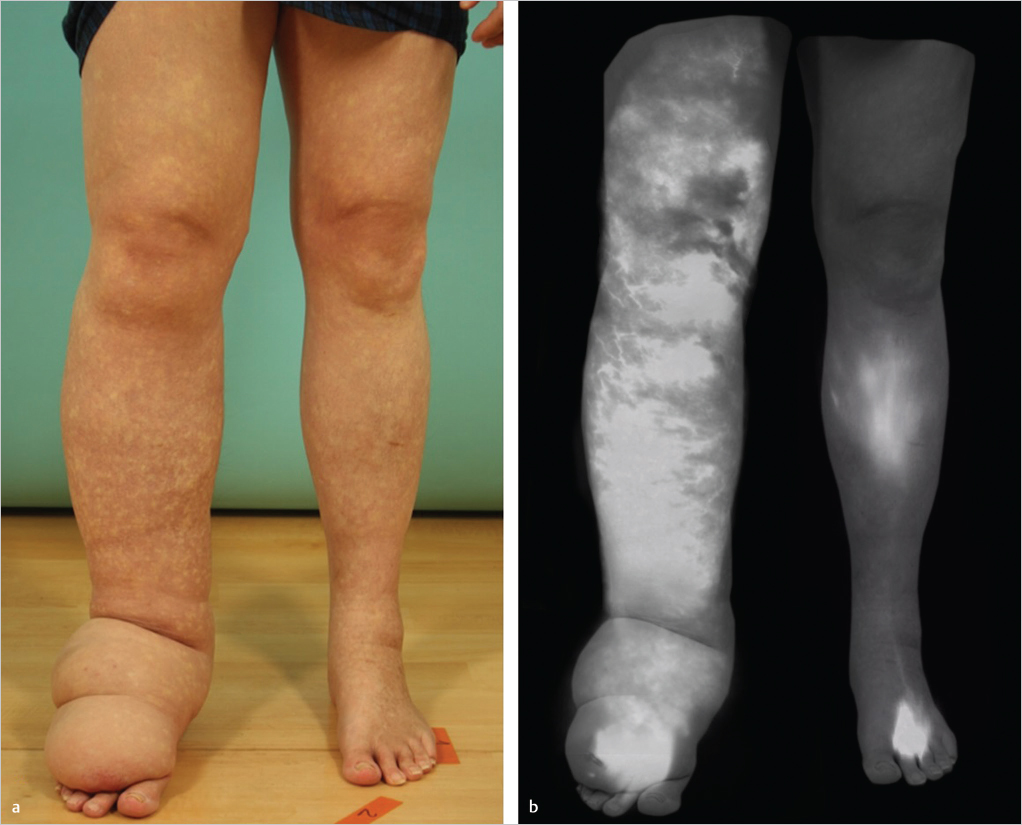
Similar to technetium-99 lymphoscintigraphy, indocyanine green (ICG) lymphography allows assessment of lymphatic physiology by peripheral injection of a contrast agent and allowing it to be taken up by the lymphatic system. Unlike lymphoscintigraphy, which uses a radioisotope, ICG lymphography relies on ICG, a fluorophore that fluoresces when optically excited at 800 nm. This property makes the study nonradioactive. The dermal backflow patterns of ICG in patients with lymphedema correspond to disease severities. Disease severity can be determined based on the presence and the locations of “splash,” “stardust,” and “diffuse” dermal backflow patterns (▶Fig. 65.1 and ▶Fig. 65.2—pictures showing diseased limb in contrast to healthy limb). ICG lymphography has also been shown to have higher sensitivity and specificity than lymphoscintigraphy. All of the above features make ICG lymphography particularly suitable as a longitudinal tracking study that can be repeated at various time points during the course of treatment. In our center, ICG lymphography has largely replaced lymphoscintigraphy as our study of choice for diagnosis, severity staging, preoperative surgical planning, and postoperative patient tracking.
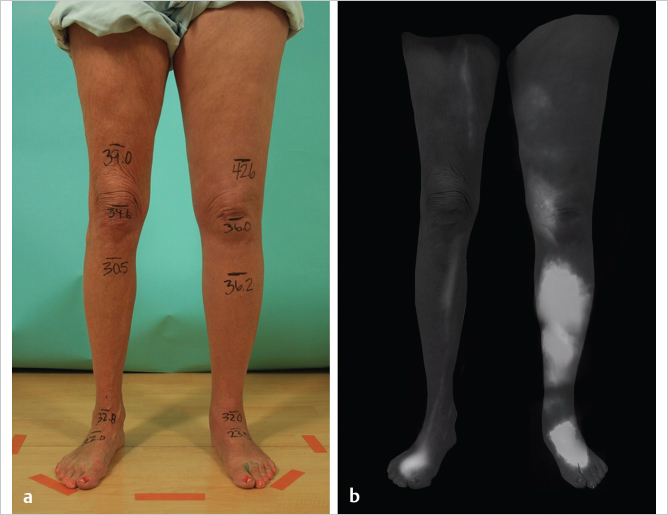
Magnetic Resonance Lymphangiography
Magnetic resonance lymphangiography (MRL) allows visualization of lymphatic vessels and surrounding structures without radiation exposure. MRL is performed with or without contrast using either T1 or T2 sequences. MRL demonstrates areas of dermal backflow, without the limited penetration depth of ICG lymphography, and the high-resolution images can be used in preoperative planning for reconstructive procedures. MRL is distinct from lymphoscintigraphy and ICG lymphography in that it can depict subcutaneous adipose deposition and fibrosis of solid phase lymphedema. Unlike the technetium-99m–sulfur colloid used in lymphoscintigraphy, the gadolinium contrast in MRL has water-soluble chelates and enhances veins in addition to lymphatic vessels.
65.3.4 Staging
Staging systems facilitate disease classification and communication among lymphologists. Commonly used staging systems include those published by the International Society of Lymphology (▶Table 65.1) and Corradino Campisi (▶Table 65.2). As reconstructive microsurgeons became increasingly involved in the field, staging systems with specific surgical considerations began to emerge. These include systems created by Ming-Huei Cheng et al, Hung-Chi Chen et al, and David Chang et al. There is currently no universally agreed upon staging system for lymphedema. Instead of examining the merits of each of the systems, it is more important to observe that all systems describe the same disease pathology and the same pathologic progression.
Stage | Description |
0 (Ia) | Impaired lymph transport without clinical evidence of swelling |
I | Early fluid accumulation that reverses with elevation |
II | Tissue swelling that pits and rarely reverses with elevation |
III | Lymphostatic elephantiasis |
Source: Adapted from International Society of Lymphology. | |
Stage | Description |
IA | Impaired lymphatic drainage without overt swelling |
IB | Swelling that completely reverses with limb elevation |
II | Swelling that only partially reverses with limb elevation |
III | Persistent swelling and recurrent lymphangitis |
IV | Fibrotic lymphedema with column limb |
V | Elephantiasis with limb deformation and skin changes |
Early lymphedema is characterized by reversible limb edema that resolves with compression and/or elevation. As the disease progresses, the limb bulk becomes progressively less reversible. This is indicative of development of the solid disease component. Gradually, the solid disease becomes more and more pervasive until the limb bulk becomes persistent and irreversible. Late, fulminant lymphedema is manifested by irreversible limb bulk and skin changes.
65.4 Treatments
Currently available lymphedema treatments can be thought of as the nonsurgical complex decongestive therapy (CDT) and several microsurgical and supermicrosurgical interventions. With few exceptions, CDT is the first-line treatment. Surgery is considered when CDT fails or when its result is unsatisfactory. CDT has the advantages of being noninvasive, highly effective, and more widely available than the surgical interventions. Lymphedema surgery was once synonymous with the Charles procedure, a purely ablative procedure created in 19th century, until recently. Modern technical advancements in micro-and supermicrosurgery make lymphatic reconstruction a reality by either bypassing lymphatic obstruction or transferring healthy functioning lymphatic tissue from a distant site to the affected limb. Due to their novelty and complex technical natures, both reconstructive modalities were initially controversial and not well accepted. After decades of experience with these procedures, more consistently favorable outcomes are now being reported in the literature.
65.4.1 Complex Decongestive Therapy
CDT is a comprehensive program that combines several nonsurgical treatment modalities including manual lymphatic drainage, pneumatic compression, bandaging, pressure garment use, Kinesio Taping, exercises, skin care, and lifestyle modifications. CDT is typically administered in two phases: an early, aggressive phase aimed at maximal edema reduction and relief of symptoms, and a late maintenance phase focused on long-term stability. When administered by trained therapists, CDT has consistently been shown to be highly successful in relieving symptoms, reducing edema, and decreasing infection rate.
The main challenge of CDT is the need for strict, lifelong adherence. Frequently, patients fail CDT because of their inability to comply with the rigor and duration of treatment. It is unknown whether CDT is disease modifying/altering, or if its impact is limited to amelioration of symptoms. It is also uncertain whether successful CDT can reverse lymphatic injury and create favorable anatomic changes. Another unanswered question of CDT is, what constitutes treatment failure? Since CDT’s role as an effective first-line treatment for lymphedema is widely accepted, most patients presenting to the surgeon have already had prior CDT that was deemed unsuccessful. Common practice describes failed CDT in terms of time. This practice is arbitrary and does not account for the varying intensities and frequencies practiced by different therapists. In our center, the definition of failure of CDT is protocol based instead of time based. Patients undergo an algorithmic tier-based CDT program and are only considered to have failed CDT after exhausting all CDT options.
65.4.2 Surgery
Lymphedema surgery is categorized into the following:
Reconstructive procedures: restore lymphatic drainage.
Debulking procedures: remove the disease-compromised tissue/skin.
Lymphaticovenular anastomosis (LVA) and vascularized lymph node transfer (VLNT) are the established workhorse reconstructive procedures. Suction-assisted lipectomy (SAL) and the Charles procedure are the mainstays of the debulking procedures. There is no one-size-fits-all procedure that treats all patients presenting with varying disease severities. Rather, the procedure of choice for a given patient should be patient and condition specific. Therefore, we recommend lymphedema surgeons become proficient with all the available surgical procedures.
Reconstructive Procedures
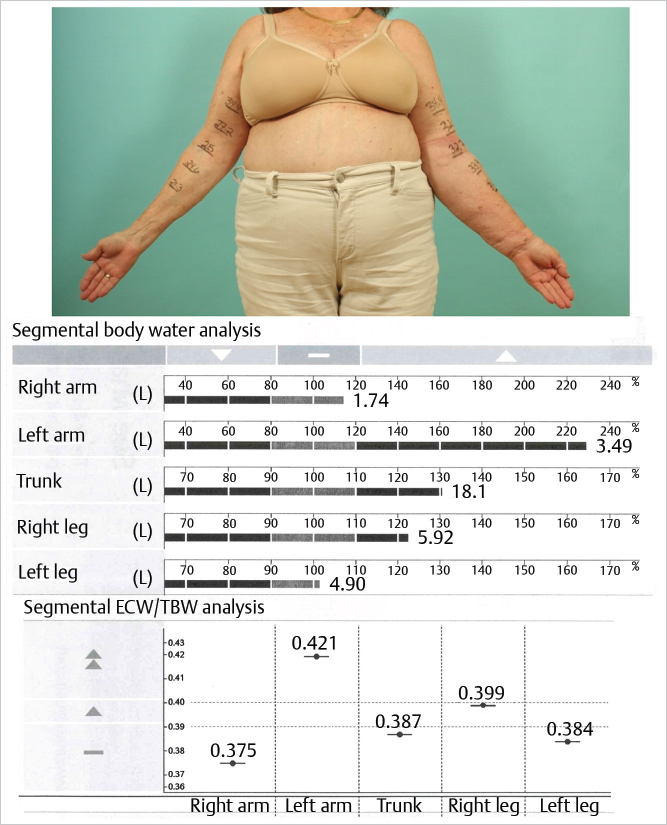
In early lymphedema, when patients report reversible edema, reconstructive procedures are appropriate. A relatively fluid-predominant state can be confirmed with bioimpedance spectroscopy (▶Fig. 65.3) or MRI. LVA and VLNT are both effective in treating fluid-predominant disease. The choice between the two rests on the conditions of the lymphatic vessels. The healthier the lymphatics, the more effective the LVA procedure. ICG lymphography can help differentiate patients with relatively preserved lymphatic systems from those with more severe injuries. If ICG lymphography demonstrates an abundance of high-quality “linear” patterns, LVA is the procedure of choice. On the other hand, severe disease with widespread “diffuse” patterns warrants VLNT (▶Fig. 65.4). Still, it is worthwhile to note that contrary to the common belief, LVA remains technically feasible even when the “linear” pattern is completely absent. The absence of “linear” pattern simply means the surgeon will no longer be able to “cut down” over the “linear” pattern to find healthy lymphatic vessels. Instead, a more labor-intensive exploratory search is necessary to find suitable lymphatic vessels. Therefore, in patients with primary lymphedema (further discussion later) or those who require a minimally invasive procedure for medical reasons, LVA may still be considered despite suboptimal ICG lymphographic findings.
When considering VLNT, the low but realistic risks of donor site lymphedema need to be thoroughly discussed with the patient. Despite the development of VLNT procedures using various anatomic donor sites, none is completely immune to this risk. Caution should especially be exercised when treating patients with primary lymphedema. Although the true incidence is unknown, it is not rare that ICG lymphography reveals asymptomatic lymphatic insufficiency in seemingly unaffected limbs of patients presenting with primary disease (▶Fig. 65.4). These patients are at particular risk for developing donor site lymphedema. If both the patient and the surgeon continue to prefer VLNT, preoperative ICG lymphography of the donor site and risk-reducing reverse lymphatic mapping are recommended.
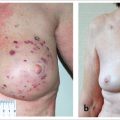
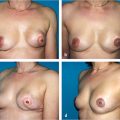
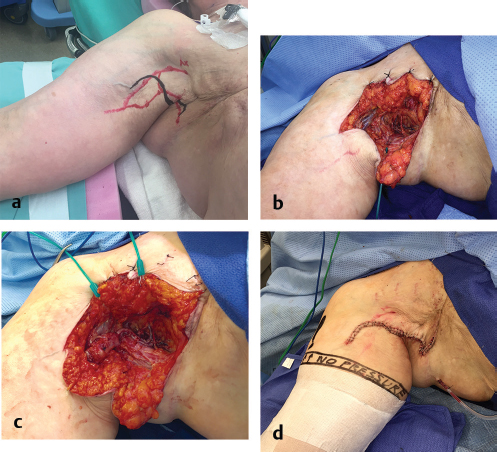
Additional VLNT technical considerations include the choice of recipient site and whether to include skin with the lymph node flap. It is controversial whether proximal or distal VLNT is more efficacious, but there is increasing consensus that the procedure works regardless of the flap’s final location. The procedure remained efficacious even when VLNT was transferred to intermediate sites such as the elbow or calf. It is also now widely recognized that thorough clearance of scar contracture in preparation for proximal transfer, such as excision of axillary scar contracture, is in itself beneficial and contributes at least partially to the therapeutic effects seen in the proximal transfer. Therefore, we recommend the decision of proximal versus distal transfer to be patient specific. Proximal transfer may be advantageous in patients with proximal scar contracture (▶Fig. 65.5). If distal transfer is planned, the expected contour deformity from the bulk of the lymph node flap needs to be discussed with the patients (▶Fig. 65.6).
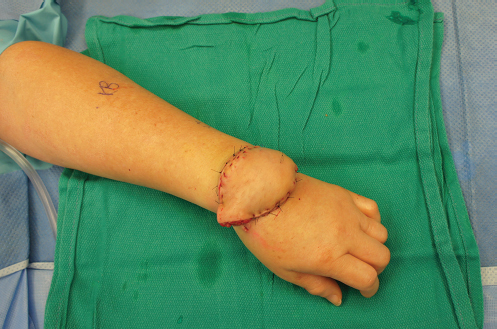
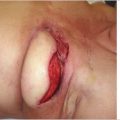
Inclusion of skin with the lymph node flap in theory includes superficial lymphatic vessels that increase the effectiveness of the procedure. At this time, there is no consensus among lymphatic reconstruction surgeons on this issue, and the VLNT procedure seems to work regardless of whether skin is included or not. In deciding whether to include the skin, the surgeon should consider both the donor and the recipient sites. The transverse cervical artery-based lymph node flap, for example, does not have overlying skin that is consistently drained by the accompanying venous pedicle, and may be more reliably performed as a skinless flap. If distal transfer to ankle or wrist is planned, inclusion of skin is advantageous to avoid the need for skin grafting.
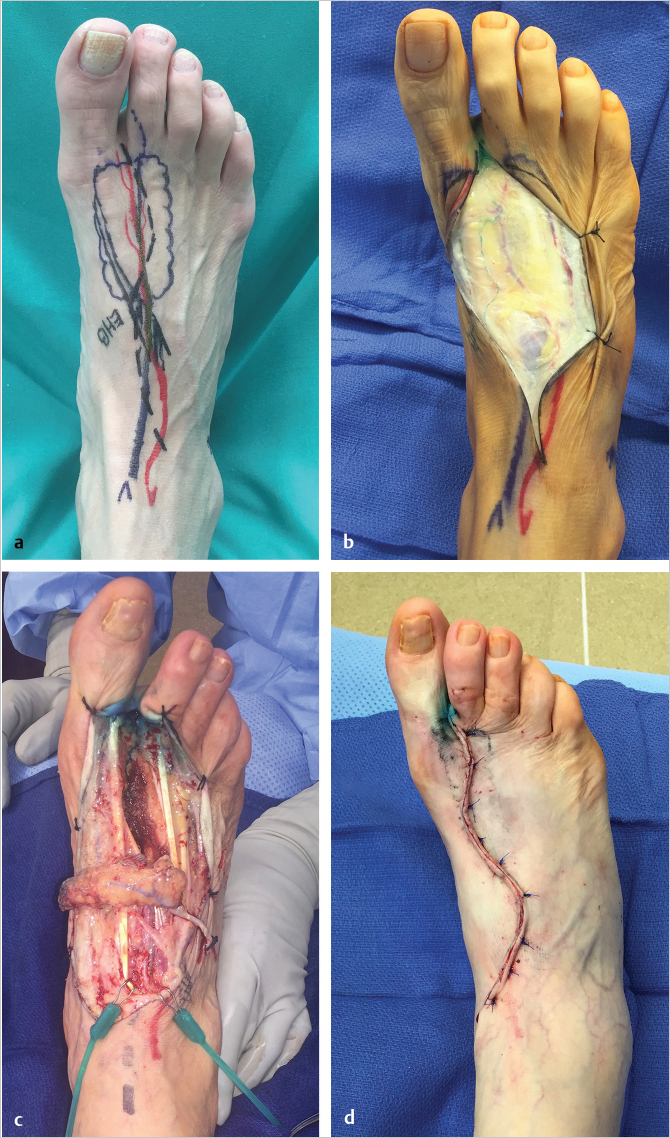
What happens when only the lymph vessels are transferred? Recently, the vascularized transfer of the first dorsal metatarsal artery (FDMA) related perivascular lymph vessels was reported as a successful treatment for patients who had failed to improve with the LVA procedure (▶Fig. 65.7). As with VLNT, how the transferred lymph vessels interact with the recipient lymph vessels and how the procedure treats lymphedema are not well understood. Further studies are clearly necessary, but the efficacy of vascularized lymph vessel transfer (VLVT) questions the need to transfer nodal tissue.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree