Diagnosis.
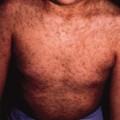
To make a diagnosis of cutaneous tuberculosis, a high index of suspicion is required when considering the aetiology of chronic eruptions in children. The history and epidemiological circumstances will be helpful, as will results of tuberculin testing and chest radiography. The important differential diagnosis to consider is whether the cutaneous lesions are caused by one of the atypical mycobacteria, such as M. marinum, M. kansasii, M. fortuitum or M. chelonae (see below). The following should also be considered in the differential diagnosis: impetigo, cat-scratch disease, sporotrichosis, syphilis and tularaemia. In those patients coming from endemic areas, cutaneous leishmaniasis, leprosy and cutaneous diphtheria also need to be considered.
A definitive diagnosis of cutaneous tuberculosis will require biopsy of a lesion; histopathology examination may reveal epithelioid granulomas and/or the presence of acid-fast bacilli. Subsequent mycobacterial cultures would identify M. tuberculosis as the aetiological agent. Culture media for M. tuberculosis include Lowenstein–Jensen, which requires 6–8 weeks for growth; Bactec systems can detect M. tuberculosis within 2 weeks but are more useful for culturing blood, urine and respiratory secretions. In general, the culture yield from tissues in children with extrapulmonary tuberculosis is around 50%, due to the paucibacillary nature of the disease [18]. Microscopy using Ziehl–Neelsen staining can detect 40–60% of culture-positive samples, with a lower limit of detection of 5000 organisms per millilitre. Newer techniques, such as fluorescent staining of samples with auramine or rhodamine, are superior to the Ziehl–Neelsen stain. The amplification of small amounts of bacterial nucleic acid, using techniques such as polymerase chain reaction (PCR), allows the detection of mycobacteria directly from clinical specimens. Several studies in children have found the PCR test on clinical samples to have a sensitivity of 40–60%, which compares favourably with standard culture [19]. Polymerase chain reation is especially useful in making a rapid diagnosis in the paucibacillary form of cutaneous tuberculosis [20].
Recently new diagnostic blood tests for tuberculosis have become commercially available. These tests, termed interferon-gamma release assays (IGRA), either measure the amount of interferon-gamma released by TB-specific T-lymphocytes or enumerate these cells. Although these tests are considered superior to tuberculin skin testing and do not require follow-up, clinical experience in children with extrapulmonary tuberculosis, such as cutaneous diseases, is limited. A high positive predictive value was observed for IGRA in diagnosing extrapulmonary tuberculosis [21].
Treatment.
There are no clinical efficacy trials for the treatment of extrapulmonary tuberculosis in children; however, most authorities recommend standard short-course chemotherapy for treatment of cutaneous tuberculosis. This involves the administration of four antituberculous drugs for the first 2 months (isoniazid 10 mg/kg, rifampicin 10 mg/kg, pyrazinamide 35 mg/kg, ethambutol 15 mg/kg), followed by 4 months of isoniazid and rifampicin [22,23]. Ethambutol has been associated with retrobulbar neuritis, although Trebucq [24] reviewed the literature and concluded that ethambutol, at a dose of 15 mg/kg/day, was safe in children older than 5 years, and in younger children. It is appropriate to obtain a baseline ophthalmological assessment in younger children before starting therapy, and repeat the assessment after 1–2 months. In older children, routine colour vision and visual acuity should be assessed at follow-ups using special charts. Treatment should be started as soon as the diagnosis is thought to be a definite possibility, based on the epidemiological history, clinical examination and the results of Mantoux testing, chest radiograph and skin biopsy. Results of drug susceptibility testing should become available within the first 2 months of the treatment period, and hence appropriate changes can be made to the antituberculous regimen if necessary. If besides cutaneous tuberculosis there is initial evidence of meningeal involvement, total treatment should be for 12 months. Local complications following BCG vaccination (e.g. excessive ulceration, subcutaneous abscess) can probably be treated with short courses of isoniazid.
References
1 Lincoln EM, Sewell EM. Tuberculosis in Children. New York: McGraw-Hill, 1963.
2 Beyt BE Jr, Ortbals DW, Santa Cruz DJ et al. Cutaneous mycobacteriosis: analysis of 34 cases with a new classification of disease. Medicine 1981;60:95–109.
3 Hill MK, Sanders CV. Cutaneous tuberculosis. In: Schlossberg D (ed.) Tuberculosis, 4th edn. Philadelphia: W.B. Saunders, 1999: 264–70.
4 Pereira CA, Webber B, Orson JM. Primary tuberculous complex of the skin. J Am Med Assoc 1976;235:942–6.
5 Miller FJW. Tuberculosis in Children. Edinburgh: Churchill Livingstone, 1982:144–55.
6 Hole LE. Tuberculosis acquired through ritual circumcision. J Am Med Assoc 1913;61:99.
7 Sehgal VN, Wagh SA. Cutaneous tuberculosis: current concepts. Int J Dermatol 1990;29:237–40.
8 Montgomery H. Histopathology of various types of cutaneous tuberculosis. Arch Dermatol 1961;35:698–702.
9 Minkowitz S, Brandt IJ, Rapp Y et al. Prosector’s wart (cutaneous tuberculosis) in a medical student. Am J Clin Pathol 1969;51:260–2.
10 Dennehy PH, Peter G. Active immunizing agents. In: Feigin RD, Cherry JD, Demmler G, Kaplan SL (eds) Textbook of Pediatric Infectious Diseases, 5th edn. Philadelphia: W.B. Saunders, 2004:3136–82.
11 Gonzalez B, Moreno S, Burdach R et al. Clinical presentation of bacillus Calmette–Guérin infections in patients with immunodeficiency syndromes. Pediatr Infect Dis J 1989;8:201–6.
12 Kumar B, Rai R, Kaur I et al. Childhood cutaneous tuberculosis; a study over 25 years from northern India. Int J Dermatol 2000;40:26–32.
13 Nepomuceno OR, O’Grady JF, Eisenberg SW et al. Tuberculosis of the anal canal: report of a case. Dis Colon Rectum 1971;14:313–15.
14 Kennedy C, Knowles GK. Miliary tuberculosis presenting with skin lesions. Br Med J 1975;3:356.
15 Sloan JB. Papulonecrotic tuberculid in a 9-year-old American girl: case report and review of the literature. Pediatr Dermatol 1990;7:191–5.
16 Ward AS. Superficial abscess formation: an unusual presenting feature of tuberculosis. Br J Surg 1971;58:540.
17 Miller ML. Evaluation of suspected rheumatic disease. In: Kliegman RM, Behrman RE, Jensen HB, Stanton BF (eds) Nelson Textbook of Pediatrics, 18th edn. Philadelphia: W.B. Saunders, 2007:995–7.
18 Shingadia D, Novelli V. Diagnosis and treatment of tuberculosis in children. Lancet Infect Dis 2003;3:624–32.
19 Smith KC, Starke JR, Eisenach K et al. Detection of Mycobacterium tuberculosis in clinical specimens from children using a polymerase chain reaction. Pediatrics 1996;97:155–60.
20 Hsiao PF, Tzen CY, Chen HC et al. Polymerase chain reaction based detection of mycobacterial tuberculosis in tissues showing granulomatous inflammation without demonstrable acid-fast bacilli. Int J Dermatol 2003;42:281–6
21 Winqvist N, Bjorkman P, Noren A, Miorner H. Use of a T cell interferon gamma release assay in the investigation for suspected active tuberculosis in a low prevalence area. BMC Infect Dis 2009;9:105.
22 American Academy of Pediatrics. Tuberculosis. In: Pickering L, Baker CJ (eds) Red Book: Report of the Committee on Infectious Diseases, 28th edn. ELK Grove Village: American Academy of Pediatrics, 2009.
23 National Institute of Clinical Excellence (NICE). Tuberculosis: Clinical Diagnosis and Management of Tuberculosis, and Measures for its Prevention and Control. London: Royal College of Physicians of London, 2006.
24 Trebucq A. Should ethambutol be recommended for routine treatment of tuberculosis in children? A review of the literature. Int J Tuber Lung Dis 1997;1:12–15.
Non-Tuberculous Mycobacterial Infections
Non-tuberculous mycobacteria (NTM), also known as atypical mycobacteria, mycobacteria other than tuberculosis and anonymous mycobacteria, are acid-fast bacilli that are found in our environment, in dust, soil and water (taps, water tanks and swimming pools), in foodstuffs, and in both wild and domestic animals [1]. Studies in the USA have estimated the prevalence of NTM to be around 20% that for tuberculosis (1–2 per 100,000). Transmission of NTM occurs from these environmental sources, aerosols and animals; however, there is no evidence of person-to-person transmission. Nosocomial outbreaks (usually by rapid growers) have been reported following surgical procedures, for example sternal wound infections after cardiac surgery, respiratory manipulations and intravenous catheter insertion [2]. Infections have also occurred, iatrogenically, after vaccination with infected needles [3]. In recent years, NTMs have represented an increasing problem in industrialized countries. This situation has been exacerbated by the AIDS pandemic, which has led to infections, especially from the M. avium-intracellulare complex (MAIC), occurring quite frequently in patients with HIV and very low CD4 counts. It may be difficult to distinguish disease from colonization, as isolating NTM from a clinical specimen does not necessarily indicate it is causing disease. Although most reports have concentrated on the pathogenicity of these organisms in immunocompromised hosts, they continue to be uncommon but important pathogens in children with intact immune systems [4]. In contrast with adults and immunocompromised individuals in whom pulmonary and bone involvement is common, the most frequent sites for NTM infection in children are the skin and lymph nodes. Indeed, in developed countries the NTM are causing an increasing proportion of all mycobacterial infections in children and cause skin infections much more commonly than M. tuberculosis [5].
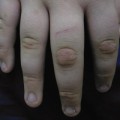
Cervical lymphadenitis is the most common non-tuberculous mycobacterial infection in immunocompetent children, with the majority of cases occurring in children under 5 years of age. In contrast with tuberculous adenitis, there is usually unilateral lymph node swelling (most commonly submandibular) and there are no clinical or radiological manifestations of pulmonary disease. The node and its overlying skin are seldom painful to the child. Because of the chronicity of the infection, the skin overlying the infected node(s) is indurated, with a dark-red to purplish hue; often the affected node may be fluctuant or there may be a draining sinus present.
Cutaneous infections caused by the NTM are less common than adenitis but are often more difficult to diagnose and require the paediatrician to have a high index of suspicion. They are generally indolent and present as non-healing lesions, which are often not diagnosed until the patient has failed several attempts at treatment.
In 1969, Wayne and Runyon [6] published a classification for mycobacteria, based on their rate of growth and pigmentation (see Table 57.1). Most infections in humans involve either group I or III. The slow-growing mycobacteria (group I) cause the most cutaneous disease. Antigenic analysis and DNA studies have shown that the rapid growers and slow growers are two distinct subgenera that probably split very early in the evolution of the genus. The rapid growers show significant overlap with the genus Nocardia. As NTM organisms are free living within the environment, in contrast to M. tuberculosis, which is an obligatory parasite, it is incorrect to consider them as a variant of tuberculosis, and because person-to-person spread is unlikely, the public health implications are also quite different.
The prevalence of the different organisms depends on the country and even varies from region to region within a country. In the UK, M. kansasii is commonest overall, although M. xenopi is more common in the south, whereas M. malmoense is more common in the north [7]. In West and Central Africa, M. ulcerans predominates [8]. The incidence of infection depends on the occurrence of these organisms within the environment. In the USA, NTM are identified in almost one-half of the cultures reported for mycobacteria in clinical laboratories, although due to their ubiquitous nature some of these are presumably contaminants. Due to the wide distribution of these organisms, it is not surprising that there is a broad base of latent, inapparent infection [9]. A large proportion of the population probably becomes infected at some time, although owing to the low pathogenicity of the organisms, disease is probably confined to people with impaired local or systemic immune responses.
The identification of these organisms is difficult and generally requires skin biopsy and culture. Most laboratories use the radiometric BACTEC culture system to culture the organisms. DNA probes are then used to identify the species of NTM isolated. More rapid tests, such as PCR, are being increasingly used to identify NTB in clinical specimens [2]. Moreover, NTB share common antigens with M. tuberculosis, and it is therefore common to have a false positive Mantoux test, although the response is usually smaller than 10 mm. Interferon-gamma release assays are usually negative for NTB, although for some atypical mycobacteria (M. kansasii, M. marinum, M. szulgai), IGRAs are positive, as these species share common antigens with M. tuberculosis [10]. Specific treatment will be discussed in the relevant sections; however, in virtually all cases of localized disease, complete surgical excision of the infected lesion(s) is the preferred option and is usually curative. When operative procedures are not possible or have not been successful, or there is more widespread disease, medical treatment can be instituted based on the results of culture and sensitivity testing. Unfortunately results of in vitro sensitivity testing do not always correlate with clinical response.
Mycobacterium Marinum Infections (Swimming Pool or Fish Tank Granuloma)
This organism was first isolated from fish in 1926 by Aronson [11], and has been shown to be identical to M. balnei, named by Linell and Norden [12] in the Swedish literature in 1954; the latter authors described 80 cases of ‘swimming pool granuloma’ amongst swimmers who used a pool filled with water from hot springs. M. marinum has a widespread distribution but is only viable in heated water in temperate areas and pool or sea water from tropical areas. Although the bacterium may colonize the skin, there is evidence that a break in the epidermal barrier is required for disease to occur [13]. In the majority of cases, infection is clinically manifest between 3 and 8 weeks after inoculation of the organism into an abrasion or puncture wound. M. marinum infection normally follows trauma, often trivial, in water or from marine life. More infections are acquired from home aquariums than from swimming pools. The most common sites for infection are the elbows, knees, fingers and dorsum of the hand. Brady and colleagues [14] described an otherwise healthy 2-year-old child with M. marinum facial infection, presumed to be from contact with his aquarium pets, and there is even a reported finger infection after a dolphin trainer was bitten by his ‘pupil’ [15].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree