25 Appendix: Update on Breast Implant–Associated Anaplastic Large-Cell Lymphoma
With increasing public awareness and as time since implant placement progresses, more cases of breast implant–associated anaplastic large-cell lymphoma (BIA-ALCL) are being reported to the U.S. Food and Drug Administration (FDA). As of July 6, 2019, the FDA’s Manufacturer and User Facility Device Experience (MAUDE) database contained 573 unique cases of BIA-ALCL, increased from 457 as of September 30, 2018, and included 33 reported deaths. 1 Of the 573, 67% were textured implants and 28% had an unknown implant type. Of the remaining 5% or 26 implants, which were reportedly nontextured, 7 had previously been exposed to textured implants, 7 had a previous exposure to an unknown type of breast prosthetic, and 12 had an altogether unknown history. Through network collaboration with other international plastic surgical societies, the American Society of Plastic Surgery was aware of 735 total unique cases of BIA-ALCL worldwide as of August 2019. 2 To date there are no known confirmed cases of BIA-ALCL in patients with strictly nontextured implants.
A recent study by Magnusson and coworkers among the 81 patients to date from Australia and New Zealand who have developed BIA-ALCL assessed the risk of developing BIA-ALCL among several different implant types. 3 It demonstrated an estimated incidence of 1:86,029 patients with Siltex (Mentor Worldwide LLC, Santa Barbara, CA) implants and a 16.5-fold higher risk with Biocell (Allergan Inc., Dublin, Ireland) and a 23.4-fold higher risk with Silimed polyurethane (Silimed Inc., Rio de Janeiro, Brazil). An analysis of the surface characteristics of the implants was performed, grading them from 1 to 4 using a combined surface area/roughness implant classification system developed by Jones and coworkers. 4 The Biocell and Silimed implants were characterized as grade 3 and 4, respectively, suggesting a potential correlation between degree of roughness and risk for developing cancer. While polyurethane implants have been banned in the Unites States for many years due to carcinogenic components, the Biocell implant is the most prevalent offender linked to BIA-ALCL worldwide, and Allergan’s CA/CARE trial estimated its risk at 1:2,207. 2
In mid-2019, following recalls in over 30 other countries, the FDA issued a recall on Biocell textured implants as well as expanders (despite no cases of BIA-ALCL from expanders having been reported to date). 5 While the recall was initially voluntary, it has since been revised to a Class 1 recall, the most serious type. Because the absolute risk of developing BIA-ALCL is low, it is not recommended that patients with Biocell implants have their implants removed. However, providers with shelved implants are mandated to return their unused supply. The FDA has more recently proposed requiring implant manufacturers to place a boxed warning on the product informing consumers of, among other things, the risk of BIA-ALCL.
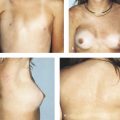
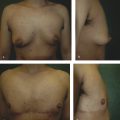
Standardized clinical practice guidelines for BIA-ALCL have been updated by the National Comprehensive Cancer Network (NCCN) in 2019. 6 Patients who present with a new periprosthetic effusion 1 year or later following implant placement, or who present with a new breast mass, rash, ulceration, or axillary lymphadenopathy, should undergo initial evaluation with ultrasound. If ultrasound findings are equivocal, magnetic resonance imaging (MRI) is recommended. Fine-needle aspiration should be performed to sample as much periprosthetic fluid as possible, and masses should be biopsied. Specimens should undergo evaluation of cellular morphology, CD30 immunohistochemistry, and flow cytometry to characterize T cells. Additional biomarkers that may be relevant in distinguishing BIA-ALCL may include CD2, CD3, CD4, CD5, CD7, CD8, CD45, and anaplastic lymphoma kinase. When confirmed, patients should undergo preoperative evaluation with a positron emission tomography/computed tomography (PET/CT) scan. If systemic involvement is suspected, patients are recommended to undergo bone marrow biopsy.
Two methods of staging BIA-ALCL have been employed: a Lugano revision to the Ann Arbor staging system and a TNM staging system initially developed by MD Anderson in 2016. Due to the improved ability of TNM staging to characterize tumor morphology and behavior more accurately as well as predict survival and recurrence, 7 it is now the recommended system.
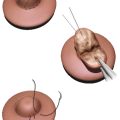
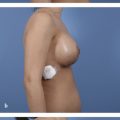
Optimal management remains removal of implant and complete surgical resection of capsule and associated masses, or en bloc capsulectomy. Adjuvant radiation therapy is recommended for incomplete resection, positive margins, or unresectable disease. Systemic adjuvant therapy is advised for nodal involvement or invasion beyond breast capsule, and first-line therapy includes combination anthracycline-based chemotherapy and brentuximab vedotin (Adcetris, Seattle Genetics, Bothell, WA). In patients who achieve a complete response, history and physical should be performed every 3 to 6 months for 2 years, and CT chest/abdomen/pelvis or PET scan should be considered every 6 months for 2 years.
References
- 1 U.S. Food and Drug Administration. Center for Devices and Radiological Health. Medical device reports of breast implant–associated anaplastic large cell lymphoma. http://www.fda.gov/medical-devices/breast-implants/medical-device-reports-breast-implant-associated-anaplastic-large-cell-lymphoma. Updated 2019. Accessed Oct 24, 2019
- 2 Clemens M. BIA-ALCL resources: by the numbers, and what they mean. https://www.plasticsurgery.org/for-medical-professionals/health-policy/bia-alcl-physician-resources/by-the-numbers. Updated 2019. Accessed December 9, 2019
- 3 Magnusson M, Beath K, Cooter R et al. The epidemiology of breast implant–associated anaplastic large cell lymphoma in Australia and New Zealand confirms the highest risk for grade 4 surface breast implants.. Plast Reconstr Surg 2019; 143 (5) 1285-1292 PubMed 10.1097/PRS.0000000000005500 30789476
- 4 Jones P, Mempin M, Hu H et al. The functional influence of breast implant outer shell morphology on bacterial attachment and growth.. Plast Reconstr Surg 2018; 142 (4) 837-849 PubMed 10.1097/PRS.0000000000004801 30252806
- 5 U.S. Food and Drug Administration. Allergan recalls Natrelle Biocell textured breast implants due to risk of BIA-ALCL cancer. https://www.fda.gov/medical-devices/medical-device-recalls/allergan-recalls-natrelle-biocell-textured-breast-implants-due-risk-bia-alcl-cancer. Updated September 12, 2019. Accessed December 9, 2019
- 6 Clemens MW, Jacobsen ED, Horwitz SM. 2019 NCCN consensus guidelines on the diagnosis and treatment of breast implant-associated anaplastic large cell lymphoma (BIA-ALCL).. Aesthet Surg J 2019; 39 (Suppl. Supplement_1) S3-S13 PubMed 10.1093/asj/sjy331 30252806
- 7 Clemens MW, Medeiros LJ, Butler CE et al. Complete surgical excision is essential for the management of patients with breast implant–associated anaplastic large-cell lymphoma.. J Clin Oncol 2016; 34 (2) 160-168 PubMed 10.1200/JCO.2015.63.3412 26628470
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree