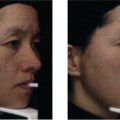
Emily C. Milam1, Alexandra Price2, Sarika Ramachandran3, and David E. Cohen1 1 The Ronald O. Perelman Department of Dermatology, New York University Grossman School of Medicine, New York, NY, USA 2 Dermatology Partners, Inc., Wellesley, MA, USA 3 Department of Dermatology, Yale School of Medicine, New Haven, CT, USA Topical cosmetic medications, cosmeceuticals, and minimally invasive cosmetic procedures play an important role in dermatologic practice. Recent advances have led to a tremendous expansion in the repertoire of nonsurgical cosmetic treatments. The wide use of cosmetics and skin care products worldwide increases the possibility of exposure to irritants and contact allergens [1]. Adverse skin reactions to cosmetics include irritant contact dermatitis (ICD), allergic contact dermatitis (ACD), phototoxic dermatitis, contact urticaria, and foreign body reactions [2, 3]. The clinician should be aware of these potential skin reactions and seek to identify the causative agents and other contributing factors [4]. Most of these reactions are treatable without sequelae once the offending agent is recognized and avoided. Among patch‐tested patients with suspected ACD, cosmetic agents are implicated in 4–9% of cases [5]. Women between 20 and 55 years of age are most affected by cosmetic contact dermatitis [5–7]. The US Food and Drug Administration (FDA) regulations on cosmetics are based on two important laws: the Federal Food, Drug, and Cosmetic Act (FD&C), which prohibits the marketing of adulterated or misbranded cosmetics, and the Fair Packaging and Labeling Act (FPLA), which states that improperly labeled or deceptively packaged products are subject to regulatory action [8]. Ingredient labeling is mandatory in the United States and Europe, and compounds are listed in descending order of amount using the nomenclature format of the International Cosmetic Ingredient Dictionary [9, 10]. Currently, listing specific fragrance allergens is voluntary, which allows cosmetic manufacturers to include individual fragrances under a generic “fragrance” listing [11]. With the exception of color additives, cosmetic products and ingredients are not subjected to FDA premarket approval and manufacturers’ reporting of adverse reactions is a voluntary process [8]. Notably, the FDA has not yet established regulatory definitions for certain cosmetic labeling terms, such as “natural,” “organic,” or “botanical.” In order to review the safety of the cosmetic ingredients, the Cosmetic, Toiletries and Fragrance Association (CTFA) sponsors the Cosmetic Ingredient Review (CIR). In 2012, CIR identified 11 ingredients that were unsafe for use in cosmetics [12]. In addition, CIR identified 38 ingredients that were not supported for use in cosmetics [12]. The Safe Cosmetics and Personal Care Products Act of 2018 seeks to greatly expand FDA oversight of chemicals in cosmetic products. This legislation would require companies to disclose all toxic ingredients, report adverse events and demonstrate that cosmetics meet safety standards prior to marketing [13, 14]. Reactions to cosmetics can present with a wide range of clinical signs. Therefore, it is important for the clinician to be familiar with the diversity of these presentations to enable prompt diagnosis and treatment. A majority of contact dermatoses are classified as ICD. ICD is a nonimmunologic consequence of a chemical or physical agent’s direct cytotoxic effect on the skin. The three main pathophysiological changes observed are stratum corneum disruption, epidermal cellular changes, and proinflammatory cytokine release [15]. Ceramides, a major component of the stratum corneum, play a key role in the protection against irritants [16]. Repeated exposure to irritants disrupts the epidermal barrier and increases transepidermal water loss (TEWL) [17]. The severity of irritant dermatitis depends on the chemical properties, amount and strength of the agent, and length and frequency of exposure. Repetitive exposures even to mild agents, such as soaps and detergents, can result in irritant dermatitis. In addition, harsh scrubbing with mechanical assistance (brushes, synthetic sponges, or cosmetics containing microabrasive spheres) increases the risk of irritation. Psychiatric disorders leading to obsessive‐compulsive skin cleansing can also damage the barrier function of the skin and cause irritant dermatitis [18]. Predisposing factors for the development of irritant dermatitis also include endogenous disorders, such as atopic dermatitis and filaggrin gene defects [19]. ACD comprises a substantial number of cases of contact dermatitis and represents a true delayed‐type (type IV) immune reaction. Previous exposure and sensitization to the causative agent are necessary [2]. Chemical agents act as haptens, which are small electrophilic molecules that bind to carrier proteins and penetrate the stratum corneum barrier of the skin. The antigen‐presenting cells (APCs) of the skin (Langerhans cells and/or dermal dendritic cells) digest and display the hapten–protein complex on their surface for presentation to T lymphocytes. Hydrophobic contact allergens may displace lipids normally bound to CD1a, a glycoprotein expressed on Langerhans cells. T cell activation may thereby occur by exposing T cell stimulatory surface regions of CD1a which are normally hidden [20]. The hapten‐specific T lymphocytes differentiate into memory T cells, which then undergo clonal expansion. The clinical manifestations of ACD are mediated by the activation of hapten‐specific memory T cells in the skin upon re‐exposure to the offending allergen. Sensitization depends on product composition, the potency of the sensitizer, the amount of product applied, the frequency and duration of application, and the physical integrity of the epidermal barrier [21, 22]. Filaggrin gene mutation carriers with concurrent atopic dermatitis or hand eczema have an increased risk of developing contact sensitization [23]. While there are no pathognomonic features that unequivocally distinguish ACD and ICD, there are certain clinical features that favor ACD over ICD. While itching can be present in both conditions, it is a key symptom in ACD, whereas burning, stinging, soreness, and pain are more characteristic of ICD [24]. ACD occurs upon re‐exposure to the allergen following an initial phase of sensitization, whereas ICD is not predicated on prior sensitization and can occur on first exposure to the causative agent [25]. Peak reaction occurs slower in ACD (within 3–4 days) than in acute ICD (within minutes to hours), whereas chronic ICD develops gradually after repeated exposure to milder irritants [24]. The clinical distinction between irritant and allergic dermatitis can be challenging because both conditions can result in eczematous reactions. Clinical manifestations range from mild erythema and scaling with minimal itch to highly pruritic vesicular, bullous, and lichenified plaques. Furthermore, the two conditions can sometimes be superimposed since an irritated and impaired epidermal barrier can facilitate the absorption of haptens that can elicit an immune response in susceptible individuals. The risk of ACD appears to be elevated among patients with atopic dermatitis, or eczema. This is likely related to increased allergen permeation due to epidermal barrier dysfunction in atopic patients, augmented antigen presentation, immune dysregulation, and the patient population’s increased exposure to potentially sensitizing agents in topical corticosteroids and moisturizers [26, 27]. Phytophotodermatitis is a nonimmunologic phototoxic skin eruption caused by topical exposure to plant‐derived photosensitizing compounds and subsequent exposure to ultraviolet (UV) light. Furocoumarins (psoralens and angelicins) are the most common photosensitizing chemical agents [28]. Plant families that produce these compounds include the Apiaceae (parsley, celery), Rutaceae (citrus fruits), Moraceae (figs), and Fabaceae (peas). Long‐wave UVA radiation (340–400 nm) is responsible for the majority of phototoxic reactions resulting in phytophotodermatitis [3]. The skin eruption of phytophotodermatitis occurs on sun‐exposed areas in contact with plants containing these photosensitizing compounds [29]. Marked hyperpigmentation, usually in a bizarre configuration or linear pattern, is often the dominant physical finding on clinical presentation. Occasionally, bullous and vesicular lesions can also develop. These phototoxic reactions cause both DNA interstrand crosslinking between UVA‐activated psoralens and the pyrimidine bases of DNA and cellular membrane damage [30, 31]. Contact urticaria syndrome is divided into immunologic and nonimmunologic subtypes. Nonimmunologic contact urticaria is the most common form and occurs in the absence of previous exposure. Localized wheals appear within 30–60 minutes of exposure and are not followed by systemic symptoms. Allergic contact urticaria is an immediate‐type (type I) hypersensitivity reaction and occurs in sensitized individuals within minutes to hours following exposure to the allergen. The binding between allergens and immunoglobulin E (IgE) triggers mast cell degranulation and subsequent release of inflammatory products, such as histamine, prostaglandins, leukotrienes, and cytokines. As a consequence, individuals experience erythema, swelling, and pruritus, which may be localized (wheals and flares) or generalized (angioedema, conjunctivitis, bronchoconstriction, hypotension). Severe reactions may be fatal. Injectable fillers are a group of exogenous substances used for soft tissue augmentation. Fillers are subdivided into agents that are degradable (hyaluronic acid, poly‐L‐lactic acid, calcium hydroxylapatite) and nondegradable (polymethylmethacrylate and silicone). Some injectable agents such as hyaluronic acids restore volume primarily through their space‐filling effects. Others, including silicone, calcium hydroxylapatite, polymethylmethacrylate, and poly‐L‐lactic acid fillers, act as scaffolds for endogenous collagen formation. Adverse immunologic reactions have been reported with practically all the products used. The frequency and degree of these reactions vary based on filler material, injection technique, and host immunologic response. The normal initial host response to foreign body implantation is the formation of a blood‐based matrix on and around the biomaterial, called the provisional matrix. The tissue injury may also lead to activation of the innate immune response and thrombus formation. The provisional matrix is rich in mitogens, chemoattractants, growth factors, and cytokines, providing an excellent medium both for wound healing and foreign body reaction. Acute inflammation is characterized by the presence of neutrophils, mast cell degranulation, and fibrinogen adsorption. The degree of the inflammation is highly dependent upon the injury produced, the site of injection, the material used, and the extent of the provisional matrix formed. The acute phase is followed by a chronic phase inflammatory response, which is characterized by the presence of monocytes, lymphocytes, and plasma cells. After resolution of the acute and chronic phases of inflammation, granulation tissue formation and neovascularization in the new healing tissue ensue [32]. A prolonged inflammatory phase (i.e. longer than 3 weeks) should prompt an investigation to exclude complications, such as infection, allergic reaction, migration, abscess formation, or granulomatous reaction [33]. Foreign body granulomatous reactions have been reported with the use of hyaluronic acid, poly‐L‐lactic acid, silicone, and other fillers [34]. Biofilm formation, protein impurities, and microsphere irregularities have been implicated in causing local granulomatous reactions to fillers [33–35]. Granulomatous reactions are delayed reactions that occur as a result of ineffective phagocytosis. An organized collection of epithelioid macrophages wall off the foreign material with a surrounding infiltrate of lymphocytes. The surrounding lymphocytes secrete a variety of cytokines that cause ongoing macrophage activation and recruitment of inflammatory cells [36]. Clinically, foreign body granulomatous reactions develop over a variable period of time ranging from 5 months to 15 years [37]. Clinical manifestations can include swelling and indurated papules, nodules, or plaques (with or without ulceration). Although rare, granulomatous reactions have been reported to migrate beyond the injection site [38]. In the clinical setting, substances that have irritant properties are used to treat acne, hyperpigmentation, and sun‐damaged skin. The depth of penetration varies with the agent, the concentration of the agent, and the time of exposure. A myriad of “peeling” agents include retinoic, glycolic, trichloroacetic and salicylic acids, resorcinol, and phenol. Irritant reactions and photosensitivity manifested by erythema, scaling, and irritation can also occur following the use of topical retinoids. Decreasing the frequency of application and applying a barrier moisturizer along with a sunscreen may improve tolerance [39]. A wide variety of substances can act as irritants given sufficient exposure time and/or concentration (Table 6.1). Mechanical, chemical, and environmental factors can act alone or in combination to produce skin irritation. Mechanical factors include cosmetic procedures (shaving, waxing, laser therapy, cleansing, dermabrasion), personal habits (excessive rubbing of the skin with soaps, scrubs, wearing tight clothes or shoes, intense exercise), and occupational exposure (latex gloves, microtrauma of the skin). Wet work (i.e. skin exposure to liquids or use of occlusive gloves for longer than two hours per day or frequent hand cleaning) is one of the most common and important causes of skin irritation [40]. Professions at risk include hairdressers, healthcare workers, and food handlers. The list of the chemical compounds capable of producing irritation of the skin is extensive. Some substances are considered universal irritants. For example, strong acids (hydrofluoric, hydrochloric, sulfuric, nitric acids) and strong caustics (sodium hydroxide, potassium hydroxide) produce severe burns even in brief and small exposures. Solvents, including alcohol, turpentine, ketones, and xylene, remove lipids from the skin, producing direct irritation and facilitating irritant reactions from other substances such as soap and water. Inappropriate skin cleansing with solvents to remove grease, paints, or oils is a common cause of skin irritation. Alkali substances such as soaps are more likely to produce irritation by disrupting the skin barrier, whereas cleansing agents with a pH of approximately 5.5 and alcohol‐based hand‐cleansing gels are less irritating and preferred for sensitive skin. Environmental elements such as dry air, extremes of temperature, and weather variations may render the skin more susceptible to cutaneous irritants. Food allergies may cause urticarial reactions; spicy foods and condiments may cause lip and perioral irritant dermatitis. Prolonged exposure to water can cause maceration and desiccation of the skin. Follicular plugging has been reported secondary to use of isopropyl myristate, an emollient and lubricant used in shaving lotions, shampoos, oils, and deodorants [41]. Sodium lauryl sulfate (SLS), a surfactant found in many topical medications including those for acne, has also been used to study the effects of sequential exposure to irritants on skin barrier function [42]. Subjective irritation, described as a tingling, burning, stinging, or itching sensation without visible skin alteration is also observed following application of topical medications. Propylene glycol, hydroxy acids, and ethanol are capable of eliciting sensory irritation in susceptible individuals. Commonly used medications such as benzoic acid, azelaic acid, lactic acid, benzoyl peroxide, mequinol, and tretinoin can elicit sensory irritation. Sorbic acid is an organic compound used as a preservative in concentrations up to 0.2% in foods, cosmetics, and drugs. Subjective irritation has been demonstrated with 0.5% sorbic acid and to 1% benzoic acid in susceptible individuals [43]. Table 6.1 List of common skin irritants: mechanical, chemical, and environmental factors known to cause irritant dermatitis. The agents can act alone or in combination to produce contact dermatitis, therefore recognition of all factors involved is crucial for proper management of patients. “Sensitive skin” or cosmetic intolerance syndrome is a condition of cutaneous hyperreactivity secondary to substances that are not defined as irritants [44, 45]. The condition is a diagnosis of exclusion, encompassing a complex combination of objective and subjective irritative symptoms and may coexist with underlying allergic processes, urticarial reactions, and/or photodermatitis. Endogenous causes include seborrheic dermatitis, psoriasis, rosacea/perioral dermatitis, atopic dermatitis, and body dysmorphia. Elimination of all cosmetic products for an extended period of time (6–12 months) followed by slow reintroduction (a new product every 2–3 weeks) is helpful when managing cosmetic intolerance syndrome. Fragrances are a common cause of cosmetic dermatitis. A single perfume may include 100 different chemicals. Allergic reactions to fragrances affect 1.7–4.1% of the general population [46]. The eruption can be restricted to areas of application (face, neck, hands, axillae) or it can present as a generalized dermatitis. Myroxylon pereira (balsam of Peru) should be considered as a potential allergen in patients presenting with a generalized dermatitis [47]. Products containing scents are ubiquitous and include cosmetics, topical medications, toiletries, cleansers, and household cleaning products. The cosmetic products most commonly associated with allergy to fragrances in descending order include the leave‐on products deodorants, scented lotions, and fine fragrances [48, 49]. In 2005, the European Union mandated that 26 fragrance ingredients known to cause contact allergy be listed individually on the label of cosmetic products sold in Europe [50]. More than 5500 European products sampled in 2018 had at least one of the 26 sensitizing fragrances listed on the label, with limonene and linalool most frequently listed [51]. Only eight of these fragrance ingredients are included in fragrance mix I (FM I) and six in fragrance mix II (FM II) (Table 6.2). Patch testing to these 26 fragrances was performed to investigate the frequency of sensitization to these allergens and to assess their importance as screening markers for fragrance hypersensitivity [50]. Patch testing with FM 1, FM 2, balsam of Peru, and hydroxyisohexyl 3‐cyclohexene carboxaldehyde (HICC) in the European baseline series failed to detect 12% of those with fragrance hypersensitivity. Nonimmunologic contact urticaria reactions can also be triggered by fragrances that contain cinnamaldehyde, a pale yellow, viscous liquid extracted from the oil of cinnamon, or from balsams such as styrax. According to the North American Contact Dermatitis Group (NACDG), the prevalence of positive patch test reactions to fragrances in the United States is approximately 20%, with positive patch test results to FM I in 8.5% of cases, M. pereira in 7.2% of cases, and FM II in 4.7% of cases [52]. The prevalence of fragrance sensitivity has increased over recent years, particularly among patch‐tested patients with reported facial dermatitis [53]. Table 6.2 List of the 26 fragrances that must be specified on the labels of cosmetic products, according to the seventh amendment to the European Directive on Cosmetic Products. a Fragrance ingredients constituting fragrance mix I. b Fragrance ingredients constituting fragrance mix II. Linalool and limonene, which offer a fresh, flowery, or citrus scent, are two increasingly relevant contact allergens, yet neither are included on the FM I or FM II screening tests. Linalool is detectable in 88% of essential oils, including lavender, ylang‐ylang, tea tree, and rosemary oils [54]. Both are weakly allergenic in their inert forms but can oxidize into potent, allergenic hydroperoxides upon exposure to air [55]. This is a relevant concern given their use in cosmetic fragrances, which are dispensed to the skin via aerosolization. Patch testing for FM I, FM II, M. pereira (balsam of Peru), and colophony fail to detect more than half of patients with sensitivities to linalool and D‐limonene. Recognizing this limitation, both linalool and D‐limonene have been added to expanded patch test series, including an oxidized preparation. Preservative allergy is a common cause of skin care product allergy [56]. Preservatives are low molecular weight, biologically active compounds that protect against microbial growth in food, cosmetic, and industrial products. As a group, preservatives are sensitizing, representing 5 of the top 20 patch‐tested allergens in North America [57]. The shift from organic solvents and mineral oils to water‐based products in the cosmetic industry has increased the need for preservatives. ACD due to preservatives most commonly affects the face, neck, hands, and axillae but also can occur in a generalized distribution. Formaldehyde allergy is common and is most often caused by formaldehyde‐releasing biocides in cosmetics, toiletries, and other products. In the United States, approximately 20% of cosmetics and personal care products contain a formaldehyde‐releaser (quaternium‐15, imidazolidinyl urea, diazolinyl urea, DMDM–hydantoin, and 2‐bromo‐2‐nitropropane‐1,3,‐diol) [58]. In Europe, formaldehyde releasers are contained in approximately 8% of cosmetic products, often in combination with other preservatives such as parabens and phenoxyethanol [59]. In a review of 81 formaldehyde‐allergic patients, allergic reaction to at least one of the 12 formaldehyde‐releasing substances was detected in 79% of the cases and isolated reactions to releasers were rare [60]. Formaldehyde allergy is also reported as a common cause of occupational contact dermatitis. The professions at risk include hairdressers, healthcare workers, painters, photographers, housekeeping personnel, metalworkers, masseurs, and workers dealing with creams, liquid soaps, and detergents [60]. The nonformaldehyde preservatives methylchloroisothiazolinone (MCI), methylisothiazolinone (MI), iodopropynyl butylcarbamate (IPBC), and methyldibromoglutaronitrile (MDGN) are also important contact allergens. A common preservative in some brands of moist toilet paper (baby wipes and moist towelettes), MCI/MI can cause perianal and perineal ACD [61]. MCI/MI in fixed combination (MCI/MI at a ratio of 3:1) is widely used for the preservation of aqueous systems in cosmetics, toiletries, and several industrial applications. Despite European restrictions on the use of the mixture of MCI and MI the frequency of positive patch test reactions to MCI/MI has not decreased [62]. Factors that increase the risk of sensitization include chronic dermatitis and the frequent use of leave‐on products containing MCI/MI [63]. MI without MCI has also increasingly been used as a preservative in cosmetics and skin care products [64, 65]. MI alone is a less effective antimicrobial agent in the absence of MCI, thus cosmetic products incorporate it at higher concentrations, with greater risk for sensitization. The number of MI‐only‐containing cosmetic products doubled between 2007 and 2010 [66]. MI now dominates as the leading preservative contact allergen, and the second most prevalent positive patch test allergen in North America (after nickel) [57]. Standard patch testing screening misses approximately 33–60% cases of allergy to MI likely due to the low concentration of MI in the MCI/MI combination patch test []. Patch testing MI alone at its higher concentration (e.g. 0.2%) is necessary to detect an allergy [69]. Parabens are also one of the most frequently used preservatives in cosmetics. Parabens are present in approximately 40% of European cosmetic products, including make‐up, facemasks, hair cleansing products, and liquid soaps [59, 70]. Despite parabens’ common use and consumers’ high‐level awareness of them, these compounds are weak sensitizers and uncommon causes of ACD, with the most recent NACDG rates of positive patch testing 0.6%, amongst the lowest in 25 years [57, 71]. Some patients sensitized to paraben‐containing medications can use cosmetic products containing parabens with no adverse effects [72]. However, parabens may have a greater sensitization potential when used on previously damaged or broken skin. This phenomenon is known as the paraben paradox. A multicenter study in patients suffering from chronic leg ulcers suggested paraben sensitivity in 3% of patients [73]. Preservatives are also commonly implicated in allergy to ophthalmic preparations. Phenylmercuric acetate has been implicated as a leading preservative allergen, although it has been suggested that positive patch tests are due to an irritant reaction rather than true contact allergy [74]. In contact lens solutions, previously thimerosal was one of the most frequent positive patch‐tested preservatives. However, it has been largely replaced by benzalkonium chloride (BAK) [75]. Eyelid dermatitis can be triggered by allergens in ophthalmic products but occurs more commonly as a result of sensitization to environmental allergens [74]. Young females with skin atopy and allergies to cosmetic and skin care products, as well as older subjects with allergy to topical medications are particularly at risk for ACD in the periorbital area [74]. The thinness of periorbital skin makes it particularly susceptible to allergens. There is widespread use of plant‐based botanicals and essential oils in topical cosmetics and therapeutic preparations owing to their natural origins and thus perceived safety. Their expanded incorporation into topical products increases the likelihood of exposure to a variety of potential herbal allergens. Botanicals commonly implicated in cosmetic contact allergy include the Compositae family plants, propolis (honeybee hive derivative), lichens, henna, as well as tea tree, ylang‐ylang, lavender, jasmine, sandalwood, peppermint oils, among others [76–78]. Some plant‐derived compounds have the potential to cause phototoxic effects; for example, bergapten (aka 5‐methoxysporalen) is a naturally occurring furanocoumarin in bergamot oil that causes phototoxicity [3]. While the International Fragrance Association has set limits on the amount of bergamot oil allowed in leave‐on products applied to sun‐exposed skin, bergamot essential oil remains one of the most widely used ingredients in modern perfumery and there are no restrictions for bergamot in rinse‐off products [79]. Bergapten and psoralens in fig leaves used to prepare a homemade tanning lotion have also been reported to cause phytophotodermatitis [80, 81]. Washout products are in contact with the skin for brief periods, making detection of allergy to these agents challenging. Cleansers are applied to remove sebum, desquamated cells, sweat, and microorganisms. Allergens in body‐cleansing products and facial cleansers include fragrances, preservatives, surfactants (cocamidopropylbetaine, alkyl and decyl glucosides, laureth/pareth sulfates, oleamidopropyl dimethylamine), and vehicle components (lanolin alcohols) [82, 83]. Allergic reactions to rinse‐off products are less common compared to leave‐on products because of the limited amount of time the substance is in contact with the skin. However, cocamidopropylbetaine (surfactant), formaldehyde, and MCI/MI have been reported as causative agents of ACD in rinse‐off agents. Moreover, weaker sensitizers preferentially incorporated into “hypoallergenic” cleansers have been reported to cause contact allergy [84].
CHAPTER 6
Contact Dermatitis and Topical Agents
Introduction
Pathophysiology and clinical presentation
Irritant contact dermatitis
Allergic contact dermatitis
Phototoxic dermatitis
Contact urticaria
Foreign body reactions
Common irritants and allergen groups
Irritants
Mechanical
Chemical
Environmental
Shaving
Water (wet work)
Excessive heat or sun exposure
Waxing
Alkalis (soaps and cleansers)
Food handling
Laser treatment
Detergents
Saunas and jacuzzis (chlorine)
Abrasive scrubs
Surfactants (sodium lauryl sulfate)
Extreme cold and windburn
Dermabrasion
Whitening agents
Stress
Microdermabrasion
Acids (salicylic, glycolic, trichloroacetic acid)
Dry air
Rubbing of the skin (e.g. when using a soap or scrubbing)
Fragrances and color additives
Hot and/or prolonged showers
Skin cleansing tools (face brushes, synthetic sponges, loofahs)
Sunscreens
Spicy foods, peppers, condiments
Closed‐weave face cloths
Oxidizing agents (sodium hypochlorite (bleach) or benzoyl peroxide)
Increased humidity (e.g. sweating from prolonged wearing of occlusive gloves)
Friction (wool, synthetic fibers, or jewelry)
Solvents (benzene, toluene, acetone, alcohol)
Low humidity (air conditioning)
Intense exercise
Vitamin A derivatives (retinoids, retinol)
Plant parts (thorns, spines, and sharp‐edged leaves)
Microtrauma (fiberglass)
Pressure (spectacle frames)
Occlusion tight clothes, rubber/latex gloves)
Fragrances
International Nomenclature of Cosmetic Ingredients Name
α‐Isomethyl ionone
α‐Amyl cinnamaldehydea
Anisyl alcohol
Benzyl alcohol
Benzyl benzoate
Benzyl cinnamate
Benzyl salicylate
Butylphenylmethylpropional
Cinnamaldehydea
Cinnamylalcohola
Citralb
Citronellolb
Coumarinb
Eugenola
Everniafurfuracea (tree moss)
Everniaprunastria (oak moss)
Farnesolb
Geraniola
Hexyl cinnamalb
Hydroxycitronellala
Hydroxyisohexyl 3‐cyclohexane carboxaldehyde (HICC)b
Isoeugenola
Linalool
Limonene
Methyl 2‐octynoate
Preservatives
Botanicals
Specific cosmetic products
Cleansing agents
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree