23 Injectable Fillers of the Face
Introduction
Perhaps the most significant change in facial rejuvenation in the last decade has been the introduction of nonsurgical techniques for restoration of volume and for the relaxation of facial wrinkles. In our time, facial rejuvenation has undergone a significant paradigm shift from initially concentrating only on tightening the skin and aponeurosis to now including repositioning and restoration of volume. A youthful appearance has a smooth contour with gradual transitions between the different facial subunits. In fact, after neuromodulators, soft tissue fillers have become the most commonly performed cosmetic, minimally invasive procedure in the United States—growing 205% from 2000 to 2012, with 1.9 million procedures performed annually. 1 This rapid growth has been possible due to the advances in soft tissue fillers, which have improved cosmetic results while simultaneously reducing adverse events.
The evolution of injection filler compounds has been driven largely by unacceptable complication rates in early materials. The use of silicone and paraffin-based products pre-2000 led to frequent postinjection complications including delayed granuloma and paraffinoma formation. 2 Although bovine collagen agents were approved by the U.S. Food and Drug Administration (FDA) in 1981 for injection, these formulations were likewise fraught with adverse reactions and short duration of action. 3 , 4 More stringent purification methods in the synthesis of injectable fillers at the turn of the century have reduced the incidence of adverse events. The subsequent introduction of new classes of dermal fillers resulted in fewer hypersensitivity reactions and longerlasting efficacy.
Hyaluronic acids (HA), calcium hydroxylapatite (CaHA), polyL-lactic acid (PLLA), and polymethylmethacrylate (PMMA) are the recent mainstays of injection filler treatment.
All wrinkle fillers are considered medical devices by the FDA. 5 Both temporary (HA, CaHA, and PLLA) fillers and the permanent filler (PMMA) have received FDA approval under the medical device classification ( Table 23.1 ). FDA approval for the injection of these compounds is restricted to the nasolabial folds, marionette lines, lips (specifically Restylane), and regions of facial lipoatrophy. While the nasolabial folds are the most commonly injected site, 6 other commonly injected areas are addressed off-label, including the temporal scalp hollow, tear troughs, and prejowl sulci. Low complication rates can be expected in experienced hands.
The physicochemical properties of fillers determine how they will behave both during and after injection. Soft tissue fillers differ in their rheological properties in terms of their viscosity, elasticity, and plasticity. Product viscosity ( η *) describes how it flows through the needle, and G′ (G prime) describes its stiffness and ability to resist deformation while being injected. 7 Once injected, these properties also affect how the product will respond to facial movements. A high G′ will resist skin forces better and may be superior at adding volume, whereas a product with a low G′ is less stiff and may be better suited for filling fine wrinkles. Fillers with higher η * will be less likely to migrate in the tissues after injected.
Choosing the appropriate filler type requires a thorough understanding of the differences between filler types, including their aesthetic advantages and disadvantages. Comprehension of vascular and musculature anatomy maximizes the efficacy of injections, and reduces the risk of complications as described in subsequent sections. We provide a description of FDA-approved fillers, injection strategies, and mitigation of complication risks (and their management).
Historical Perspective
Injectable facial fillers have been used since the 1800s, shortly after the invention of the syringe. The first facial filler to be used was paraffin. Paraffin injection led not only to facial disfigurement but life-threatening complications as well which eventually led to its abandonment. 2 Paraffin was discovered in 1830 by the German chemist Baron Karl Ludwig von Reichenback (1788–1869) by distillation of beechwood tar. 8 The material was noted to be largely inert and the medical community started utilizing the material. Notably, the famous Austrian surgeon who is considered the father of modern abdominal surgery, Theodur Billroth (1825–1899), would use paraffin for joint lubrication. Paraffin was first used cosmetically by Robert Gersuny (1844–1924), who trained under Billroth in Vienna. Gersuny would employ paraffin for breast and nasal augmentation. In 1875, Vaseline (Unilever) appeared as a combination of solid and liquid paraffin. 9 Because of the 40°C melting point of Vaseline, it could easily be liquefied, and after injection into the human body it rapidly hardened. Gersuny advocated sterile injections and small amounts to avoid complications. Vaseline quickly gained popularity for cosmetic nasal augmentation. It was not until the early 1900s that the complications began to be described. Inflammation, infection, migration, granulomatous reaction to paraffin, white plaque discoloration, and pulmonary and cerebral emboli were occurring as a result of Vaseline injections. 10 , 11 Perhaps the most famous account of complications was that of Gladys Spencer-Churchill (1881–1977), the Duchess of Marlborough. 12 The Duchess was a socialite who was famous for her beauty and her blue eyes, which were painted in the portico ceiling in the main doors of the palace. Preoccupied with her nose, the duchess had paraffin injected into her nasal dorsum, which migrated throughout her face. She became deformed and died a recluse in her house, where she did not allow mirrors. Over the years, different oils have been used for cosmetic injection including lanolin, mineral oil, and vegetable oil, which have ultimately been abandoned due to undesirable complications. It was not until 1981 that the FDA approved the use of the first filler for soft tissue augmentation: bovine collagen. 3
Materials
Collagens
Bovine collagen (Zyderm I Collagen Implant, Collagen Corp.) was the first filler to be approved by the FDA in 1981. Six years of development and testing prior to its FDA approval were necessary to demonstrate its safety as a facial filler. 13 In 1988, actress Barbara Hershey underwent off-label collagen injection into her lips prior to her role in the movie “Beaches,” creating a media sensation and rage for facial collagen injections. 14 Despite the exhaustive purification process, bovine collagen carries a risk of allergic reaction to the bovine content. Prior to injection, all patients required subdermal skin testing of 0.1 mL of the product in the forearm. Repeated testing 2 weeks after the initial test was recommended to avoid false-negative skin testing which was reported to occur in 6.2% of cases. 15 , 16 The duration of Zyderm was 3 months, and in an effort to increase the duration of the product, newer collagen products were developed. Zyderm II showed longer efficacy with a more concentrated formulation, increasing from 3.5% to 6.5%. 15 Zyplast increased the duration of the product by cross-linking with glutaraldehyde. Inadvertently, this change also increased its antigenicity. Specific immunoglobulin responses to bovine collagen were reported in the 1990s with complications including dermatomyositis and polymositis. 17 , 18 In more recent years, collagens have largely fallen out of favor in exchange for newer facial fillers, with less antigenicity and improved longevity. 19 , 20 , 21
Hyaluronic Acids
Chemical modifications to naturally occurring hyaluronic acids (HAs) have revolutionized the world of facial fillers. HA is a highly hydrophilic glycosaminoglycan found in the extracellular compartments of the dermis, umbilical cord, synovial joint fluid, and hyaline cartilage. The abundance of HAs in skin decreases as it ages, leading to increased rhytides and decreased hydration. Injection of HAs filler can restore facial volume. An important mechanism for volume restoration is the increase in water content given that HA can hold over 1,000 times its mass in water. Its profile makes HAs ideal fillers given that they are biodegradable, biocompatible, and nonimmunogenic. Naturally occurring HAs have a half-life of 12 to 24 hours. 22 To increase the half-life in injectable HA-based products, cross-linking between the HA chains was introduced. HA chains were cross-linked with alternating D-glucuronic acid and N-acetyl-D-glucosamine monosaccharides. 23 Cross-linking is increased with butanediol diglycidyl ether (BDDE), divinyl sulfone, bidcarbodiimide, or 1,2,7,8-diepoxyoctane bonds. Ideally, cross-linking increases the longevity of the product. Excessive cross-linking, however, can lead to foreign body reactions. 24 The half-life of cross-linked products is 4 to 12 months. 25 Studies demonstrated superiority of various HAs over collagen in longevity and safety. 19 , 20 , 21 An early doubleblind, randomized, split-face trial for nasolabial fold treatment showed 60% of patients had persistent improvement at 6 months with HAs versus only 8% with collagen. 20
The earliest HA facial fillers were derived from rooster combs, which led to some allergic reactions due to residual avian proteins. Currently, nonanimal stabilized HAs (NASHAs) derived from fermentation of Streptococcus equi bacterium are the only type of HAs available in the market. 5 NASHA fillers received approval by the FDA for cosmetic injectable use in 2003. HAs products available differ in the degree of cross-linking, the gel consistency, and the concentrations. In the United States, currently available FDA-approved HAs include Restylane and Perlane (Medicis Aesthetics, Inc); Juvéderm Ultra, Ultra Plus, and Voluma (Allegran, Inc.); and Belotero (Merz Aesthetics, Inc.) (see Table 23.1). HA products can be divided into biphasic or monophasic gels. The first HAs to become commercially available were biphasic gels. In biphasic gels, cross-linked HA particles are suspended in non–cross-linked HAs used as carrier. The non–cross linked HA will increase fluidity, which allows injection through fine needles. Restylane and Perlane are both biphasic NASHAs cross-linked with BDDE. They were approved by the FDA in 2003 and 2007, respectively. The difference between the two formulations is the particle size. Restylane molecules are half of the size of Perlane (250 µm vs. 550 µm). Monophasic fillers are monodensity gels, which are mixed and cross-linked in one step. Monophasic gels have a more even distribution after injection in comparison to biphasic gels. 6 Juvéderm and Belotero are both monophasic gels. Juvéderm Plus and Juvéderm Ultra Plus vary in their degree of cross-linking. Juvéderm Ultra Plus is denser and lasts longer than its counter product. 21 , 26 Biphasic HAs gels have been suggested to have greater longevity than monophasic gels. A recent evaluator-blind, split-face trial for treatment of glabellar lines reported biphasic gels provided persistent improvement at 1 year in 70% of patients while monophasic gels had persistent improvement in only 10% of patients. 27 Belotero is a polydense monophasic gel. To create several densities, an initial cross-linking step is followed by the addition of new HA and a second cross-linking step. Polydense monophasic gels create smoother deposits than other HAs. An evaluator blinded, randomized, split-face trial for treatment of the nasolabial folds showed significantly smoother contour 1 month postinjection when comparing Belotero versus Restylane as measured by optic threedimensional scanners. 28 The different properties of the gels allows for different amounts of diffusion after injection. 6 Therefore, different anatomical regions of the face will benefit from the use of particular products. Juvéderm Voluma XC was developed to add volume to the midface. Its higher G′ results in greater hardness and cohesivity which results in improved lifting capability. 29
Calcium Hydroxyapatite
Calcium hydroxyapatite (CaHA) injectable fillers are composed of synthetic CaHA microspheres measuring 25 to 45 µm in diameter suspended in a carboxymethylcellulose and glycerine carrier gel. CaHA is a mineral naturally present in bone and teeth and therefore not immunogenic. Prior to its cosmetic use, CaHA had been use for vocal fold augmentation and to treat urinary incontinence. 30 , 31 In 2006, Radiesse (Merz Aesthetics, Inc.) was the first filler of its kind to receive FDA approval as a facial filler. 32 Radiesse is composed of 30% CaHA microspheres and 70% gel carrier. CaHA is currently FDA approved for the treatment of nasolabial folds and human immunodeficiency virus (HIV)-associated facial lipodystrophy. 33 , 34 When injected as facial filler, CaHA microspheres serve as a scaffold and recruit fibroblasts, which then secrete new collagen. Eventually, the CaHA microspheres are degraded into calcium and phosphate and reabsorbed. 35 , 36 , 37
CaHA fillers have greater longevity than previously available facial fillers. A split-face trial in 2007 demonstrated longer efficacy of Radiesse over collagen. At 6 months, results were graded by blinded evaluators, and they found that 82% of the Radiessetreated folds had improvement compared to 27% in the collagen control group. In addition, the amount of CaHA required for optimal correction was half of that needed for collagen. 38 Likewise, when comparing CaHA to HAs, studies suggest CaHA is longer lasting. 39 At 12 months in a split-face randomized trial in 2008, 79% of the CaHA folds were still improved versus 43% of the NASHAs folds. 39 The longevity of the fillers will depend in part on injection technique, patient’s age, and site of injection. CaHA longevity ranges from 10 to 14 months with an average correction time of 1 year. 40 , 41
The 2007 consensus recommendations confirmed the efficacy of CaHA for the correction of volume loss in the midface and lower face. The nasolabial folds, oral commissures, prejowl area, chin, and the midface may all be treated with CaHA. 41 However, because of the risk of nodule formation and necrosis, CaHA is contraindicated in the lips and glabella. Nodule formation rates of has high as 5.9% have been reported in the literature when injected in these areas. 42 Superficial injections are also associated with higher nodule formation and hence subdermal injections are recommended. Of note, CaHA, in contrast to almost all other fillers, does not come premixed with local anesthetic. The FDA has approved premixing CaHA with lidocaine prior to the injection to increase patient comfort. 43
Poly-L-lactic Acid
Poly-L-lactic acid (PLLA) is a synthetic polymer from the α-hydroxyl-acid family, similar to absorbable suture material and immunologically inert. Sculptra (Valeant Aesthetics, Inc.) is the current PPLA facial filler available on the market. In contrast to traditional fillers that provide immediate results, PLLA fillers provide gradual results by stimulating the synthesis of collagen. Several treatment sessions 4 to 8 weeks apart are needed for optimal facial volume restoration. The results may last 2 to 5 years; however, PLLA is biodegradable and is not permanent. 44 PLLA particles are reabsorbed 9 months after injection. 45 PLLA was initially approved by the FDA for treatment of facial lipoatrophy in patients with HIV in 2004. Subsequently in 2009, PLLA was approved for cosmetic treatment of facial contour deficiencies and other facial rhytides.
Sculptra comes as a freeze-dried powder with sodium carboxylmethylcellulose and mannitol. The powder requires hydration to generate a homogeneous suspension. Sterile water and lidocaine can be used to hydrate the powder. The reconstitution is very important to prevent patients from developing subcutaneous nodules as well as to prevent needle clogging during injection. 46 , 47 Reconstitution for at least 2 hours prior to use and preferably overnight prior to injection is recommended. Gentle shaking of the syringe prior to the injection may be necessary to maintain the particles in suspension. Once reconstitution is performed, the product should be used within 72 hours. Prior to reconstitution, PLLA can be stored in its freeze dried form for up to 2 years at room temperature.
Injection of Sculptra should be deep to the dermis into the superficial subcutaneous or preperiosteal tissues, which may help decrease nodule formation. Early studies of PLLA in HIVassociated lipoatrophy found subcutaneous nodules in as many as 52% of patients. 48 , 49 Subsequent studies reported decreasing nodule formation of 0 to 13% could be achieved by increasing the dilutions of PLLA in water and by performing deeper injection. 47 , 50 , 51 To help prevent nodules and clumping of the particles after the injection, patients are advised to massage the area of injection for several days.
Polymethylmethacrylate
Polymethylmethacrylate (PMMA) PMMA is the only facial injectable filler that is considered to be permanent. 52 This material had been previously used for years as bone cement. More recently, preparations of bovine collagen with microspheres of PMMA measuring between 30 and 50 µm are being used as facial fillers. Smaller particles of PMMA are easily degraded by phagocytosis, while in contrast bigger particles are associated with higher granuloma formation. The collagen prevents PMMA particles from clustering. The collagen carrier is absorbed 1 month after injection and replaced by the patient’s neocollagenesis. The final augmented volume is considered to be 80% attributed to neocollagenesis and 20% to PMMA. 53
ArteFill (Suneva Medical, Inc.) is a thirdgeneration PMMA facial filler and the only PMMA FDA approved filler. Early formulations of the PMMA (Artecoll and Arteplast, originally manufactured by Artes Medical, Inc.) were used in Europe for 10 years, producing granulomas at an unacceptable rate of 2.5%. The formulation was changed to decrease the granuloma formation by removing the negative charge on the particles and producing a smoother sphere. After the changes, the rate of granuloma formation decreased significantly to less than 0.01%. 53 In 2006, ArteFill received FDA approval for treatment of deep nasolabial folds. Because the formulation contains bovine collagen, patients should undergo skin testing 1 month in advance to ensure no allergic reactivity to the product. The collagen in ArteFill has been modified to remove antigenic portions of the molecule. A recent ArteFill skin test study of 1,000 patients revealed a positive allergy rate of only 0.2%. 54 ArteFill can be stored in the refrigerator for 1 year (4°C). Comparison of the thirdgeneration PMMA filler versus collagen fillers by blinded observers showed significant superiority in the rhytid correction PMMA group at 6 months (p < 0.001). 52
Given that PMMA is a permanent filler, the longevity of the ArteFill should be taken into consideration and discussed with patients prior to injection. The permanent microspheres are not degradable; thus, the only option for reversal is complete local excision. Blinded observers rated correction after ArteFill to the nasolabial folds to be equally preserved at 6 months and 5 years after injection. 55 The use of temporary fillers prior to PMMA can be considered to evaluate the results prior to moving to permanent filler. The most common adverse reaction reported is mild lumpiness. Because of the significant nodule formation after lip injections, the use of PMMA in the perioral region is not recommended. 56
Autologous Fibroblasts
Advances in cell culture technology have made it possible to use the patient’s own skin to restore facial volume and rejuvenate the face, although limited experience is available with these newer techniques. In 2011, LaViv (azficel-T; Fibrocell Science, Inc.), a dermal filler derived from autologous fibroblast, was approved by the FDA for correction of nasolabial folds. The process of culturing fibroblast involves harvesting a punch biopsy from the postauricular area of the patient, then shipping the sample to the company, which then processes and expands the sample, and, finally, returns autologous filler for injection. Because LaViv utilizes autologous cells, it has low antigencity and low hypersensitivity reactions. 57 In a 2007 doubleblinded, randomized phase III clinical trial comparing autologous fibroblasts versus placebo for the treatment facial contour defects, fibroblasts were shown to significantly improve facial defect. The vast majority of the facial defects studied were nasolabial folds and acne scars. 57 , 58 Longevity past 6 months remains to be been determined. The use of LaViv for other facial defects, including burns, is currently being investigated.
Preinjection Considerations
Patient counseling prior to injection should discuss the healing period and the duration of the results. Preinjection photos should be taken. Mild edema is the most common side effect after filler injections. Patients should be advised to schedule their injections appropriately. Lip and tear trough injections have a higher rate of swelling and ecchymosis than other sites. Ice, bromelain (a derivative of pineapple), or Arnica Montana tablets may reduce swelling and bruising after injection. 59 , 60
Patients with systemic diseases such as diabetes, autoimmune disorders, and HIV can have facial filler injections and are considered to have relatively normal wound healing capabilities. However, if there is any indication of impaired wound healing, careful consideration should be given prior to filler injection. Continued usage of aspirin, warfarin, and other antiplatelet and anticoagulation medications should be taken into account on an individual case basis and are not considered absolute contraindications to facial fillers. Skin should be healthy prior to facial filler injection. If patients have undergone prior laser or chemical peels, facial fillers should be delayed until the skin is healed. Consider antiviral prophylaxis prior to lip injection in patients with known oral herpes simplex infection. 61 Patients who have a history of hypertrophic scarring or keloid scars should be counseled about the potential risk. However, hypertrophic scarring is only a risk with intradermal injection and not with deeper injections.
Topical anesthetic may be applied prior to injection of fillers to minimize discomfort. The most popular available combination creams include BLT (benzocaine 20%, lidocaine 6%, and tetracaine 4%) and EMLA (eutectic mixture of local anesthetics with 2.5% lidocaine and 2.5% prilocaine). BLT has an onset of action of approximately 20 minutes, and EMLA has an onset of action of about 1 hour. Regional blocks with local anesthetic such as 1 to 2% lidocaine can be used in the areas of the mental and infraorbital nerves. Careful evaluation of the patient should be performed prior to administering the anesthetic, as even mild swelling from the topical cream can result in a false temporary improvement in finer rhytides and regional blocks can create temporary asymmetries.
Areas of Treatment
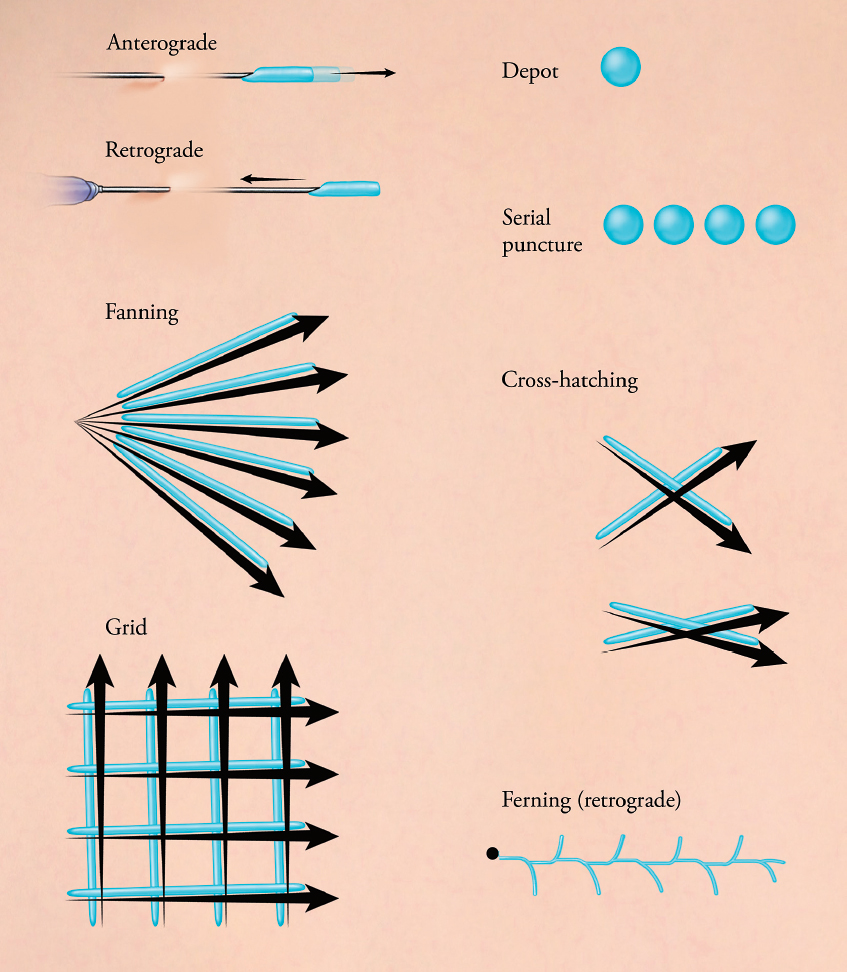
When rejuvenating the aging face, volume restoration and softening of facial rhytides can help achieve a natural, more youthful appearance. In order to achieve appropriate rejuvenation in the different areas of the face, the clinician will require different materials and injection techniques. Fine superficial wrinkles are preferentially treated with HAs, particularly polydense monophasic gels. Supraperiosteal injections for cheek restoration require large volume replacement and CaHA with high density HAs are often chosen for these areas. Available injection techniques include linear threading, fanning, crosshatching, and serial punctures ( Fig. 23.1 ). Linear threading deposits the product in a linear pattern, using either an anterograde or retrograde injection technique. It is often used to soften nasolabial folds. Fanning uses the same insertion point, with redirection of the needle in multiple directions, and is often used in the cheek and temples. Cross-hatching uses multiple parallel lines that intersect and is key for discrete isolated depressions such as the oral commissure. The serial puncture technique deposits small aliquots of product using direct injections over the desired area and is often used in tear trough treatment. The filler material, depth of the injection, and the injection technique should be chosen based on the area of the face being treated and will determine the expected results and their duration. The duration of filler effect will vary depending not only on the filler material, but also on the area of injection. Locations with significant underlying muscle motion have a shorter duration than areas without underlying muscle motion. Filler injected to the lips tends to have very low duration, whereas injections to the tear trough or the ear lobe have a longer duration.
Upper Third of the Face
Hollow Temple
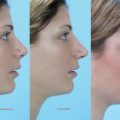
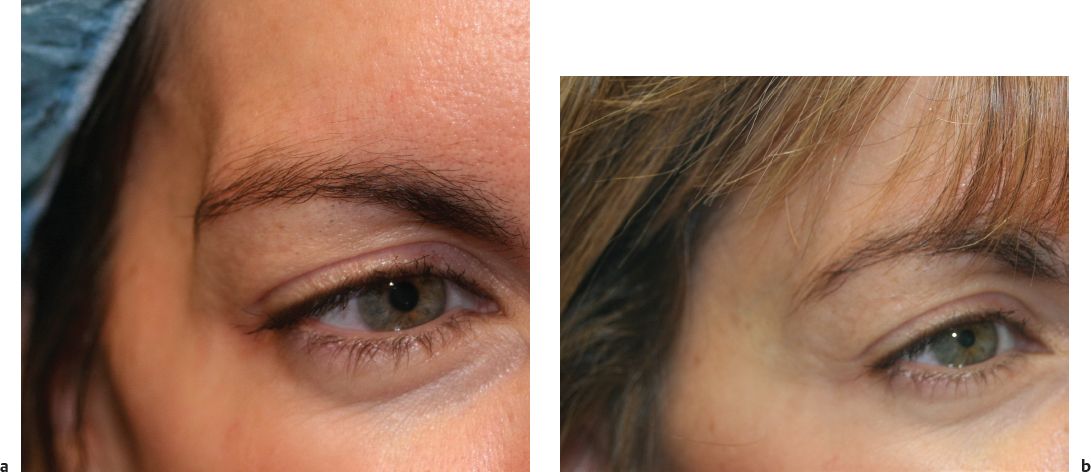
Temporal fossa wasting and hollowing lead to an aged appearance and a general gaunt and unhealthy look. In a youthful face, the temple is convex and continuous with the zygomatic arch and the orbit. Hollow temples will make the lateral orbital rim visible and contribute to a gaunt skeletonized look. Hollowed temples are more common in thin patients and with HIVassociated lipodystrophy. Most patients do not specifically recognize or request temporal augmentation but rather complain of an overall sense of appearing older or tired. The practitioner must explain how volumization of the temples will help restore volume and overall roundness to the face. In addition to addressing facial contour, temporal augmentation will provide a lateral brow lift to the portion of the brow that overlies the temporal fossa, further rejuvenating the upper third of the face. 62
Augmentation of the temples can be achieved with multiple fillers including CaHA, PPLA, and HA ( Fig. 23.2 ). 63 , 64 Filler injections in this region are performed deep to the temporalis muscle in the preperiosteal layer. A 1-inch needle is needed to penetrate the temporalis muscle and reach the appropriate depth. The medial and superior aspects of the temple provide the most benefit aesthetically. The injection is usually a bolus depot injection followed by massage of the area to smooth the contour. The superficial temporal artery is easily palpated in the region and should be avoided. Often 1 mL of CaHA or HA is needed for each side. Because of the depth of injection, CaHA is preferred over HA in this region. PLLAs also work particularly well for this region; however, serial injections are required to achieve volumization. If PLLA is chosen, a 25-gauge needle of at least 1-inch is used. Immediate improvement is noted that is short lasting, followed by gradual neocollagenesis. Massage is performed for 5 minutes by the injector immediately after injection, and subsequently by the patient for several days. Patients may require three treatment sessions of PLLA 4 to 8 weeks apart.
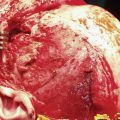
Localized bruising is the most common side effect of temporal injections. The patient should be advised of temporary mild soreness, particularly with chewing, and even possible transient trismus secondary to temporalis muscle inflammation or intramuscular hematoma formation. Superficial vein prominence has also been noted as a side effect of the injection. 65
Glabella
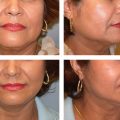
Aging in the forehead region is characterized by brow ptosis and deep hyperfunctional rhytides. Currently, neuromodulators to the forehead muscles are the main rejuvenation strategy prior to forehead lift. Deep static rhytides resulting from repeated muscle contraction of the frontalis, corrugulator, and procerus muscles and volume loss associated with aging may not completely resolve with botulinum toxin injection alone. Static rhytides can be treated with a combination of neuromodulators and/or facial fillers to smooth and soften the forehead, which will rejuvenate the face and also give a more relaxed look ( Fig. 23.3 ). 66
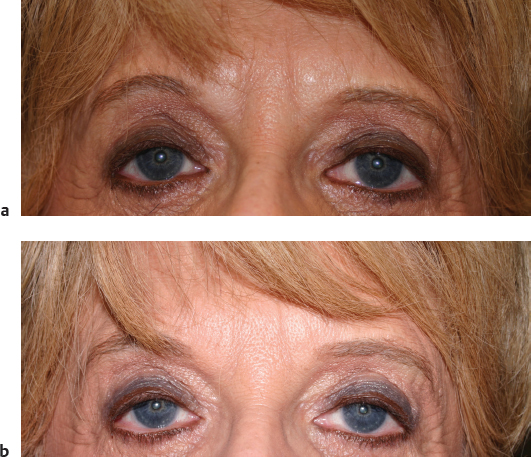
The injection technique is usually intradermal or immediately subdermal below the rhytid in the area where the loss of volume has occurred. In a randomized controlled trial, the use of concurrent botulinum toxin and filler was superior to filler alone. 67 Mild side effects such as swelling and tenderness have been reported in studies looking at glabellar filler injection. Although rare, case reports of glabellar necrosis due to filler embolization have been published in the literature. 68 , 69 The blood supply to the glabella comes mainly from the supratrochlear and supraorbital arteries. These vessels lie in the superior periorbital region, and careful superficial injection technique should prevent this dreaded complication.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree