Abstract
This chapter examines the different types of grafts and their clinical application in wound reconstruction. Assessment of wounds is discussed, followed by the proper operative techniques for each type of wound. Instructions are also provided involving postoperative care. Receiving special treatment are the various grafts available to the surgeon, including fat, cartilage, bone, tendon, and nerve grafts. The various types of flaps are described, along with the proper technique for flap transfer. Of special significance the attention is given to the principles and techniques of microsurgery paramount, reconstruction of complex defects.
5 Surgical Principles: Grafts, Flaps, Microsurgery
5.1 Goals and Objectives
Describe different types of grafts and their clinical application in wound reconstruction.
Describe different types of flaps, their clinical application, and relevance.
Understand the basic concepts of applications of reconstructive microsurgery, microvascular operative techniques, postoperative management, and outcomes.
5.2 Grafts
5.2.1 Anatomy
The skin varies among individuals depending on their age, ethnicity, genetics, diet, sun exposure, and area of the body. In early childhood the skin is thin, but from age 10 until about 35 years it continues to progressively thicken. Sometime in the fourth decade, the dermis starts to thin again, elasticity decreases and sebaceous skin content decreases, and this phenomenon is progressive until the person dies.
The skin also varies according to area of the body, for example, the skin of the eyelids, upper extremity, medial thigh, and postauricular area is thinner than skin on the buttock area, palms, and soles of the feet. Approximately 95% of the skin is dermis, while the other 5% is epidermis. 1 The dermis contains sebaceous glands. Sweat glands and hair follicles are located in the subcutaneous fat just beneath the dermis. The skin vasculature is superficial to the superficial fascia and parallels the skin surface.
5.2.2 Terminology and Properties of Skin Grafts
Split-thickness skin graft (STSG) contains epidermis and variable amount of dermis. It can be used to resurface large surface areas as the donor site does not require closure and generally can be left to heal via secondary intention. STSG has good “take” and vascularizes well. On the minus side, thin STSG tends to shrink considerably, pigment abnormally, and is very susceptible to trauma. 2 Full-thickness skin graft (FTSG) contains epidermis and the entire dermis. The donor site, unlike that of STSG, has to be closed primarily or reconstructed with a STSG or other means making this type of graft limited in supply. FTSGs are thicker and therefore require a well-vascularized wound bed to survive. On the plus side, they have better color match, have less secondary contraction, and tend to look better and feel more natural when compared to STSGs. Common donor sites for FTSG include, but are not limited to, the supraclavicular area, pre- and postauricular skin, the neck region, forearm/arm, abdomen, and the groin region. Common STSG donor sites include inner and outer thigh region, buttock region, and scalp.
5.2.3 Graft Take
Skin graft take occurs in three phases. The first phase consists of serum imbibition and lasts 24 to 48 hours. This is followed by an inosculatory phase and a process of capillary ingrowth that occurs essentially simultaneously, until generalized blood flow has been established by the fifth or sixth postoperative day (Table 5‑1).
Retention of the function of skin appendages often depends on graft thickness. Thin grafts often have deficient functions of sebaceous glands and thus have dry and brittle appearance. Only FTSGs retain the full ability to grow hair and have normal sweat and oil secretion.
5.3 Preoperative Considerations
Wounds considered for skin grafting must have a well-vascularized recipient bed. If a wound does not have a vascularized bed, a flap which by definition brings its own blood supply should be considered. Other factors such as cleanliness of the wound, absence of infection, nutritional status, and the presence of healthy granulation tissue should be considered. A value of 105 CFU/g of tissue is a good indicator and prognostic factor for graft take; however, nothing can substitute for sound clinical judgment from an experienced surgeon in regard to wound condition. Thus, unhealthy appearance far supersedes the value of microbiologic studies.
For wounds amenable to skin grafting, the next important decision is whether to use an FTSG or an STSG. Despite better take of STSG and ability to reconstruct larger surface areas, one needs to consider the degree of graft contracture and cosmetic appearance. The decision should be individualized based on specific patient and anatomic wound location. For instance in areas such as over a joint (i.e., elbows, axilla, and neck) or facial region, FTSG would likely produce a superior outcome due to lower rate of secondary contracture and better color match. 2
5.4 Operative Technique
5.4.1 Split-Thickness Skin Graft Harvest
Thigh wound should be prepped widely or circumferentially. Any povidone iodine should be completely removed prior to harvest. The size of the graft required should be known and measured out and marked on the thigh. Generally, the graft should be 10 to 15% larger than the size of the wound unless meshing is planned. Lateral or medial thigh regions can be used and it is preferable to harvest the graft higher toward the groin to avoid visible scars. Mineral oil is commonly used to lubricate the skin prior to harvest. The harvest is commonly performed with a dermatome set at appropriate thickness and width. Thickness is often set at 0.014 to 0.015 in and can be checked with a no. 15 blade. Tension on the skin is applied with proximal and distal traction. The dermatome is angled at 45 degrees, and with one smooth motion the graft is harvested and placed in sterile saline. The donor site should be immediately covered with epinephrine solution–soaked sponges to prevent excessive bleeding. If meshed graft is desired, meshers are available with variable templates for meshing from 1.5:1 to 3:1 ratios. The meshed graft is then secured to the recipient wound either with chromic sutures or staples. The graft is dressed with a bolster dressing of choice or a NPWT dressing at 75 mm Hg of suction. The donor site is generally covered with a semiocclusive dressing (i.e., Opsite, Tegaderm, etc.) or xeroform. 3
5.5 Postoperative Care
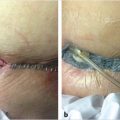
A pressure dressing or bolster is left in place for 5 to 7 days and then the graft is examined. If an NPWT dressing is used, it can be left on for a week. For FTSG, this period is varied and could be anywhere from 5 days for eyelids to 2 to 3 weeks for nipple grafts. If an extremity was grafted, early postoperative immobilization, splinting, compression, and extremity elevation are required. If the graft is adherent, Xeroform dressing or Adaptec is applied and reinforced with gauze and bandages that should be changed every day or every other day for the first 3 weeks to prevent desiccation. This can be switched to Vaseline and/or lotion once the graft is healed to prevent drying out. The patient should continue to avoid any shearing to the graft or excessive ambulation or activity to avoid injuring the new skin graft (Fig. 5‑1a,b).
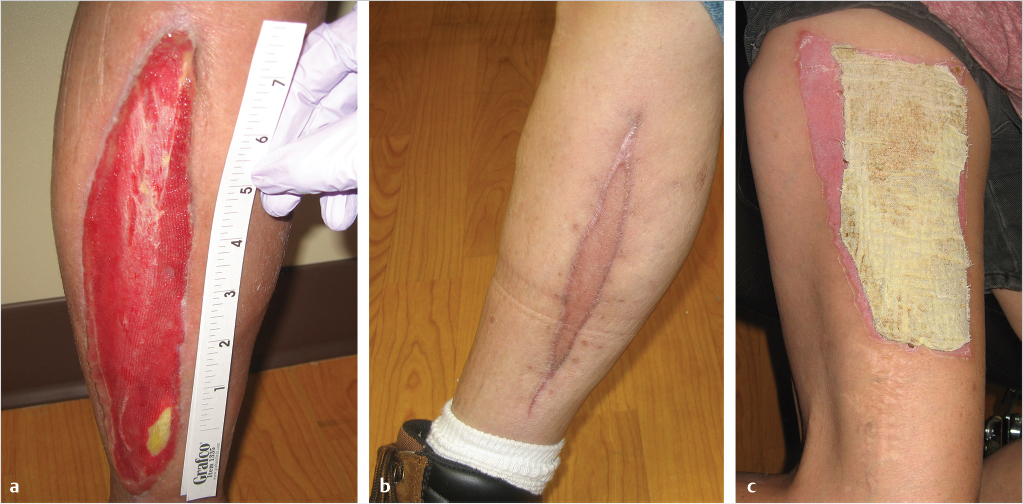
The donor-site dressing remains in place for about 10 days while allowing epithelialization of the wound and generally can be removed after this period. When Xeroform is used, it is typically not removed but gradually trimmed as the wound heals and epithelializes (Fig. 5‑1c). It is also important to warn patients that they will have a potentially visible patch of discolored skin on their thighs. 3
5.5.1 Other Types of Grafts
Fat Grafts
Fat grafting has gained popularity recently among plastic surgeons. It can be a useful adjunct to both reconstructive and cosmetic surgeons. Fat is commonly used in breast reconstruction to correct asymmetries and add volume, in facial rejuvenation as a permanent filler to add volume and restore youthful facial appearance, and in many other challenges such as scar management and body contouring procedures. The literature is replete with studies and reports about fat-grafting techniques and outcomes, and many of these present contradictory findings. This makes a choice of methods/practice confusing for one new to the field. In general, the goal of fat grafting is to achieve a predictable and reproducible outcome. Tissue fragments within the graft survive initially by diffusion, just as with a skin graft. Although relatively simple from a technical standpoint, fat grafting is actually a complex process when viewed in its entirety or from a process engineering perspective. It involves a number of steps, including donor-site preparation, tissue/graft harvest, tissue/graft washing and preparation, tissue/graft delivery, and graft/recipient site preparation and care. Each of these steps is associated with a large number of variables and options that may alter the final outcome of the procedure. For this reason, it is difficult to design and carry out well-controlled studies that will yield robust data.
Although the topic of fat grafting could cover an entire chapter itself, a few basic principles will be mentioned. First, logic would dictate that repeatable and predictable volume maintenance requires that the tissue/graft physiology be returned to normal physiological status as much as possible prior to grafting. 4 A large burden of free oil, for instance, can release fatty acids to the surrounding tissues and/or reduce pH, resulting in cell membrane damage. Ideally, the graft should have a known and reproducible aqueous fraction, as this is a volume that will be lost after grafting via resorption. If the graft has a large aqueous component, it will be easier to inject and will be associated with less shear force, but will require relatively more overcorrection to maintain volume. If the graft is particularly concentrated, it will be harder to push through a cannula and result in more cellular damage from shear stress. To minimize cell trauma and damage, it makes sense to use larger bore syringes (i.e., catheter tip syringe vs. Luer lock syringe) and larger bore cannulae for graft delivery. A standard Luer syringe has an internal diameter of 1.8 mm at the tip. The use of syringes, needles, and cannulae with a diameter of 2.5 to 3.0 mm is likely to result in less shear force and cell damage when all other variables are held constant. Further discussion of the many facets and considerations surrounding fat grafting is beyond the scope of this chapter, but the reader is referred to a number of reviews on the subject. 5 , 6 , 7 , 8
Cartilage Grafts
Cartilage grafts are often helpful during nasal, ear, or eyelid reconstruction or during rhinoplasty. The three most common sites for cartilage harvest include costal rib cartilage, conchal bowl of the ear, and nasal septum. Cartilage can be harvested with or without perichondrium. The characteristics of the cartilage are usually maintained at the recipient site, and will usually maintain the natural shape of the cartilage. Nasal septal and auricular cartilage are most often used in nasal surgery, as spreader grafts, or tip reconstruction/augmentation. Costal cartilage is most often used for nasal dorsum and external ear reconstruction. Cartilage donor sites are generally inconspicuous and the material itself is indispensable for achieving the desired results.
Bone Grafts
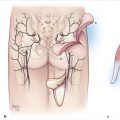
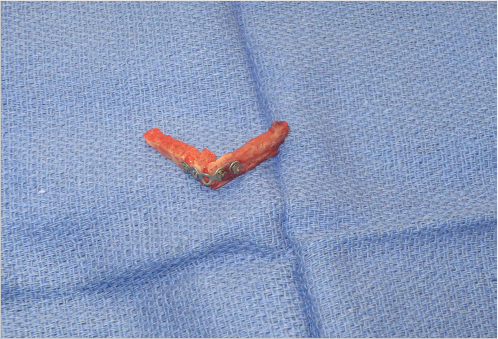
Bone grafts are common and are often used for reconstruction of the mandible, long bones of extremities, and the facial skeleton (Fig. 5‑2). Autogenous bone grafts or allografts can be used according to surgeon’s preference and the type and extent of the defect. For example, in a patient who needs reconstruction of a large mandibular defect with a history of prior radiation, a free fibula graft is a far superior option as compared to an allograft or a nonvascularized bone graft. In general, allografts have less osteoinductive and osteoconductive properties than autogenous bone grafts but have the distinct advantage of absence of donor-site morbidity. Four general types of autologous bone grafts are described: cancellous grafts, cortical grafts, cortico-cancellous nonvascular grafts, and free vascularized grafts. Cancellous grafts are typically used for reconstruction of the alveolar cleft, nasal reconstruction, or in hand reconstruction after removal of a bone cyst. They tend to vascularize better than cortical grafts but require longer time to gain structural stability and strength. The main mechanism for new bone formation is osteoconduction. Cortical bone grafts take longer to vascularize but may provide better initial stability. They have more limited osteoinductive and osteoconductive properties. Fixation of bone grafts has been shown to reduce graft resorption when grafts are placed under mobile tissues. Some of the common donor sites include calvarium, iliac crest, fibula, rib, and distal radius. 3
Tendon Grafts
Tendon grafting is useful in the management of various upper extremity injuries and defects. It can be done acutely in one stage if the wound is clean with adequate soft-tissue coverage and tendon donor sites are available outside of the zone of injury. If this is not possible, reconstruction can be performed in two stages. In the first stage, silicone rods can be placed to allow for tendon bed preparation, and in the second stage these can be removed and replaced with an appropriate graft. The palmaris longus and plantaris tendons are ideal tendon grafts as they are expendable and easily accessible. It is important to remember that about 20% of patients lack either a palmaris longus or plantaris tendons.
Nerve Grafts
Nerve grafts are commonly used in hand trauma, upper extremity, and facial reconstruction and facial reanimation. Nerve grafts can be allografts or autografts depending on surgeon’s preference. When autograft is used, one has to consider denervation somewhere and sensory deficit. Sural nerve is by far the most common nerve utilized for grafting. It courses along the posterolateral ankle and is easily accessible. It is important to inform the patient that grafted nerve is not immediately healed and it may take months until the final result is achieved. Nerve graft essentially serves as a conduit for axonal growth, which typically begins at 2 weeks after grafting and continues 1 mm/day until the target is reached.
5.5.2 Flaps
A flap is defined as a tissue with its own blood supply. This is contradistinction to grafts, which are transplanted without a blood supply. Flaps can include skin, combination of skin and fascia, musculocutaneous flaps, muscle flaps, bone flaps, or composite bone, muscle, and skin flaps (osteocutaneous).
McGregor and Morgan categorized flaps as random or axial. Random pattern flaps lack significant bias in their vascular pattern, whereas axial pattern flaps are single-pedicle flaps that encompass an anatomically recognizable arteriovenous system along their long axes. Random cutaneous pattern flaps obtain their blood supply from the subdermal plexus and have limited dimensions as determined primarily by base width and may require surgical delay. 9
Technique of Flap Transfer
Cutaneous flaps can be classified according to the technique used for flap transfer and the distance between donor and recipient site.
Rotational flaps are semicircular and design and rotate about a pivot axis. The donor site can be closed primarily or skin grafted. To facilitate closure, a burrows triangle or a back cut can be made. If a back cut is made, one has to make sure not to undermine the blood supply to the flap.
Transposition flaps are rotated (laterally) about a pivot point into an immediately adjacent defect. Because the effective length of the flap becomes shorter the farther the flap is rotated, the flap must be designed longer than the defect to be covered, otherwise a back cut may be necessary. The flap donor site can be closed primarily, with a skin graft or another flap such as a bilobed flap.
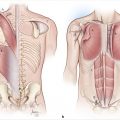
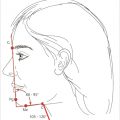
Rhomboid flap is another type of transposition flap. Limberg flap is commonly used to reconstruct cutaneous defects. It is designed with 60- and 120-degree angles opposing each other. Four different types of flaps can be designed around angles of 60 degrees with recruitment of skin from the area of most laxity (Fig. 5‑3). 10
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree