21 The Diagnosis and Management of Epiphora
Abstract
“The Diagnosis and Management of Epiphora” deals with the very common complaint of a watering eye, which may affect patients of any age. It is important to establish the true nature of the patient’s complaint. The term epiphora refers to the overflow of tears onto the cheek. The complaint by a patient of a “watering” or “watery” eye, or “tearing,” may not imply that tears actually overflow from the eye. There are many abnormalities that can lead the patient to seek attention for this complaint. It is important to establish the underlying cause by obtaining an accurate detailed history and by performing an appropriate clinical examination and, where indicated, supplementary investigations. It should not be assumed that this complaint automatically signifies a lacrimal drainage system obstruction. The surgical procedures required for the management of a variety of congenital and acquired lacrimal drainage system abnormalities are described in detail.
21.1 Introduction
The complaint of a watering eye is very common and may affect patients of any age. It is important to establish the true nature of the patient’s complaint. The term epiphora refers to the overflow of tears onto the cheek. The complaint by a patient of a “watering” or “watery” eye, or “tearing,” may not imply that tears actually overflow from the eye. There are many abnormalities that can lead the patient to seek attention for this complaint. It is important to establish the underlying cause by obtaining an accurate detailed history and by performing an appropriate clinical examination and, where indicated, supplementary investigations. It should not be assumed that this complaint automatically signifies a lacrimal drainage system obstruction.
21.2 Applied Anatomy
The anatomy of the lacrimal drainage system and nose is described in detail in Chapter 2. This anatomy should be carefully reviewed.
21.3 History
A history of irritation, foreign body sensation, or allergy should alert the clinician to the possibility of reflex hypersecretion of tears. Patients with a dry eye may paradoxically present with a complaint of epiphora related to increased reflex tear secretion. It is essential to exclude such an underlying etiology, because these patients will not benefit from any form of lacrimal drainage surgery, however well performed. The use of topical medications, such as glaucoma drops, should raise the suspicion of allergy with an associated lower lid dermatitis or a cicatrizing conjunctivitis with punctal or canalicular obstruction. A history of previous dacryocystitis indicates the presence of a nasolacrimal duct obstruction.
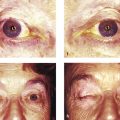
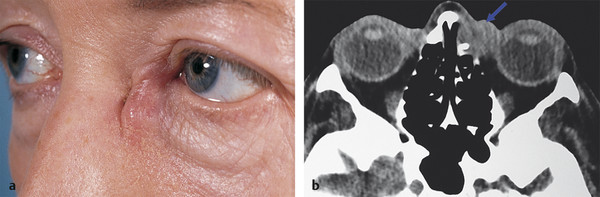
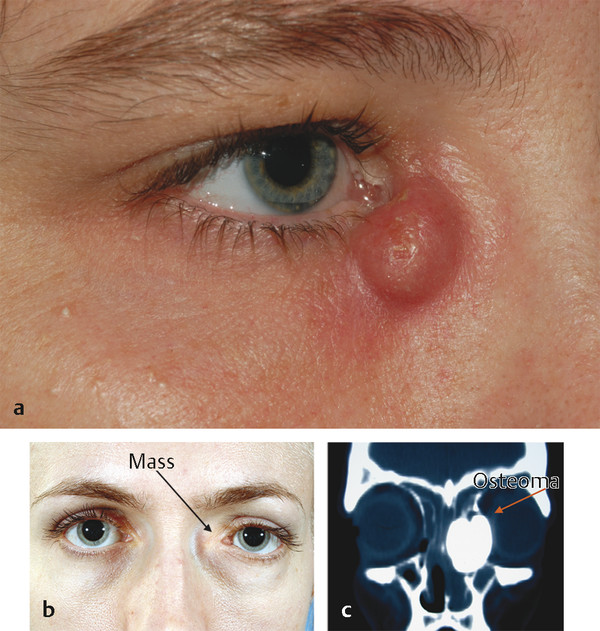
A history of previous intranasal surgery or facial trauma should alert the clinician to the possibility of a nasolacrimal duct obstruction and may require more detailed preoperative investigations, such as coronal computed tomography (CT) to determine the presence and location of microplates or bone grafts and the position of the cribriform plate. A history of bloody tears, nasal obstruction, or epistaxis should raise the suspicion of nasal, sinus, or lacrimal sac malignancy or Wegener’s granulomatosis (Fig. 21‑1). Such patients should be evaluated with the assistance of an ear, nose, and throat (ENT) specialist. A previous history of a facial palsy should alert the clinician to the possibility of “crocodile tears,” a residual incomplete blink, or frank lagophthalmos.
The history taking should be tailored to the patient’s age, bearing in mind the common causes of epiphora in each age group:
Infants and children
Congenital nasolacrimal duct obstruction.
Congenital anomalies of the lacrimal drainage system.
Young adults
Trauma: canalicular or lacrimal sac lacerations, nasoethmoidal fractures.
Canalicular scarring: herpes simplex canaliculitis.
Middle-aged adults
Dacryoliths: actinomyces infection.
Cicatricial disorders of the lower eyelid anterior lamella.
Older adults
Idiopathic primary acquired nasolacrimal duct obstruction
Involutional eyelid malpositions.
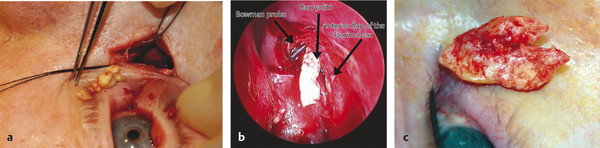
Dacryoliths (lacrimal sac stones) are found in 2 to 5% of dacryocystorhinostomies (DCRs). Dacryoliths consist of dried mucus, lipid, and inflammatory debris and are more often seen in patients with chronic dacryocystitis. They can be seen as small soft flakes, can consist of multiple small stones, or can form an entire cast of the lacrimal sac (Fig. 21‑2, Video 21.1). They can sometimes be seen as filling defects on dacryocystography (DCG).
The examination of infants and children with epiphora is considered separately under Nasolacrimal Duct Probing and Intubation.
21.4 External Examination
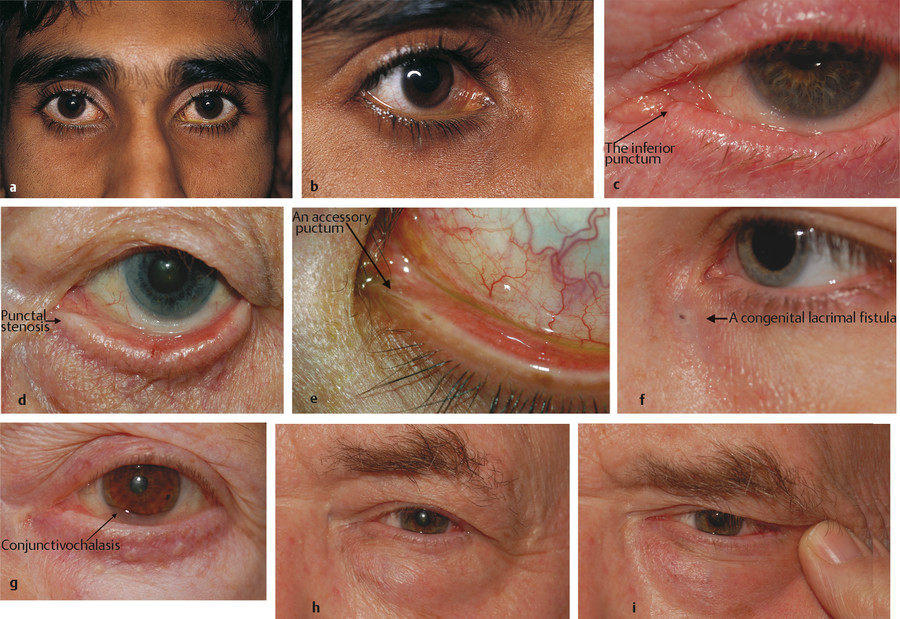
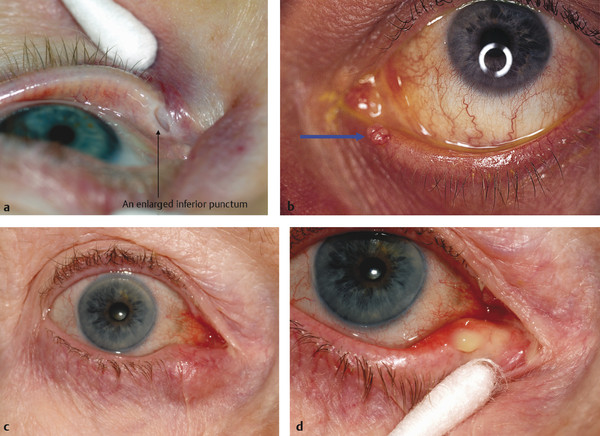
A number of abnormalities of the eyelids may be responsible for the complaints of epiphora and can be overlooked with a cursory examination (Fig. 21‑3a,b):
Lower lid ectropion (Fig. 21‑3c).
Punctal stenosis, eversion, or obstruction (Fig. 21‑3d).
Accessory puncta and/or fistulas (Fig. 21‑3e,f).
Lower lid or upper lid entropion.
Trichiasis.
Eyelid lesions (e.g., molluscum contagiosum).
Conjunctivochalasis (Fig. 21‑3g).
An incomplete reflex blink.
Aberrant reinnervation of the facial nerve (crocodile tears).
“Kissing puncta” (Fig. 21‑3h,i).
The patient should be examined closely for the presence of conjunctivochalasis, a redundant fold of conjunctiva that can prevent the access of tears to the inferior puncta (Fig. 21‑3g).
Stenosis of the inferior punctum is commonly associated with an early punctal ectropion.
A lower eyelid ectropion should be carefully evaluated as described in Chapter 6. It is important to exclude shortening of the anterior lamella or vertical eyelid tightness as causes of a cicatricial ectropion (Fig. 21‑4). Subtle changes are easily overlooked and may be related to dry, “weather-beaten” skin or a midface ptosis.
The degree of laxity of the lower eyelid and the medial and lateral canthal tendons should be assessed. The degree of horizontal lower eyelid laxity is assessed by performing a distraction test, a pinch test, and a snap test (Chapter 6).
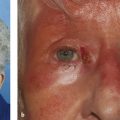
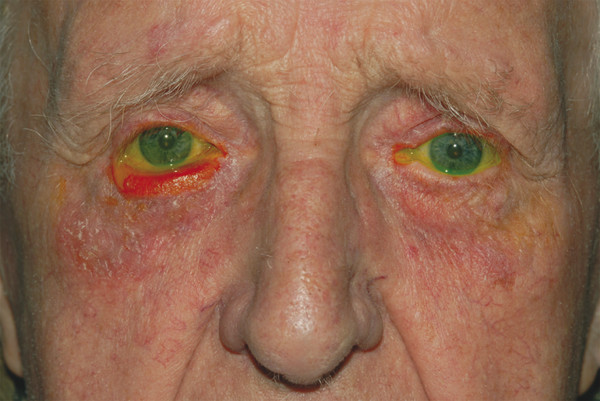
A lower eyelid entropion may be intermittent. The patient should be asked to look down and to forcibly close the eyes to ascertain whether an entropion can be provoked. This also enables an assessment of orbicularis function. The medial canthus should be palpated for any intrinsic lacrimal sac lesions, such as a lacrimal sac mucocele or tumor (Fig. 21‑5a), or an extrinsic lesion compressing the lacrimal sac, such as an ethmoid sinus tumor (Fig. 21‑5b,c).
21.4.1 Slit Lamp Examination
Biomicroscopy should be used to exclude causes of reflex hypersecretion of tears and tear film abnormalities, such as blepharitis or a dry eye. The vertical height of the tear meniscus should be noted before the instillation of any drops. The puncta should be examined carefully to ensure normal position and to exclude an early eversion, stenosis, abnormal iatrogenic enlargement (Fig. 21‑6a), or obstruction by cilia or other lesions (Fig. 21‑6b). The puncta should not normally be visible on slit lamp examination without digital manipulation to evert them. The puncta should face slightly posteriorly toward the lacus lacrimalis. All four puncta should be carefully examined to assess their presence and patency. The puncta should be examined for discharge with and without digital pressure applied to the lacrimal sac. A chronic inflammatory swelling of the eyelid medial to the puncta may indicate a canaliculitis related to an actinomyces infection (Fig. 21‑6c,d). Such a swelling should not be confused with a chalazion or meibomian cyst, which would be located within the tarsus lateral to the puncta. Pressure applied to the canaliculi may cause a cheesy material to be expressed.
The lacrimal sac should be massaged and the puncta observed for any discharge. Reflux of mucoid material is pathognomonic for a lower lacrimal drainage system obstruction.
21.5 Clinical Evaluation of the Lacrimal Drainage System
The following simple clinical tests should be performed:
Fluorescein dye disappearance test.
Syringing of the lacrimal drainage system.
Probing of the canaliculi.
Endoscopic nasal examination.
21.5.1 Fluorescein Dye Disappearance Test
The fluorescein dye disappearance test is a very simple physiological method for assessing the lacrimal drainage system and can be used in children. A drop of 2% fluorescein is instilled into the inferior fornix of each eye. Complete disappearance of the dye after a period of 4 to 5 minutes excludes any significant lacrimal drainage system obstruction. A delayed or asymmetrical dye disappearance is an indication to progress with further clinical tests of the lacrimal drainage system. (I do not use the Jones dye tests because I do not find them practical to perform or useful in clinical practice.)
21.5.2 Syringing of the Lacrimal Drainage System
Key Point
The patient should be advised that syringing of the lacrimal drainage system is not a therapeutic but a diagnostic procedure, although occasionally this can result in an improvement in symptoms. It should be performed by the surgeon evaluating the patient and never delegated to a nurse. The details of the findings should be carefully recorded.
A local anesthetic drop is instilled into the conjunctival sac and the residual fluorescein dye is washed from the conjunctival sac. The patient should be placed in a semirecumbent position on an examination couch, and an illuminated magnifier should be used. The surgeon should always stand on the side that is being syringed. Dilatation of the puncta should only be required if the puncta are stenosed. If this is necessary, it should be done with great care using a very fine Nettleship dilator to avoid creating a false passage. A fine lacrimal cannula on a 2-mL syringe of sterile saline is gently manipulated through the inferior punctum and along the inferior canaliculus with the eyelid pulled laterally. The cannula should be advanced along the correct anatomical line of the canaliculus, taking great care not to concertina the canaliculus, which creates a false impression of a canalicular or common canalicular obstruction (a common mistake).
Very gentle pressure should be applied to the syringe. Normal patency of the system will allow easy passage of saline to the nasopharynx with no regurgitation. It should be noted, however, that irrigation is applied at a far higher hydrostatic pressure than normal tear outflow. Patients with epiphora related to relative stenosis of the nasolacrimal duct may have apparently normal findings on irrigation. This is an indication for the use of lacrimal scintillography. Passage of saline to the nasopharynx after applying more pressure to the syringe suggests a partial obstruction of the lacrimal drainage system or the presence of a dacryolith.
Regurgitation of saline stained with fluorescein through the opposite punctum indicates patent canaliculi but suggests a distal obstruction. It excludes lacrimal pump failure as a cause of epiphora as the fluorescein reached the lacrimal sac. The procedure should be repeated via the upper punctum if the lower punctum is absent or if there is an inferior canalicular obstruction. Regurgitation of mucoid or mucopurulent material suggests a nasolacrimal duct obstruction. Regurgitation of blood is an indication to exclude the possibility of a lacrimal sac malignancy.
21.5.3 Probing of the Canaliculi
If syringing of the lacrimal drainage system has suggested a canalicular obstruction, careful probing of the canaliculi using a 00 Bowman probe is undertaken. The probe should not be forced through any obstructions. If concretions are present, such as after chronic actinomyces infection, the surgeon may feel a gritty sensation with the probe.
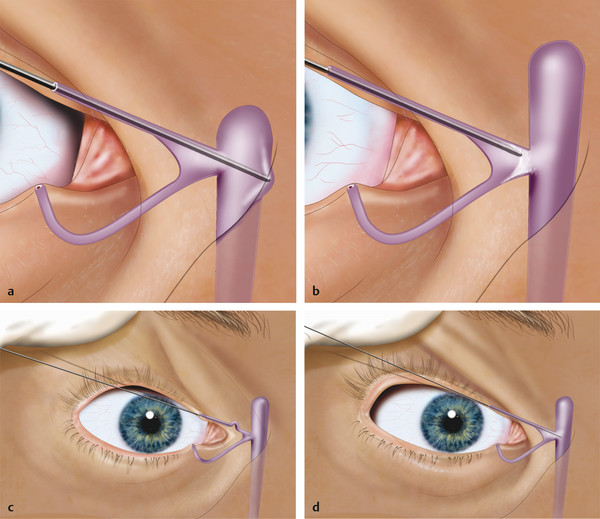
If the probe enters the lacrimal sac, a “hard stop” is felt as the probe abuts bone (Fig. 21‑7a). The presence of a “soft stop” suggests a canalicular or common canalicular obstruction as long as the probe has not been inadvertently pushed into the side wall of the canaliculus, which creates a false impression of a soft stop (Fig. 21‑7b–d).
The site of any obstruction is determined by withdrawing the probe after grasping it by the punctum with forceps and measuring the length of the probe that is withdrawn. By using this maneuver a canalicular or common canalicular obstruction may be diagnosed.
Key Point
Care should be taken to avoid the false impression of a soft stop, which can occur if the canaliculus is allowed to kink during probing. This can be avoided by ensuring that the eyelid is stretched laterally during the examination. Under no circumstances should the nasolacrimal duct be probed as a diagnostic procedure in an adult.
21.5.4 Endoscopic Nasal Examination
It is essential to perform a nasal examination before embarking on lacrimal drainage surgery. This should be performed using a rigid nasal endoscope and is undertaken to exclude intranasal pathology, such as allergic rhinitis, polyps, or tumors that may contribute to obstruction of the lacrimal drainage system, and to exclude anatomical variations that may adversely interfere with lacrimal drainage surgery, such as a deviated nasal septum or concha bullosa. Although the nose can be examined using a headlight and a nasal speculum, an endoscope provides a far superior view of the intranasal structures.
A rigid 4-mm 0-degree endoscope is ideal for most patients. Other viewing angles (e.g., 30 degrees) are not required for routine nasal endoscopy in a clinic. A rigid 2.7-mm endoscope has the advantage that it can be used routinely for adult nasal examination without the need for nasal decongestion, but it has the disadvantage that it is relatively fragile. Sterilization of endoscopes between patients is another practical problem that requires special attention for clinic organization.
Nasal Preparation
The nasal mucosa can be decongested in the office using a simple decongestant spray (e.g., oxymetazoline) or a combined decongestant and local anesthetic spray (e.g., co-phenylcaine). If a local anesthetic is used, the patient should be warned to avoid any hot drinks for an hour after the application. In children who are sufficiently cooperative, only a simple pediatric decongestant drop, such as pediatric oxymetazoline, should be used.
21.5.5 Technique of Nasal Endoscopy
Patient Preparation
The patient should be reclined at 45 degrees and should be asked to breathe gently through the mouth. If the examiner is right handed, he or she should stand to the right side of the patient to examine both the right and left nasal cavities.
Patient Examination
The tip of the endoscope should be gently wiped with cotton wool soaked in warm sterile water. The examiner should insert the tip of the endoscope very carefully into the nasal vestibule, taking great care not to touch any intranasal structures. Only after the tip of the scope has been inserted into the nose should the examiner look through the endoscope. The endoscope can then be gently advanced.
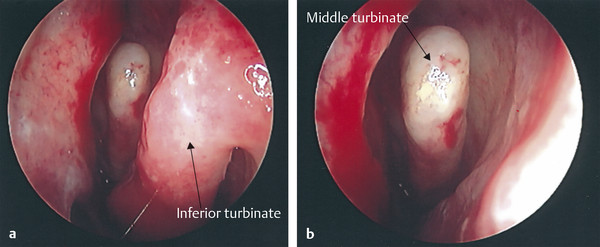
The floor of the nose can be inspected along with the lower part of the nasal septum, but it is difficult to examine the inferior meatus with a 4-mm endoscope, and if it is necessary to do so a 2.7-mm endoscope should be used. The endoscope should then be directed above the inferior turbinate where the maxillary line is seen on the lateral nasal wall, extending up to the root of the middle turbinate, the anterior part of the middle turbinate, and the middle meatus. The endoscope should not be directed to a higher position, because this is likely to induce sneezing.
21.6 Anatomical Variations of the Middle Turbinate
The middle turbinate normally curves away from the lateral wall of the nose and has a slightly curved lateral surface (Fig. 21‑8). There are a number of variations in the anatomy of the middle turbinate that should be recognized, because these will otherwise confuse the inexperienced examiner. These variations can also have implications for the surgical management of lacrimal drainage system obstructions:
Paradoxical (convex laterally rather than medially).
Concha bullosa (a pneumatized anterior portion of the middle turbinate continuous with the ethmoid air cells).
Bifid.
Duplicated.
Lateralized.
A large middle turbinate that extends anteriorly can cause problems with the correct placement of a Lester Jones (LJ) tube. A partial middle turbinectomy may have to be performed to address the problem. Great care should be taken, however, bearing in mind that the middle turbinate is attached to the skull base at the level of the cribriform plate. A twisting action should be avoided. Such a surgical procedure should only be undertaken if it is deemed necessary.
If the lateral surface of the middle turbinate is in very close proximity to or in direct contact with the lateral wall of the nose, there is an increased risk of the development of postoperative adhesions after lacrimal drainage system surgery. Such adhesions can be responsible for the occlusion of the drainage ostium after a DCR.
The inferior turbinate is also prone to some anatomical variations. The casual observer must also be wary of not mistaking a hypertrophied inferior turbinate for a nasal mass or polyp.
21.6.1 The Nasal Septum
A significantly deviated nasal septum may make lacrimal drainage surgery particularly difficult and may adversely affect the success of lacrimal bypass surgery using an LJ tube. A decision can be made after nasal endoscopy in the office about the likely requirement for a submucosal resection (SMR) of the septum. The patient can then be counseled and consented appropriately before surgery and appropriate adjustments made to the anticipated time allocated for the surgery.
21.7 Imaging
In most patients presenting with epiphora, a good history followed by a meticulous clinical evaluation alone will suffice in enabling the surgeon to make a correct diagnosis and to determine the appropriate therapeutic option for the patient (e.g., a patient with a long history of epiphora who has a lacrimal sac mucocele with no prior history of nasal surgery or trauma and a normal endoscopic nasal examination does not require further investigations). In a small proportion of patients (depending on the specialist nature of the service and the referrals), further investigations may be required. More than one investigation from the following list may be necessary to help to establish the cause of the patient’s epiphora:
DCG.
Dacryoscintigraphy.
CT.
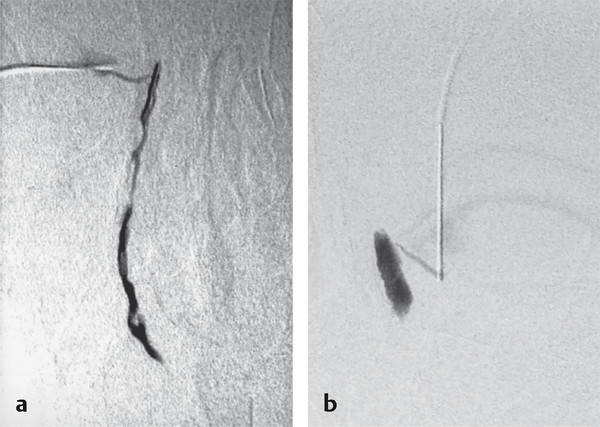
21.7.1 Dacryocystography
DCG, which involves the injection of a radiopaque dye into the lower or upper canaliculi followed by the taking of magnified radiological images, provides an anatomical assessment of the lacrimal drainage system.
Indications
Suspected lacrimal sac tumor.
Abnormal anatomy—previous trauma, craniofacial surgery, congenital anomalies.
Suspected dacryoliths in a patient patent to syringing.
Partial or functional obstruction of the nasolacrimal duct.
Superior images of the lacrimal drainage system are obtained using computerized digital subtraction DCG (Fig. 21‑9). Additional films should be obtained 10 minutes after injection of dye to evaluate dye retention.
21.7.2 Dacryoscintigraphy
Dacryoscintigraphy provides a physiological assessment of the lacrimal drainage system. The investigation involves the instillation of a radionuclide tracer into the conjunctival sac. The lacrimal system is then imaged with a gammagram. It is a more sensitive for the diagnosis of incomplete obstructions, particularly of the more proximal system. It is very rarely required.
21.7.3 Computed Tomography
CT is necessary in the following situations:
After trauma.
To evaluate a patient with a suspected lacrimal sac malignancy (Fig. 21‑1).
To evaluate an infant with a medial canthal mass.
21.8 Surgical Management: Infants and Children
21.8.1 Nasolacrimal Duct Probing and Intubation
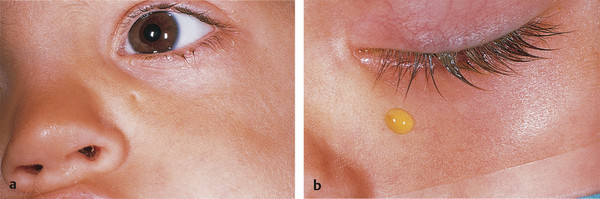
Congenital lacrimal drainage system obstruction is present in approximately 3 to 6% of newborns. Of these, approximately 0.3% are bilateral. The most common cause is a membranous obstruction of the distal end of the nasolacrimal duct, although there is a variety of rarer anatomical variations that can also cause obstruction of the duct, including nasolacrimal duct atresia, a complete bony obstruction of the duct, impaction of the inferior turbinate, a duct ending within the inferior turbinate, and nasolacrimal duct diverticula. There are a number of other congenital anomalies that must be excluded as a cause of epiphora, including punctal atresia, supernumerary puncta, congenital absence of the canaliculi, duplication of the canaliculi, and lacrimal sac fistula (Fig. 21‑10). There are also anomalies associated with facial clefts, for example, in Goldenhar’s syndrome.
Other ocular or eyelid abnormalities that can cause tearing must be excluded, including buphthalmos and distichiasis.
Most infants present with epiphora and a recurrently sticky eye that has failed to respond to topical antibiotic treatment. Pressure applied over the lacrimal sac may produce a regurgitation of mucopurulent material. A frank mucocele may be present. Dacryocystitis is unusual.
A much rarer condition of newborn infants is an amniocele. This appears as a soft bluish mass at the medial canthus below the level of the medial canthal tendon. It may simply respond to firm pressure applied to the mass if the associated membranous obstruction of the nasolacrimal duct gives way. It is important to differentiate the lesion from a meningocele or a capillary hemangioma, because these can have very similar appearances.
Patient Assessment
In most cases the diagnosis can be established from a good history and careful clinical examination. A dye disappearance test is easy to perform and causes the patient no pain or discomfort. It may demonstrate leakage through a dimple in the eyelid skin, indicating a congenital fistula. Syringing and probing can only be performed under general anesthesia. A dacryocystogram and CT may be necessary for patients with craniofacial anomalies. Dacryocystography is not required for the vast majority of pediatric patients with a congenital nasolacrimal duct obstruction, but this can be performed in the operating room before any intervention in select cases if the facility to do so is available.
Management of Congenital Nasolacrimal Duct Obstruction
Congenital nasolacrimal duct obstruction should be managed conservatively initially. The parents should be instructed to apply firm massage to the lacrimal sac in a downward direction after feeding. They must understand how to do this properly. This increases hydrostatic pressure in the lacrimal drainage system and may accelerate opening of the lower end of the nasolacrimal duct. Topical antibiotics can be prescribed intermittently. More than 90% of congenital nasolacrimal duct obstructions resolve spontaneously within the first year, with the figure increasing further by the age of 24 months. Surgical intervention should therefore be deferred until this age unless the patient is experiencing recurrent severe infections or there is an amniocele or dacryocystitis.
Probing should not, however, be deferred beyond the age of 2 years, because the effectiveness of probing decreases with age.
Key Point
More than 90% of congenital nasolacrimal duct obstructions resolve spontaneously within the first year, with the figure increasing further by the age of 24 months. Surgical intervention should therefore be deferred until this age unless the patient is experiencing recurrent severe infections or there is an amniocele or dacryocystitis.
Nasolacrimal Duct Probing
Nasolacrimal duct probing is performed under general anesthesia with the patient’s airway protected with a laryngeal mask. Oxymetazoline is sprayed into the patient’s nostril immediately after the induction of anesthesia. The nose is packed beneath the inferior turbinate with small neurosurgical patties moistened with oxymetazoline. The superior canaliculus is dilated, taking great care to avoid creating a false passage. Next, a lacrimal cannula is inserted into the midcanaliculus and a gentle irrigation of saline is performed. If there is reflux of saline, the surgeon should note which canaliculus the saline refluxes from. The cannula should then be gently advanced into the lacrimal sac, and further irrigation should be performed. The patency of the lower canaliculus should then be similarly checked.
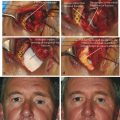
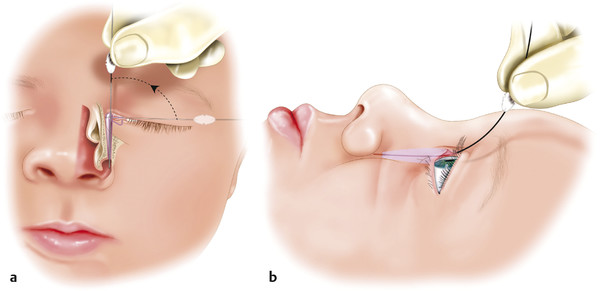
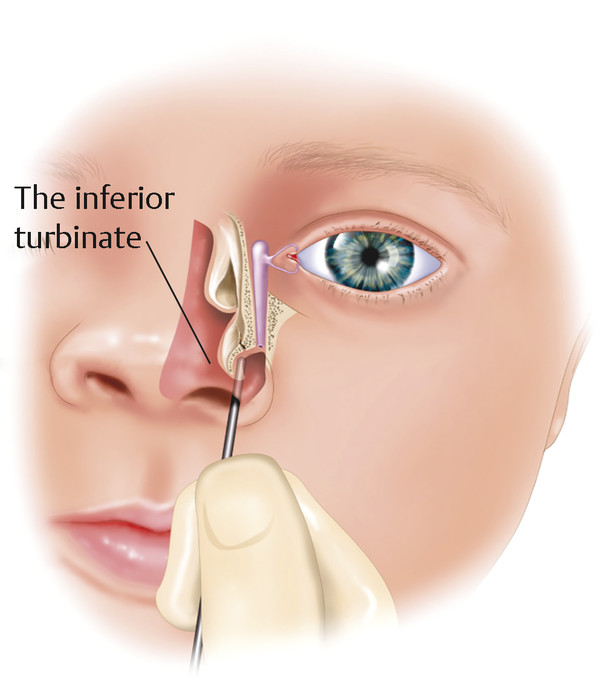
A no. 00 Bowman probe is then passed along the canaliculus while drawing the upper lid laterally, remembering the anatomical configuration of the canaliculus. Once a hard stop is felt, the probe is withdrawn minimally into the lacrimal sac and rotated 90 degrees into a vertical position and then passed inferiorly, laterally, and posteriorly, respecting the anatomical configuration of the nasolacrimal duct and without any force. The probe should lie against the infant’s brow. It is often necessary to bend the probe slightly, particularly in the presence of a prominent brow (Fig. 21‑11).
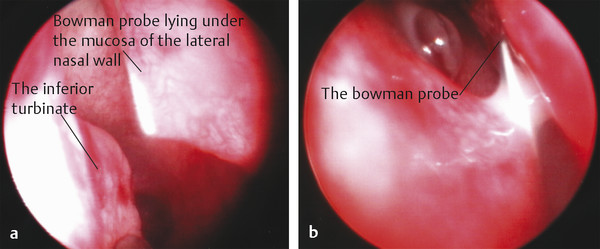
Usually the membranous obstruction is felt to give way. The probe can be visualized beneath the inferior turbinate with a small endoscope (2.7 mm, 0 degrees). This allows confirmation that a false passage has not been created through the nasal mucosa of the lateral nasal wall or inferior turbinate, a common cause of failure (Fig. 21‑12).
A second syringing is then performed using saline stained with fluorescein. Patency is confirmed by means of a simultaneous endoscopic examination. If the inferior turbinate is found to be abnormally positioned or impacted, it can be gently in-fractured using the blunt end of a Freer periosteal elevator placed under the turbinate (Fig. 21‑13). The turbinate is pushed toward the nasal septum. No postoperative ocular medications are necessary.
Management of a Failed Nasolacrimal Duct Probing
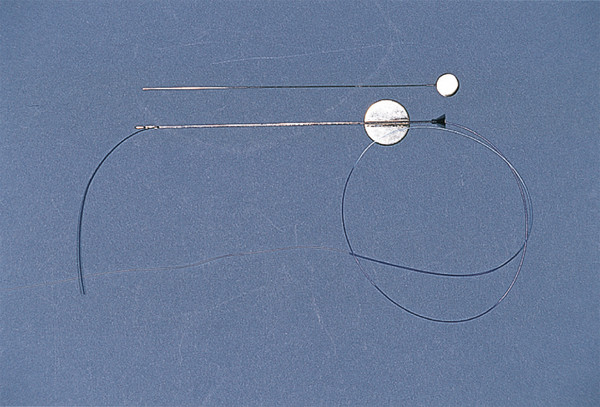
If a single probing has failed, the next step is to repeat the probing with placement of a silicone stent. The nose is packed beneath the inferior turbinate with small neurosurgical patties soaked in oxymetazoline. If necessary the nasal mucosa around the turbinate can be injected with 0.25% bupivacaine with 1:200,000 units of adrenaline. The patties are left for 5 minutes and removed. After probing the nasolacrimal duct, a Crawford silicone stent is placed, again confirming that the stent wire has passed to the correct anatomical location beneath the inferior turbinate. The olive tip of the Crawford stent is easily engaged in the end of an Anderson–Hwang grooved director or with a Crawford retrieval hook and withdrawn from the nose (Fig. 21‑14). This can be achieved in a blind fashion by the feel of metal on metal or with the use of a 2.7-mm, 0-degree endoscope.

A Ritleng stent is even simpler to remove by using a small blunt hook along the floor of the nose and hooking the nylon suture that precedes the silicone stent (Fig. 21‑15). I prefer the less rigid Crawford stent introducer.
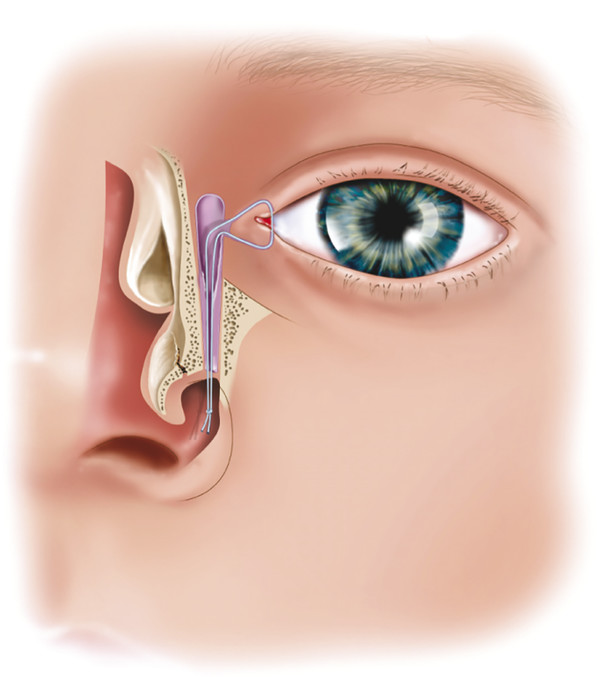
The stents are grasped with a locking Castroviejo needle holder at the tip of the inferior turbinate after spreading apart the eyelids to ensure that the stent is not under tension. A single surgeon’s knot is tied tightly. The stent is released and the ends trimmed below the knot, leaving the cut ends just visible beneath the turbinate (Fig. 21‑16). It is unnecessary to secure the stent using any other device. The position of the stent at the medial canthus is again checked. An overly tightened stent will cause cheese-wiring of the puncta. The stent is left in place for at least 6 months and removed under a very short general anesthetic. If the patient has only one patent canaliculus, a monocanalicular Crawford-style stent is used. No postoperative ocular medications are required. The use of drops or ointments is more likely to lead to a stent prolapse.
If this procedure fails or if the patient develops a dacryocystitis, an external DCR is indicated. The operation is performed in precisely the same manner as in an adult (see the next section) and can be performed relatively easily in infants. It is my preference to avoid endoscopic DCRs in most infants because the nasal space is too narrow to safely manipulate the instrumentation. In addition, the patient is not able to undertake the postoperative nasal douching that is required to aid the success of endoscopic surgery.
If a patient presents with a completely prolapsed bicanalicular silicone stent, it can be removed by rotating the knot via the inferior canaliculus before cutting the stent. It is for this reason that a single surgeon’s knot is used when tying the stent in the nose. The parents should be warned of this possibility and instructed to tape the stent to the side of the nose before making arrangements to be seen. The parents should be instructed not to cut or to pull on the stent.
21.8.2 Dacryocystocele
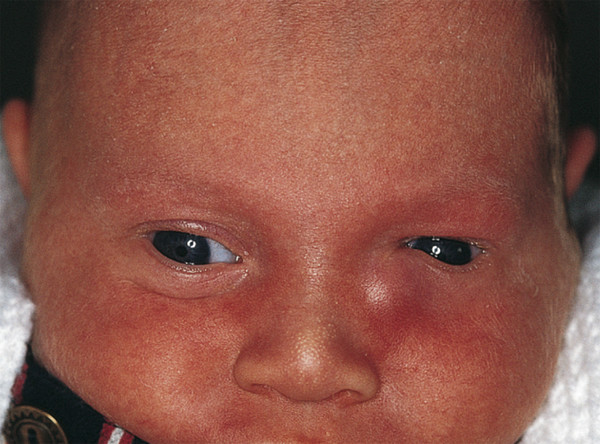
A dacryocystocele is an unusual lesion that forms in utero when there is a combination of a congenital nasolacrimal duct obstruction and a competent valve of Rosenmüller. The lacrimal sac expands, because the mucus produced within the sac is unable to escape. A medial canthal mass is visible at birth and may become infected if not treated with a degree of urgency (Fig. 21‑17).
The dacryocystocele is managed by simple probing under a short general anesthetic. It is unwise to attempt to perform the probing without general anesthesia, because this is painful and may act as a powerful stimulant to the oculocardiac reflex.
Lacrimal Sac Fistula
A lacrimal sac fistula is a rare congenital abnormality that is easily overlooked: it usually occurs in conjunction with a congenital nasolacrimal duct obstruction. The fistula may connect to the common canaliculus, the lacrimal sac, or the nasolacrimal duct. Tears may be seen emanating from a tiny hole in the inferior aspect of the medial canthus. This is more easily demonstrated with a fluorescein dye disappearance test (Fig. 21‑10). The nasolacrimal duct obstruction is managed as described previously, with placement of a silicone stent, and the fistula is completely excised.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree