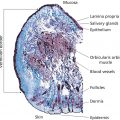
Duane Charbonneau Procter & Gamble Company, Global Bioscience, Mason, OH, USA Hand cleansers and hand sanitizers were developed to reduce transient microbes on the skin to help limit the spread of infectious disease. In fact, handwashing and appropriate hand hygiene are cornerstone of CDC recommendations for the prevention of transmissible infectious diseases. This class of products includes liquid and solid soaps, antimicrobial liquid and solid soaps, alcohol‐based hand sanitizers, and non‐alcohol‐based hand sanitizers. Growing concern exists about the spread of infectious disease. Despite historical expectations that such disease would wane in the United States over the course of the 20th century, between 1980 and 1992, the death rate due to infectious diseases increased 58% from 41 to 65 deaths per 100,000 population [1]. The cross‐species transfer of infectious viral agents to humans, which initiated the 2002–2003 outbreak of severe acute respiratory syndrome (SARS), the 2004 outbreak of H5N1‐linked avian influenza [2], and the 2009 US outbreak of H1N1‐linked porcine influenza [3], elevated concerns about the potential for human pandemics. In 2019, these concerns became a reality with the SARS‐CoV‐2 (COVID‐19) pandemic with tremendous numbers casualties in excess of 300,000 worldwide and greater than 100,000 in the United States alone [www.cdc.gov/coronavirus/2019‐ncov/covid‐data/forecasting‐us.html]. In the United States, about 1 in 6 people contract a foodborne illness each year [4], resulting in an estimated 52 million cases annually. A growing number of outbreaks have been associated with Campylobacter or Escherichia coli O157:H7 through consumption of contaminated meats and produce in the home. Half of all cases of norovirus, the most common cause of food‐borne illness in the United States, occur in long‐term care facilities [5]. Rotavirus, another viral cause of gastroenteritis, spreads by the fecal–oral route among children cared for in group settings [6]. Healthcare‐associated infections have gained attention as the leading cause of death in the US healthcare settings [7] and the fourth leading cause of death overall in Canada [8]. Pathogens once largely confined to healthcare facilities, such as methicillin‐resistant Staphylococcus aureus (MRSA) and Clostridium difficile (C. difficile), have now moved into the community. Multidrug resistant Acinetobacter baummanii has entered nursing homes and long‐term care facilities through discharge of hospital patients into these settings [9]. The emergence of these antibiotic‐resistant organisms limits the therapeutic arsenal available to combat infection; moreover, it has become economically less desirable for pharmaceutical companies to develop and register novel antibiotics, further limiting potential new therapeutics. Because of these concerns, reducing the burden of infectious disease must emphasize strategies to limit transmission. Good hand hygiene is sentinel to preventative measures, for the spread of infectious agents. Because hand contact plays such a crucial role in the transmission of infectious agents both for the individual and their contacts. Efficacious hand hygiene products are a vital component of preventive regimens. This chapter reviews the formulation, mode of action, and efficacy of hand cleansers and sanitizers, discusses regulatory and safety issues related to the use of antimicrobial ingredients, and proposes future directions for improving these products and technologies. The skin is an ecosystem coinhabited by symbiotic and commensal microbes collectively termed the skin microbiome [10]. Most skin bacteria fall into four dominant phyla – Actinobacteria, Firmicutes, Bacteriodetes, and Proteobacteria – but a great deal of diversity exists at the species and strain level [11]. Compared to the oral or gut microbiota, the skin microbiota shows the greatest variability over time [12]. Although a core set of taxa inhabits skin surfaces, considerable intraindividual and interindividual variability in skin microbiome composition exists, reflecting both host and environmental influences. Diversity is greater between individuals: only 13% of bacterial phylotypes on the surface of the palms are shared between unrelated people [13]. Members of the same household share more of their skin microbiota, particularly on the palms, than people from different households [14]. The diversity of microbial populations within households arise from various internal and external sources, but surfaces with high contact bear the signature of the occupants’ skin microbiome [15]. Microbes that inhabit the hand are generally divided into two categories: the transient and the resident microbiota (Figure 15.1). The transient microbiota are microbes that inadvertently become attached to the hands by touching contaminated materials and surfaces, including raw food items during food preparation, body fluids in healthcare or group care settings, or fomites in the daily environment. Microbes on the dominant and nondominant hands of the same individual are quite significantly different [13], most likely because the dominant hand comes into contact with different types of surfaces than the nondominant hand. Furthermore, the transfer of transient microbiota from hands to mouth or to other parts of the body, either in a single person or between individuals, is well documented in the literature. The classic example is the work of Gwaltney and others in the 1970s [16, 17], which demonstrated the importance of hand‐to‐hand transmission of the common cold virus. Figure 15.1 The common microbiota of the hand. The resident hand microbiome is composed of the community of microbes that consistently inhabit the hand and routinely are not washed off with nonmedicated soaps. A summary of bacteria reportedly isolated as resident flora is presented in Table 15.1. Handwashing alters overall resident community composition in a temporary, limited fashion, either because washing fails to remove most taxa on the skin surface (resident taxa) or because depleted communities rapidly reestablish themselves [13]. In a University population, taxa such as Staphylococcaceae, Streptococcaceae, and Lactobacillacaeae were relatively more abundant on hands that have been recently washed, whereas Proprionibacteria, Neisseriales, Burkholderiales, and Pasteurellaceae taxa became more abundant with longer times after handwashing [13]. Frequent exposure to certain transient microbes may lead to them becoming established as constituents of the resident microbiome. For example, nurses who perform similar tasks within a hospital share similarities in their resident microbiome but exhibit differences from those assigned to other tasks [18–21]. Furthermore, the resident hand microbiome of homemakers includes microbes that are identical to those environmental isolates identified within the home [22]. The functional role of the skin microbiome in health and disease is incompletely understood, but it is speculated that the resident skin microbes are as essential to the overall health of the skin as the gut microorganisms are to overall health of the individual [23]. Staphylococcus epidermidis, a commensal bacterium, maintains health by modulating the host immune response (reviewed in [24]). However, the organism is also the most common cause of hospital‐acquired infection, for example when virulent forms create biofilms on in‐dwelling medical devices such as catheters and heart valves [25]. Proprionibacterium acnes, a commensal skin bacterium, causes acne, an inflammatory disorder of the pilosebaceous unit that occurs when this structure matures during puberty. In addition, it is estimated that approximately 32% of the population carries the common pathogen, Staphylococcus aureus (S. aureus), as a member of the resident skin and nasal microbiome [23]. As many as 90% of patients with atopic dermatitis are colonized with S. aureus on lesion and nonlesion skin [26] and colonization rates are highest on acute lesions [27]. Treatment of patients with atopic dermatitis leads to a more diversified skin microbiome [28, 29]. Table 15.1 Constituents of the resident microbiota of the hand. The critical role of hand hygiene as means of infection control dates to the mid‐1800s, when the groundbreaking work of Professor Ignaz Semmelweis demonstrated a reduction in puerperal sepsis after initiating a mandatory handwashing policy for medical personnel [30]. By 1980, the role of hand transmission of bacterial and viral pathogens was well‐documented [16, 31]. Today, handwashing with soap and water, or hand antisepsis using antimicrobial washes or hand sanitizers, is paramount to infection control programs. Guidelines for handwashing and hand antisepsis in healthcare settings were published by the Association for Professionals in Infection Control (APIC) in 1988 and updated in 1995 [32]. In 2002, the Centers for Disease Control and Prevention (CDC) published guidelines for hand hygiene in healthcare settings in collaboration with the HICPAC/SHEA/APIC/IDSA hand hygiene task force [33]. This guideline recommends using alcohol‐based hand rubs routinely for hand antisepsis unless hands are visibly dirty, contaminated, or if exposure to spores is suspected; in such cases, washing with soap (nonantimicrobial or antimicrobial) and water is recommended. In 2009, the World Health Organization (WHO) published Guidelines for Hand Hygiene in Healthcare as part of their “Clean Care Is Safer Care” Initiative [34]. The guidelines are intended to apply to all healthcare settings, whether care is delivered continually (as in healthcare institutions), or occasionally (e.g. home care by birth attendants). WHO recommended the adoption of alcohol‐based hand rubs to rapidly and effectively inactivate a wide array of potentially harmful microorganisms on hands. Consistent with the earlier CDC guidelines, this body recommends the routine use of alcohol‐based hand rubs unless hands are visibly soiled or contaminated. Posters and brochures depicting optimal methods for using hand rubs or washing with soap and water (and the situations in which they apply) are available online [35]. The US Food and Drug Administration (FDA) Food Code, updated in 2013, contains specific hand hygiene guidance for retail and food service workers that describes when, where, and how to wash and sanitize hands [36]. The Food Code calls for proper handwashing with soap and water to control the transmission of enteric bacteria, enteric nonlipophilic viruses, and protozoan oocysts that may be encountered in settings where food is prepared. Alcohol‐based hand sanitizers are not effective enough against these particular microbes. Furthermore, fatty or proteinaceous soils encountered during food preparation may interfere with alcohol efficacy [37]. Consequently, hand sanitizers that meet specific criteria described in Section 2–301.16 of the Food Code may be used in retail and food service settings only after proper handwashing is used to remove soil/debris. When done properly, handwashing is considered to be the gold standard for removing transient pathogenic bacteria from the hands. The CDC has published a description of handwashing technique for healthcare settings in both text and pictorial poster format [38]. The CDC also promulgates general handwashing advice aimed at homes, schools, and businesses as part of a “Handwashing: Clean Hands Save Lives” initiative as well as prevention of COVID‐19 [39]. The handwashing protocol for healthcare providers provides explicit descriptions for each step, including activating the faucet, the modes of scrubbing all aspects or the dorsal and palmar surfaces and fingers, drying the hands, and shutting off the faucet. It specifies that the full procedure should have a 60 second duration from start to finish. The description aimed at consumers is consistent with this healthcare protocol, but uses much simpler instructions, being most explicit about lathering and scrubbing for 20 seconds (see Table 15.2 for comparison). The basic elements of proper handwashing are as follows: Although healthcare professionals recognize the importance of handwashing, observational studies indicate that compliance is poor. A CDC review of available studies found that the baseline level of compliance with hand hygiene guidelines among healthcare workers averaged 40% (range 5–81%) [33]. Handwashing compliance also tends to fall to lower levels at night [40], and fewer than 50% of hospital healthcare workers washed their hands after toileting [41]. A similar deficiency exists in the community. An observational study of food service workers found that 30% were compliant with standard guidelines [42]. Studies in public restrooms on a university campus indicated that 61% of the women and 37% of the men observed washed their hands with soap in the absence of a visual prompt; 97% of the women and 35% of the men washed their hands in the presence of a sign prompting handwashing [43]. When college students were evaluated in four restroom settings (presence of soap and water; soap and water and visual prompts; soap and water and hand sanitizers; or soap and water, hand sanitizers, and visual prompts), 72.9% of students either washed or just rinsed their hands, 58.3% practiced hand hygiene (using either soap or hand sanitizer), and 26.1% washed their hands for the adequate length of time [44]. Table 15.2 Comparison of hand hygiene instructions for healthcare workers and consumers. a Centers for Disease Control/World Health Organization. Hand Hygiene in Health Care Settings. Hand Hygiene Basics. 2012. www.cdc.gov/handhygiene/Basics.html. b Centers for Disease Control and Prevention. Hand Washing: Clean Hands Save Lives. When and How to Wash Your Hands. 2013. www.cdc.gov/handwashing/when‐how‐handwashing.html. The duration of handwashing averages about 8–9 seconds in both healthcare and community settings [45, 46]. These conditions are not ideal, as evidenced by studies showing mean cell densities on the hands of homemakers to be 5.72 CFU before washing and a 5.69 CFU after handwashing indicating that the effectiveness of handwashing was limited [47]. Compliance with handwashing guidelines at a teaching hospital was shown to be inversely proportional to education levels, so understanding the guidelines is not the issue [48]. In certain situations where handwashing compliance is low, hand sanitizers, especially those with a persistent benefit, may be considered to promote hand hygiene if appropriate to the setting in question. However, merely providing the product is insufficient. University students typically do not use hand sanitizers when they are installed in restrooms without any reinforcement of their use [44, 49]. However, prospective trials have shown that when hand sanitizers were provided in elementary school classrooms along with appropriate education about their use, or when sanitizers were used in the classroom as an adjunct to handwashing, intervention groups had significantly lower absenteeism due to illness [50–52]. Formulation choices must be appropriate to the target pathogens of concern. For example, alcohol‐based hand sanitizers may exhibit limited effectiveness against norovirus unless alcohol concentrations are high enough or the formula is supplemented with other actives [53, 54]. It bears repeating that substituting alcohol‐based hand sanitizers for soap and water cleansing is inappropriate in food service settings; however, regimens that combine handwashing with the subsequent application of high efficacy hand sanitizers can be very effective in reducing hand contamination in food handling situations [55]. Antimicrobial handwashes are primarily water‐based formulations composed of mixtures of surfactants, antimicrobial actives, perfumes, and, in some cases, emollients. Because frequent use of surfactants can be drying to the skin, emollients and other agents to improve skin feel are incorporated to improve the consumer experience with the hope of raising compliance. In the United States, antimicrobial actives for handwash and handrub products are regulated under the Food and Drug Administration. In the 1994, Tentative Final Monograph (TFM) for over‐the‐counter (OTC) antiseptic drug products [56]. The ingredients are classified into three categories: Under the 1994, TFM only active ingredients in categories I and III could be lawfully marketed in products within the United States. From 2013 to 2016 several changes were to the Monograph made including separation of the Health Care and Consumer products, requests for additional safety and efficacy data were requested for GRAS and GRAE determinations by FDA. In 2015 (FDA‐2016‐N‐0124), alcohol 60–95% previous Category I was reclassified as Category IIISE indicating that the FDA required additional efficacy data for acceptance of GRAE claims. In 2016 (FDA‐1975‐N‐0012), the Consumer Antiseptic Final Monograph was issued (21 CFR Part 310). In the 2016 document, Category I distinction was replace with “monograph conditions” and Category II and Category II were replaced with “nonmonograph conditions.” As a result, several of the actives previously utilized in antimicrobial handwash formulations were no longer allowed. Three actives were deferred until further safety and efficacy data could be collected this included benzalkonium chloride, benzethonium chloride, and chloroxylenol. Antimicrobial soaps vary in the nature of the active ingredient as well as their overall formulation. Each antiseptic agent has a distinct range of activity against various bacteria and viruses (reviewed in [33, 34]). Apart from iodophors (at concentrations higher than used in antimicrobial washes or handrubs), neither alcohol nor any of the agents listed above are reliably efficacious against spore‐forming bacteria, such as against Clostridium spp. or Bacillus spp. [57]. Glove use and strict adherence to recommended handwashing protocols are critical if there is reason to suspect contamination with such microbes. Hand sanitizers can be categorized into three main classes: Although the 1994 TFM placed isopropanol 70–91.3% in category III (i.e. insufficient data to classify as effective), 60% isopropanol has subsequently been adopted in Europe as the reference standard against which alcohol‐based hand‐rub products are benchmarked [58]. Hand sanitizer product forms include liquids, gels, and foams. Germicidal efficacy is based on the fact that they are leave‐on products that rapidly kill microbes. Hand sanitizers based solely on alcohol as the antiseptic deliver an immediate benefit but provide no residual activity. Supplementation of the alcohol‐based formula with additional antimicrobial actives can provide more persistent activity, delaying the reestablishment of transient microorganisms. Other combinations have also proved efficacious. For example, a synergistic combination of a humectant (octoxyglycerine) and preservatives has resulted in prolonged activity of an alcohol‐based handrub against transient pathogens without raising the concentration of antimicrobial active [59]. An experimental alcohol‐based surgical hand disinfectant containing a synergistic combination of farnesol and benzethonium chloride demonstrated both immediate and persistent activity against resident hand flora of volunteers [60]. In its 2009 guidelines, the WHO recommended two alcohol‐based handrub formulations for hand hygiene in healthcare settings, taking into account cost and efficacy for resource‐poor countries. The first has final concentrations of ethanol 80% v/v, glycerol 1.45% v/v, and hydrogen peroxide (H2O2) 0.125% v/v; the second has final concentrations of isopropyl alcohol 75% v/v, glycerol 1.45% v/v, and hydrogen peroxide 0.125% v/v. However, these formulations did not meet the most stringent European efficacy requirements for surgical hand disinfection (the prEN 12791 standard). Proposed modifications (raising alcohol content, reducing glycerol, modifying rub times) raise efficacy to the required standards [61–63]. The formulation of non‐alcohol‐based hand sanitizers or antimicrobial handwashes must take into consideration the bioavailability of the antimicrobial active. Certain surfactants may complex with or otherwise inactivate the antiseptic, which results in products with the same antimicrobial active having different levels of antimicrobial efficacy [64]. The final variable to be considered in formulating antimicrobial handwashes and hand rubs are pH. The relatively acidic pH of the skin plays a role in the innate antimicrobial hostility of the hand surface. The normal pH of the hands is approximately 4–5, but alkaline soaps often raise the skin pH. In one report, pH increased between 0.6 and 1.8 units after handwashing with plain soap and then gradually declined to baseline levels over a period of 45 minutes to 2 hours [65]. Historically, three approaches have traditionally been used to demonstrate the efficacy of antimicrobial hand soaps and hand sanitizers: In 2016, the FDA issued a rule to update the conditions for establishing that OTC antimicrobial products are generally recognized as safe and effective (GRASE) [66]. The proposal requires manufacturers of antimicrobial hand or body washes to demonstrate that the products are more effective than plain soap and water in preventing illness and the spread of infection in a community setting. Currently, the regulations rely on in vivo models of germicidal activity to determine efficacy. The does not include over‐the‐counter antiseptic hand sanitizers or antiseptics used by healthcare professionals [66]. Standard in vitro assays evaluate the minimum inhibitory concentration (MIC) for germicidal activity or assess the time to kill target organisms, using indicator bacteria or viruses of interest. The research community has debated the relevance of such assays for decades, as they describe the potency and spectrum of activity of a formulation or antiseptic under very specific laboratory conditions. Other in vitro
CHAPTER 15
Hand Cleansers and Sanitizers
Introduction
Hand microbiome
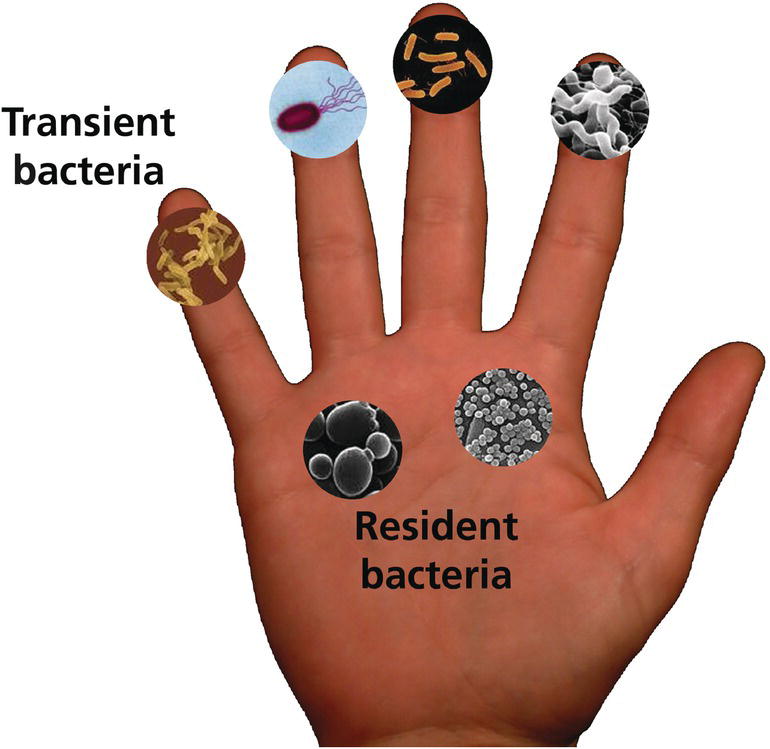
Organism
Acinetobacter baumannii
Acinetobacter johnsonii
Acinetobacter lwoffii
Corynebacterium spp.
Enterobacter agglomerans
Enterobacter cloacae
Klebsiella pneumoniae
Propionibacterium acnes
Pseudomonas aeruginosa
Staphylococcus aureus
Staphylococcus epidermidis
Staphylococcus warneri
Streptococcus mitis
Streptococcus pyogenes
Hand hygiene guidelines
Hand hygiene techniques and compliance
Hand hygiene in healthcare settings a
Hand hygiene consumer guidelines b
Wet hands with water
Wet your hands with clean, running water (warm or cold), turn off the tap, and apply soap
Apply enough soap to cover all hand surfaces
Lather your hands by rubbing them together with the soap. Be sure to lather the backs of your hands, between your fingers, and under your nails
Rub hands palm to palm
Scrub your hands for at least 20 seconds. Need a timer? Hum the “Happy Birthday” song from beginning to end twice
Right palm over left dorsum with interlaced fingers and vice versa
Rinse your hands well under clean, running water
Palm to palm with fingers interlaced
Dry your hands using a clean towel or air dry them
Backs of fingers to opposing palms with fingers interlocked
Rotational rubbing of left thumb clasped in right palm and vice versa
Rotational rubbing, backward and forwards with clasped fingers of right hand in left palm and vice versa
Rinse hands with water
Dry hands thoroughly with a single‐use towel
Use towel to turn off faucet
Your hands are now safe
Antimicrobial handwash and hand sanitizer formulations
Efficacy of antimicrobial handwashes and hand sanitizers
In vitro assays for potency and spectrumof activity
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree