Congenital haemangioma
Rapidly involuting
Non-involuting
Low-flow: CVM, CLM, CLVM, LVM
Fast-flow: CAVM, CLAVM
A, arterial; AVF, arteriovenous fistula; C, capillary; L, lymphatic; M, malformation; V, venous.
Infantile haemangiomas are unique neoplasms that proliferate in infancy and eventually involute spontaneously in childhood. They do not occur in older children or adults. The terms ‘infantile haemangioma’ or ‘haemangioma of infancy’ are typically limited to vascular tumours arising in the first few weeks of life that display a period of active growth, in the vast majority of cases, followed by a period of apparent inactivity and subsequent involution. When histological confirmation is needed, these tumours stain positively for the glucose transporter protein, GLUT1 [4], but the diagnosis is usually made clinically. For the sake of simplicity, the term ‘haemangioma’ in this chapter will refer to such lesions unless otherwise specified.
Haemangiomas can be superficial, deep or mixed and all three types of lesions undergo the same natural history. Superficial haemangiomas (formerly called ‘capillary’ or ‘strawberry’ haemangiomas) are vivid red or scarlet, sharply circumscribed plaques or nodules, whereas deep haemangiomas (formerly called ‘cavernous’) are skin-coloured or bluish purple and less well circumscribed. Mixed haemangiomas combine the features of superficial and deep tumours. Focal lesions confined to a relatively small area of the body are ‘localized’, whereas larger, plaque-like tumours that are distributed in a developmental region or territory have been termed ‘segmental’ [5]. IH can also be defined by their stage of maturation and are referred to as nascent, proliferative, involuting or involuted.
References
1 Mulliken JB, Glowacki J. Hemangiomas and vascular malformations in infants and children: a classification based on endothelial cell characteristics. Plast Reconstr Surg 1982;12:412–20.
2 Enjolras O, Mulliken JB. Vascular tumors and vascular malformations (new issues). Adv Dermatol 1997;13:375–423.
3 Garzon MC, Enjolras O, Frieden IJ. Vascular tumors and vascular malformations: evidence for an association. J Am Acad Dermatol 2000;42:275–9.
4 North PE, Waner M, Mizeracki A et al. Glut1: a newly discovered immunohistochemical marker for juvenile hemangiomas. Hum Pathol 2000;31:11–22.
5 Chiller KG, Passaro D, Frieden IJ. Hemangiomas of infancy: clinical characteristics, morphologic subtypes and their relationship to race, ethnicity and sex. Arch Dermatol 2002;138:1567–76.
Aetiology and Pathogenesis.
The pathogenesis of haemangiomas is still being determined; there is not yet a unifying mechanism that accounts for all aspects of the haemangioma phenotype nor a known single event that leads to haemangioma development. However, the patterns found in segmental haemangiomas suggest that the stage for at least some haemangiomas is set as early as 4–6 weeks of gestation, via a developmental error [1]. IH only occur in humans, and adequate animal or laboratory models have been difficult to develop. Several recently published articles comprehensively review proposed theories regarding IH pathogenesis [2–6].
The recognition that a glucose transporter protein, GLUT1, is expressed in all stages of haemangioma maturation helped spur new hypotheses on the pathogenesis of IH [7]. GLUT1 expression is absent in normal cutaneous vasculature but is found in placental blood vessels as well as in other so-called barrier tissues such as the blood–brain barrier. This, together with other immunohistochemical markers shared by IH and human placenta (see Histopathology, below), led to speculation that these tumours are of placental origin from either embolized placental cells or invading angioblasts that have differentiated towards a placental phenotype [8]. IH are unlikely to be placental emboli as they lack a villous architecture and do not express known placental trophoblastic markers [9]. A recent study determined that haemangioma endothelial cells are of fetal, not maternal origin [10]. There are marked similarities in genetic transcripts between haemangioma and placental endothelial cells, however [11], and IH and placental vasculature arise under similar physiological conditions [6], possibly suggesting a common link.
Further investigations into the cellular origin of haemangioma endothelial cells have demonstrated that these cells have features of immature mesenchymal cells. Their immunophenotype is similar to an early embryological vessel, the cardinal vein [12], and they express CD133, a primitive cell marker, during proliferation [13,14]. Implantation into immunodeficient mice of CD133+ cells isolated from IH gives rise to GLUT1+ vessels that later diminish and are replaced by adipocytes [15]. While not a perfect replica of IH growth, this model merits attention and additional study. Some studies have shown clonality [16,17] but it is not yet clear whether cells in segmental haemangiomas are clonal.
Additionally, while previous work suggested that IH arise because of aberrations in angiogenesis, recent investigations show that IH are not simply cutaneous capillaries with excessive growth but more likely represent de novo vasculogenesis in the skin and other sites. IH endothelial cells express known mediators of vasculogensis and in vitro studies indicate that hypoxia and oestrogen synergistically enhance haemangioma proliferation [18].
References
1 Haggstrom AN, Lammer EJ, Schneider RA et al. Patterns of infantile hemangiomas: new clues to hemangioma pathogenesis and embryonic facial development. Pediatrics 2006;117:698–703.
2 Boye E, Jinnin M, Olsen BR. Infantile hemangioma: challenges, new insights and therapeutic promise. J Craniofac Surg 2009;20(suppl 1): 678–84.
3 Bischoff J. Progenitor cells in infantile hemangioma. J Craniofac Surg 2009;20(suppl 1):695–7.
4 Boscolo E, Bischoff J. Vasculogenesis in infantile hemangioma. Angiogenesis 2009;12:197–207.
5 Chang EI, Chang EI, Thangarajah H et al. Hypoxia, hormones and endothelial progenitor cells in hemangioma. Lymphat Res Biol 2007;5:237–43.
6 Barnés CM, Christison-Lagay EA, Folkman J. The placenta theory and the origin of infantile hemangioma. Lymphat Res Biol 2007;5:245–55.
7 North PE, Waner M, Mizeracki A et al. GLUT1: a newly discovered immunohistochemical marker for juvenile hemangiomas. Hum Pathol 2000;31:11–22.
8 North PE, Waner M, Mizeracki A et al. A unique microvascular phenotype shared by juvenile hemangiomas and human placenta. Arch Dermatol 2001;137:559–70.
9 Bree AF, Siegfreid E, Sotelo-Avila C et al. Infantile hemangiomas: speculation on placental trophoblastic origin. Arch Dermatol 2001;137:573–7.
10 Pittman KM, Losken HW, Kleinman ME et al. No evidence for maternal-fetal microchimerism in infantile hemangioma: a molecular genetic investigation. J Invest Dermatol 2006;126:2533–8.
11 Barnés CM, Huang S, Kaipainen A et al. Evidence by molecular profiling for a placental origin of infantile hemangioma. Proc Natl Acad Sci USA 2005;102:19097–102.
12 Dadras SS, North PE, Bertoncini J et al. Infantile hemangiomas are arrested in an early developmental vascular differentiation state. Mod Pathol 2004;17:1068–79.
13 Kleinman ME, Tepper OM, Capla JM et al. Increased circulating AC133+ CD34+ endothelial progenitor cells in children with hemangioma. Lymphat Res Biol 2003;1:301–7.
14 Yu Y, Flint AF, Mulliken JB et al. Endothelial progenitor cells in infantile hemangioma. Blood 2004;103:1373–5.
15 Khan ZA, Boscolo E, Picard A et al. Multipotential stem cells recapitulate human infantile hemangioma in immunodeficient mice. J Clin Invest 2008;118:2592–9.
16 Boye E, Yu Y, Paranya G et al. Clonality and altered behavior of endothelial cells from hemangiomas. J Clin Invest 2001;107:745–52.
17 Walter JW, North PE, Waner M et al. Somatic mutation of vascular endothelial growth factor receptors in juvenile hemangioma. Genes Chromosomes Cancer 2002;33:295–303.
18 Kleinman ME, Greives MR, Churgin SS et al. Hypoxia-induced mediators of stem/progenitor cell trafficking are increased in children with hemangioma. Arterioscler Thromb Vasc Biol 2007;27:2664–70.
Histopathology.
Biopsies of both superficial and deep haemangiomas reveal relatively uniform vessel morphology [1,2]. In the proliferative stage, haemangiomas comprise syncytial aggregates of plump endothelial cells and pericytes, some of which form lumina and others solid cords. Ultrastructurally, these endothelial cells contain microtubular Weibel–Palade bodies and also demonstrate thickened multilaminated basement membranes. As involution progresses, the endothelial cells flatten and the vascular channels become more ectatic, producing large, thin-walled vessels. Islands of fatty tissue and fibrous strands gradually replace the tumour cells, giving the lesion a lobular architecture. Fully involuted IH contain few capillary-like feeding vessels and draining veins with flattened endothelium in a stroma of fatty and fibrous tissue, collagen and reticulin fibres.
In addition to endothelial cells, haemangiomas contain pericytes, fibroblasts, interstitial cells and mast cells [3]. An abundance of mast cells has been reported in both proliferating and involuting tumours but the significance of this finding is unclear [3–6]. In late-proliferating tumours, the mast cells acquire finger-like processes and appear to interact with the adjacent connective tissue cells, forming cytoplasmic bridges and membrane fusions [7].
Not surprisingly, immunohistochemical studies demonstrate that haemangiomas express many of the same markers as mature endothelial cells, such as alkaline phosphatase, factor VIII antigen, CD31, von Willebrand factor and urokinase [1,5,6]. These cellular markers have also defined distinctions between IH and vascular malformations as well as differences in the varying stages of haemangioma evolution. In the phase of rapid growth, haemangioma endothelia express large amounts of proliferating cell nuclear antigen, type IV collagenase, vascular endothelial growth factor (VEGF) and E-selectin [5,8]. In contrast, elevated expression of TIMP-1 is a specific marker for the involution phase [5]. Use of these markers in biopsy tissue could conceivably provide a means for staging haemangiomas.
Positive immunohistochemical staining for GLUT1 confirms the diagnosis of haemangioma and helps differentiate it from other vascular tumours such as tufted angioma, pyogenic granuloma and congenital haemangioma as well as vascular malformations [9]. In addition to GLUT1, haemangiomas also demonstrate intense immunoreactivity for the placenta-associated vascular antigens FcγRII, merosin and LeY [10].
References
1 Mulliken JB, Glowacki J. Hemangiomas and vascular malformations in infants and children: a classification based on endothelial characteristics. Plast Reconstr Surg 1982;69:412–20.
2 Mulliken JB, Young AG. Vascular Birthmarks. Hemangiomas and Malformations. Philadelphia: W.B. Saunders, 1988.
3 Gonzalez-Crussi F, Reyes-Mugica M. Cellular hemangiomas (‘hemangioendotheliomas’) in infants: light microscopic, immunohistochemical and ultrastructural observations. Am J Surg Pathol 1991;15:769–78.
4 Glowacki J, Mulliken JB. Mast cells in hemangiomas and vascular malforma-tions. Pediatrics 1982;70:48–51.
5 Takahashi K, Mulliken JB, Kosakewich HPW et al. Cellular markers that distinguish the phases of hemangioma during infancy and childhood. J Clin Invest 1994;93:2357–64.
6 Tan ST, Velickovic M, Ruger BM et al. Cellular and extracellular markers of hemangioma. Plast Reconstr Surg 2000;106:529–38.
7 Dethlefsen SM, Mulliken JB, Glowacki J. An ultrastructural study of mast cell interactions in hemangiomas. Ultrastruct Pathol 1986;10:175–83.
8 Kraling BM, Razon MJ, Boon LM et al. E-Selectin is present in proliferating endothelial cells in human hemangiomas. Am J Pathol 1996;148:1181–91.
9 North PE, Waner M, Mizeracki A et al. GLUT1: a newly discovered immunohistochemical marker for juvenile hemangiomas. Hum Pathol 2000;31:11–22.
10 North PE, Waner M, Mizeracki A et al. A unique microvascular phenotype shared by juvenile hemangiomas and human placenta. Arch Dermatol 2001;137:559–70.
Clinical Features.
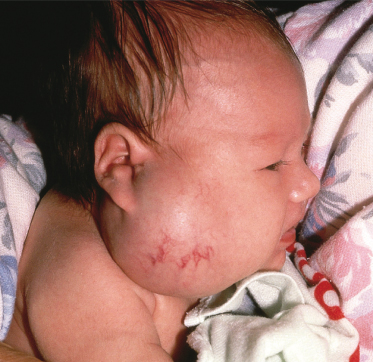
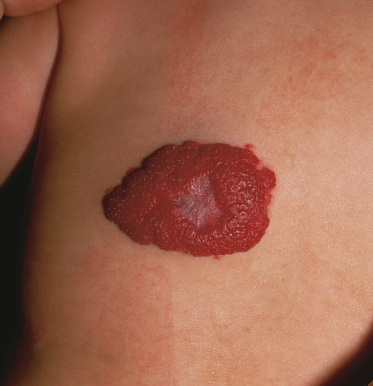
Infantile haemangiomas show considerable variation in appearance depending on their size, anatomical location, depth and stage of evolution (Figs 113.1–113.3). Approximately one-third to one-half are present at birth in some premonitory form and the remainder usually become evident during the first month of life [1,2]. The precursor lesions often appear as a blanched macule, an erythematous or telangiectatic patch with or without a pale halo, a closely packed cluster of bright-red papules, or a blue-tinged bruise-like area (Figs 113.4, 113.5) [3,4]. Precursor lesions are sometimes confused with port-wine stains or a ‘scratch’ or ‘bruise’ attributed to perinatal or postnatal trauma.
Fig. 113.4 Large precursor lesion on the back of a newborn infant, consisting of an area of pallor and telangiectases.
Courtesy of Dr James Hogan.
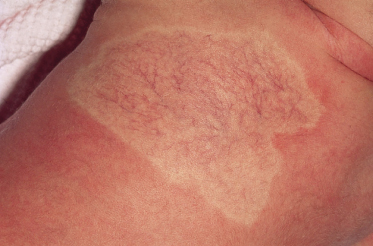
Fig. 113.5 Early, plaque-type haemangioma. Note the numerous cherry-red capillary proliferations developing within the telangiectatic patch.
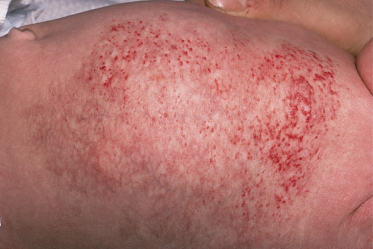
Superficial haemangiomas evolve into vivid-red, elevated, dome-shaped nodules or plaques of a rubbery consistency. Deep haemangiomas are skin-coloured or bluish, somewhat more compressible, and may have telangiectatic vessels on the surface as well as visible feeder vessels and draining veins. These tumours characteristically fluctuate in size and deepen in colour with crying or activity. Some IH have both superficial and deep components, so-called ‘mixed haemangiomas’.
Infantile haemangiomas are identified in approximately 1–2.5% of newborns [4–7] and while an incidence of 10% in the first year of life is typically quoted [7], a better estimate is 4–5% [8]. They are more common in white non-Hispanic infants [9] and females are more frequently affected than males, with a sex ratio ranging from 2 : 1 to 5 : 1 [2,3,9]. Infants with IH are more likely to be born prematurely, to be the product of multiple gestation and to have mothers older than 30 years [9]. Low birthweight (less than 2500 g) is an independent risk factor associated with IH [10]. When comparing term and preterm infants with IH, preterm infants are more likely to have multiple tumours and the sex ratio is less skewed toward females [11]. Contrary to prior observations, newer prospective studies do not support chorionic villus sampling as a major risk factor [10,11]. Although most haemangiomas occur sporadically, presumed autosomal dominant transmission of these tumours, with and without vascular malformations, has been documented in six kindreds [12]. In total, 15–30% of infants with haemangiomas have multiple lesions, usually three or fewer [13–17]. Superficial haemangiomas are the most common subtype, constituting approximately 50–60% of cases, whereas deep haemangiomas constitute about 15% and combined superficial and deep lesions 25–35% [13,15].
Infantile haemangiomas are not randomly distributed. They show a clear predilection for the head and neck region with tumours at these sites constituting at least 50% of lesions in some series [1,14,15]. The majority of facial IH are focal tumours and a minority, so-called segmental haemangiomas, affect a broader, geographic region [18,19]. Four distinct segments that correspond to facial development units have been identified [20]. Segmental IH are more likely to be associated with complications and/or structural anomalies (see Associations, below) [19].
Virtually all IH undergo a proliferative phase followed by a period of stabilization and then gradual involution. IH tend to ‘mark out’ their area early on. Subsequent changes in size correlate with increases in volume, with 80% of proliferation being completed by 5 months of age [21]. A minority of haemangiomas, particularly those with segmental morphology, a deep component and often those affecting the parotid gland, may continue to enlarge slowly for months to years longer [22].
Involution is clinically apparent in most IH at 1 year of age [21]. The first perceptible change is loss of the brilliant red colour, which is replaced by a more dull red colour in superficial haemangiomas. As this process continues, the colour becomes grey or milky-white, first centrally, then peripherally and as involution progresses the haemangioma begins to flatten and soften, gradually losing volume (Fig. 113.6). Unfortunately, an exact timetable for regression of a particular lesion cannot be predicted but involution is almost invariably slower than the proliferative phase. The time and rate of involution do not necessarily correlate with the sex of the child, or with the location, age of onset or type of haemangioma, but very small lesions do tend to complete involution sooner than very large ones [1].
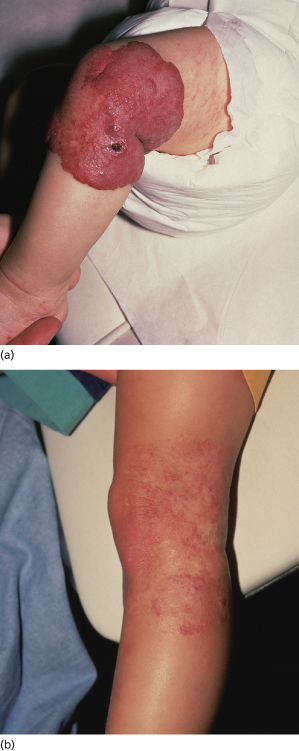
Fig. 113.6 (a) Large haemangioma on the knee of an infant. (b) The same child at 3 years of age. The haemangioma was allowed to involute spontaneously.
References
1 Finn MC, Glowacki J, Mulliken JB. Congenital vascular lesions, clinical application of a new classification. J Pediatr Surg 1983;18:894–900.
2 Mulliken JB, Fishman SJ, Burrows PE. Vascular anomalies. Curr Probl Surg 2000;37:519–84.
3 Esterly NB. Cutaneous hemangiomas, vascular stains and malformations and associated syndromes. Curr Prob Dermatol 1995;7:65–108.
4 Hidano A, Nakajima S. Earliest features of the strawberry mark in the newborn. Br J Dermatol 1972;83:138–44.
5 Pratt AG. Birthmarks in infants. Arch Dermatol 1953;67:302–5.
6 Jacobs AH, Walton RG. The incidence of birthmarks in the neonate. Pediatrics 1976;58:218–22.
7 Jacobs AH. Strawberry hemangioma, natural history of the untreated lesion. Calif Med 1957;83:8–10.
8 Kilcline C, Frieden IJ. Infantile hemangiomas: how common are they? A systematic review of the literature. Pediatr Dermatol 2008;25:168–73.
9 Haggstrom AN, Drolet BA, Baselga E et al. Prospective study of infantile hemangiomas: demographic, prenatal and perinatal characteristics. J Pediatr 2007;150:291–4.
10 Drolet BA, Swanson EA, Frieden IJ. Infantile hemangiomas: an emerging health issue linked to an increased rate of low birth weight infants. J Pediatr 2008;153:712–15.
11 Garzon MC, Drolet BA, Baselga E et al. Comparison of infantile hemangiomas in preterm and term infants: a prospective study. Arch Dermatol 2008;144:1231–2.
12 Blei F, Walter J, Orlow SJ et al. Familial segregation of hemangiomas and vascular malformations as an autosomal dominant trait. Arch Dermatol 1998;134:718–22.
13 Lampe I, Latourette HB. Management of hemangiomas in infants. Pediatr Clin North Am 1959;6:511–28.
14 Margileth AM, Museles M. Current concepts in diagnosis and management of congenital cutaneous hemangiomas. Pediatrics 1965;36:410–16.
15 Moroz B. Long-term follow-up of hemangiomas in children. In: Williams HB (ed) Symposium on Vascular Malformations and Melanotic Lesions. St Louis: C.V. Mosby, 1983: 162–71.
16 Nakayama H. Clinical and histologic studies of the classification and natural course of the strawberry mark. J Dermatol 1981;8:277–91.
17 Bowers EA, Graham EA, Tomlinson EM. The natural history of the strawberry nevus. Arch Dermatol 1960;82:667–80.
18 Waner M, North PE, Scherer KA et al. The nonrandom distribution of facial hemangiomas. Arch Dermatol 2003;139:869–75.
19 Chiller KG, Passaro D, Frieden IJ. Hemangiomas of infancy: clinical characteristics, morphologic subtypes and their relationship to race, ethnicity and gender. Arch Dermatol 2002;138:1567–76.
20 Haggstrom AN, Lammer EJ, Schneider RA et al. Patterns of infantile hemangiomas: new clues to hemangioma pathogenesis and embryonic facial development. Pediatrics 2006;117:698–703.
21 Chang LC, Haggstrom AN, Drolet BA et al. Growth characteristics of infantile hemangiomas: implications for management. Pediatrics 2008;122:360–7.
22 Brandling-Bennett HA, Metry DW, Baselga E et al. Infantile hemangiomas with unusually prolonged growth phase. Arch Dermatol 2008;144:1632–7.
Prognosis.
Although the outcomes of haemangiomas as an aggregate are excellent in the majority of cases, the wide heterogeneity of the condition mandates that prognosis be assessed individually based on location, extent and any associated complications. All haemangiomas involute but they nearly always do so at a slower rate than their growth. Approximately 50–60% will have completed involution by 5 years of age, 70–75% by 7 years of age and upwards of 90% by 9 years of age [1–3]. Precise figures regarding the percentage of children left with permanent skin changes from IH are lacking. These changes can be minor and trivial but in some patients they are severe and life-altering. Our best estimate is that at least 20% of children will have residual skin changes, including pallor, atrophy, altered skin texture or overlying telangiectasia. IH may also destroy hair follicles, resulting in partial alopecia, but many lesions of the scalp resolve without this sequela. More significant changes, such as redundant skin and fibrofatty residuum, may occur if the tumour had a predominantly exophytic dermal component, especially if it was sharply demarcated from the surrounding skin. Scarring is a virtual certainty if ulceration has occurred, although the location of the lesion and the extent of ulceration may affect the prominence of the residual scar.
References
1 Lampe I, Latourette HB. Management of hemangiomas in infants. Pediatr Clin North Am 1959;6:511–28.
2 Margileth AM, Museles M. Current concepts in diagnosis and management of congenital cutaneous hemangiomas. Pediatrics 1965;36:410–16.
3 Bowers EA, Graham EA, Tomlinson EM. The natural history of the strawberry nevus. Arch Dermatol 1960;82:667–80.
Complications.
While the majority of IH are localized and pose no immediate or long-term threat, a significant minority develop complications. Haggstrom et al. [1] noted that 24% of patients with IH referred to tertiary paediatric dermatology centres had complications. Large size, segmental morphology and facial and perineal location are associated with increased likelihood of complications [1]. Early recognition of potentially problematic lesions, coupled with prompt intervention, may help to minimize future problems.
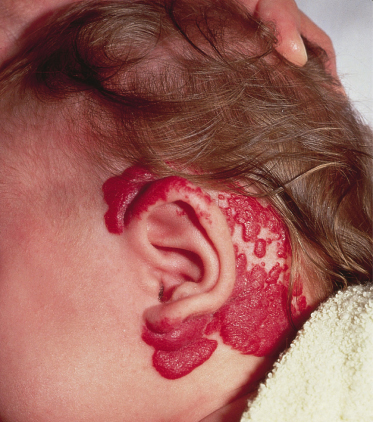
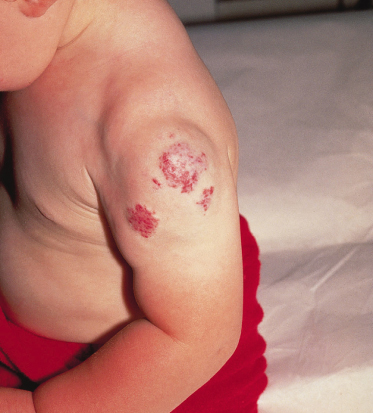
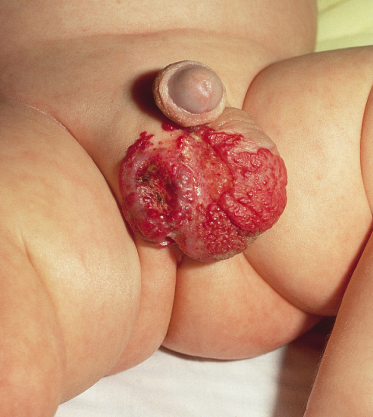
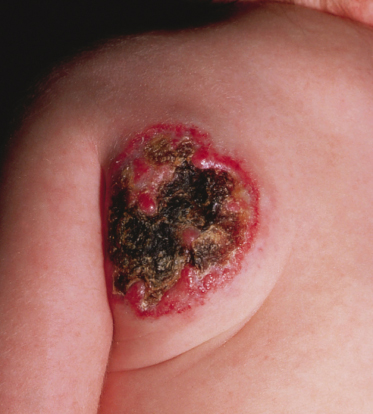
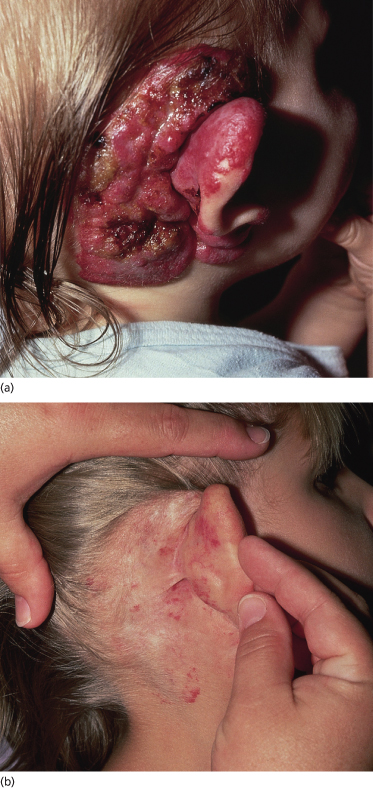
Ulceration is the most common complication, occurring in approximately 15% of haemangiomas [2] (Figs 113.7–113.10). Characteristics associated with ulceration are large size, segmental morphology, superficial or mixed subtype and location on the lower lip, neck, or perineal area [2–4]. Ulceration typically occurs during proliferation, with a median age of 4 months, and infants with ulcerated IH are likely to be referred to a specialist at a younger age [2]. Ulceration causes considerable pain, particularly when the lips, genitalia, perirectal area or flexures are involved. It almost invariably results in scar formation, causing disfigurement and, in some locations, functional impairment. Intervention is indicated to control pain, hasten re-epithelialization and prevent infection, although no single treatment modality is uniformly effective and studies comparing the efficacy of different treatment approaches are lacking.
Fig. 113.10 (a) Ulcerated haemangioma in an infant several months of age. (b) The same child at 5 years of age. No treatment was given.
Local wound care is indicated in all cases. Gentle cleansing with lukewarm water or compresses helps to debride crusts or scabs. Generous application of an antibiotic ointment and/or petrolatum with or without a non-adhesive dressing may help induce re-epithelialization [4]. In areas such as the perineum, where the adherence of a dressing is difficult, use of petroleum jelly-impregnated gauze or a barrier cream may suffice. Occlusive dressings help to protect the ulcer from further trauma and the entry of irritating substances, thus alleviating pain [5]. Similar results may be attained with the use of a polyurethane film dressing [6]. Alternatively, the pulsed dye laser (PDL) can be an effective stimulus for re-epithelialization and is recommended as an early intervention by some authors [7–9]. Healing usually occurs within 2 weeks but larger ulcers may require subsequent treatments at 2–4-week intervals [7–9]. In a minority of patients, however, the ulceration does not respond to PDL or may actually worsen after treatment [4]. The use of topical becaplermin (recombinant human platelet-derived growth factor, BB) for ulcerated haemangiomas has been reported to be very effective in a small number of cases [10]. A boxed warning issued by the United States Food and Drug Administration due to studies suggesting increased cancer-related mortality (though not cancer incidence) in adult patients has raised concerns about its use, though the relevance of such information for young infants with ulcerated haemangiomas is less than clear. In general, becaplermin is not used first-line but rather reserved for ulcerated IH that have failed other treatment modalities [11].
While bacterial colonization of ulcerated haemangiomas is relatively common, actual infection is infrequent. When it does occur, there may be increased redness, swelling, tenderness and purulent drainage. Such infections are usually localized to the skin but more invasive spread to deeper tissues or the bloodstream can occur [12,13]. Polymicrobial colonization with a mixture of aerobic and/or anaerobic bacteria is common. The most common aerobic pathogens are Staphylococcus aureus and group A β-haemolytic streptococci, followed by organisms populating mucous membranes near the IH [14].
Localized infection will often improve with the use of topical antibiotics such as mupirocin ointment or metronidazole gel. The empirical use of systemic antibiotics, such as cephalexin, is indicated if localized infection fails to improve or if cellulitis is present, pending culture results. Life-threatening septicaemia may supervene, particularly if infection is not detected early and treated properly. Group A β-haemolytic streptococci are frequently incriminated in these instances [12,13].
With rare exceptions, bleeding from an eroded or traumatized haemangioma in the absence of a coagulopathy is usually insignificant and responds to direct pressure. Nevertheless, it is distressing to parents, who often need reassurance and specific instructions for handling this problem. The use of an enuresis blanket, which will detect even small amounts of bleeding during the night, may relieve parental anxiety [15]. Caretakers should be instructed to report evidence of bleeding in a timely fashion and should be made aware of signs of blood loss, such as pallor or fatigue. In the event of uncontrolled or persistent bleeding, the infant should be taken to an emergency room for haematological evaluation and additional therapeutic measures.
Symptomatic relief from the pain of an ulcerated haemangioma may often be attained with the simple measures of occlusive dressings and/or PDL treatments. In addition, oral medications such as acetaminophen with or without codeine may be used until healing begins. The sparing application of lidocaine ointment may be helpful in temporarily alleviating pain as well. Application of a pea-sized amount no more than four times daily appears to be safe, even in very young infants [4]. Owing to the risk of side-effects, parents must be cautioned regarding overuse of this medication and educated about the signs and symptoms of toxicity.
Ophthalmological complications occur in up to 80% of patients with untreated periocular haemangiomas [16]. The most common complication is anisometropia which can occur due to obstruction of the visual axis (due to ptosis or mass effect), compression or displacement of the globe leading to asymmetrical refractive errors, or due to strabismus [17]. Abnormal visual input during the critical period when normal vision is required for visual development can result in permanent amblyopia. Although the age at which infants are most susceptible and the amount of time necessary to develop amblyopia are not precisely known, animal studies suggest that even a few days of abnormal visual input within the first few months of life can result in permanent visual defects [17].
Periocular IH less than 1 cm in size are unlikely to cause complications, while 53% of localized tumours greater than 1 cm and 78% of diffuse eyelid tumours produce amblyopia [18]. Tumour location also seems to be an important predictor of amyblopia, with lesions affecting the nasal third of the eyelid associated with the highest risk, central lesions with moderate risk and temporal lesions with the lowest risk [18]. Upper eyelid tumours constitute approximately 75% of periocular IH and are more likely to be associated with complications but problems can occur with lower lid lesions as well [19]. Other less common complications of periocular IH include proptosis and exophthalmos which are signs of deeper orbital involvement. Orbital involvement can cause compression of the optic nerve in rare cases [17]. It is mandatory that patients with periocular haemangiomas undergo an ophthalmological assessment early in their course and periodically thereafter in order to detect and treat visual impairment.
Sequelae from airway haemangiomas are among the most alarming of all potential complications. Sites of involvement include both supra- and subglottic mucosa but subglottic location is most associated with airway compromise. Infants with subglottic disease typically present at between 6 and 12 weeks of age with noisy breathing, hoarse cry, inspiratory or biphasic stridor and, if severe, cyanosis [20,21]. Approximately 50% of these infants have IH elsewhere on the body, with the segmental mandibular and neck (‘beard’) area being strongly associated with symptomatic airway disease [22]. The airway haemangiomas themselves are most often segmental but can be more focal [23]. Definitive diagnosis is made by direct laryngoscopy. Biopsy is warranted if the diagnosis is not clear as airway IH histologically and immunohistochemically mirror cutaneous IH and express GLUT1 [24]. Treatment modalities have ranged from corticosteroids (intralesional or systemic) to laser ablation and surgical excision [25] and early reports of using propranolol for this indication are also favourable [26,27]. Some infants have required tracheostomy as a temporizing measure. Extrinsic compression from very large, bulky cervicofacial haemangiomas can occasionally result in feeding difficulties or sleep apnoea.
Cardiac failure is a rare complication most often associated with hepatic haemangiomas (see Hepatic haemangiomas, below) and occasionally with very large haemangiomas at other sites [28]. High-output congestive heart failure may respond to digitalis and diuretics but ultimately requires reduction of blood flow by drug-induced regression, embolization or excision of the haemangioma.
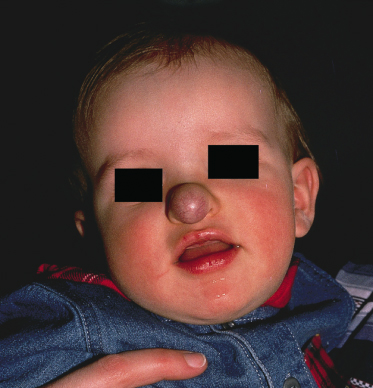
Disfigurement, particularly from extensive and rapidly expanding IH of the head and neck, can be a serious problem and is an important reason for intervention. Certain locations are particularly prone to leaving permanent skin changes that can be problematic, particularly from a psychosocial standpoint [29,30]. Haemangiomas of the nasal tip (Cyrano nose, Fig. 113.11) are a case in point as they can significantly distort the nasal contour, causing permanent nasal deformity as well as tremendous distress and embarrassment to the child and family. The lips are another central facial site where even relatively small haemangiomas can leave permanent scarring. Early intervention with systemic agents, intralesional steroids or surgery may help to minimize disfigurement and psychosocial distress [31].
References
1 Haggstrom AN, Drolet BA, Baselga E et al. Prospective study of infantile hemangiomas: clinical characteristics predicting complications and treatment. Pediatrics 2006;118:882–7.
2 Chamlin S, Haggstrom A, Drolet B et al. Multicenter prospective study of ulcerated hemangiomas. J Pediatr 2007;151:684–9.
3 Hermans DJJ, Boezeman JBM, van de Kerkhof PCM et al. Differences between ulcerated and non-ulcerated hemangiomas, a retrospective study of 465 cases. Eur J Dermatol 2009;19:152–6.
4 Kim HJ, Colombo M, Frieden IJ. Ulcerated hemangiomas, clinical characteristics and response to therapy. J Am Acad Dermatol 2001;44:962–72.
5 Yan A. Pain management for ulcerated hemangiomas. Pediatr Dermatol 2008;25:586–9.
6 Oranje AP, de Waard-van der Spek FB, Devillers ACA et al. Treatment and pain relief of ulcerative hemangiomas with a polyurethane film. Dermatology 2000;200:31–4.
7 Morelli JG, Tan OT, Yohn JJ et al. Treatment of ulcerated hemangiomas in infancy. Arch Pediatr Adolesc Med 1994;148:1104–5.
8 Morelli JG, Tan OT, Weston WL. Treatment of ulcerated hemangiomas with the pulsed tunable dye laser. Am J Dis Child 1991;145:1062–4.
9 David L, Malek M, Argenta L. Efficacy of pulse dye laser therapy for the treatment of ulcerated haemangiomas: a review of 78 patients. Br J Plast Surg 2003;56:317–27.
10 Metz BJ, Rubenstein MC, Levy ML et al. Response of ulcerated perineal hemangiomas of infancy to becaplermin gel, a recombinant human platelet-derived growth factor. Arch Dermatol 2004;140:867–70.
11 http://novanews.org/index.php?s=regranex.
12 Yagupsky P, Giladi Y. Group A β-hemolytic streptococcal septicaemia complicating infected hemangioma in children. Pediatr Dermatol 1987;4:24–6.
13 Armstrong AL, Finch RG, Bailie FB. Serious group A streptococcal infections complicating cryotherapy to lip hemangiomas. Clin Exp Dermatol 1993;18:537–9.
14 Brook I. Microbiology of infected hemangiomas in children. Pediatr Dermatol 2004;21:113–16.
15 Mallory SB, Morris P. Bleeding hemangioma detected by enuresis blanket. Pediatr Dermatol 1989;6:139–40.
16 Stigmar G, Crawford JS, Ward CM et al. Ophthalmic sequelae of infantile himangiomas of the eyelids and orbit. Am J Ophthalmol 1978;85:806–13.
17 Ceisler EJ, Santos L, Blei F. Periocular hemangiomas: what every physician should know. Pediatr Dermatol 2004;21:1–9.
18 Schwartz SR, Blei F, Ceisler E et al. Risk factors for amblyopia in children with capillary hemangiomas of the eyelids and orbit. J AAPOS 2006;10:262–8.
19 Haik BG, Jacobiec FA, Ellsworth RM et al. Capillary hemangioma of the lids and orbit: an analysis of the clinical features and therapeutic results in 101 cases. Ophthalmology 1979;83:760–89.
20 Shikani AH, Marsh BR, Jones MM et al. Infantile subglottic hemangiomas: an update. Ann Otol Rhinol Laryngol 1986;95:336–47.
21 Riding K. Subglottic hemangioma: a practical approach. J Otolaryngol 1992;21:419–21.
22 Orlow SJ, Isakoff MS, Blei F. Increased risk of symptomatic hemangiomas of the airway in association with cutaneous hemangiomas in a ‘beard’ distribution. J Pediatr 1997;131:643–6.
23 O TM, Alexander RE, Lando T et al. Segmental hemangiomas of the upper airway. Laryngoscope 2009;119(11):2242–7.
24 Badi AN, Kerschner JE, North PE et al. Histopathologic and immunophenotypic profile of subglottic hemangioma: multicenter study. Int J Pediatr Otorhinolaryngol 2009;73:1187–91.
25 O-Lee TJ, Messner A. Subglottic hemangioma. Otolaryngol Clin North Am 2008;41:903–11.
26 Denoyelle F, Leboulanger N, Enjolras O et al. Role of propranolol in the therapeutic strategy of infantile laryngotracheal hemangioma. Int J Pediatr Otorhinolaryngol 2009;73:1168–72.
27 Buckmiller L, Dyamenahalli U, Richter GT. Propranolol for airway hemangiomas: case report of novel treatment. Laryngoscope 2009;119:2051–4.
28 Vaksman G, Rey C, Marache P et al. Severe congestive heart failure in newborns due to giant cutaneous hemangioma. Am J Cardiol 1987;60:392–4.
29 Dieterich-Miller C, Cohen B, Liggett J. Behavioral adjustment and self-concept of young children with hemangiomas. Pediatr Dermatol 1992;9:241–5.
30 Tanner JL, Dechert MP, Frieden IJ. Growing up with a facial hemangioma: parent and child coping and adaption. Pediatrics 1998;101:446–52.
31 Frieden IJ. Which hemangiomas to treat: and how? Arch Dermatol 1997;133:1593–5.
Differential Diagnosis.
Although numerous conditions can mimic haemangiomas [1,2], the diagnosis is usually made easily on the basis of the history and clinical findings. In a minority of cases, however, the true nature of the lesion may be in question and arriving at the correct diagnosis is critical, particularly when complications arise or if surgery is contemplated.
Haemangiomas may be mistaken for other types of birthmarks or neoplasms, depending on their stage of development. Nascent lesions may be confused with naevus anaemicus or naevus depigmentosus, capillary malformations or traumatic injury (a scratch or bruise). Superficial haemangiomas may resemble other vascular tumours such as pyogenic granulomas, congenital haemangiomas, tufted angiomas, kaposiform haemangioendotheliomas or haemangiopericytomas. Deeper tumours may mimic dermoid cysts, nasal gliomas, infantile myofibromatosis, lipomas, plexiform neurofibromas, neuroblastomas or other soft tissue sarcomas. Perhaps the greatest difficulty lies in the distinction from vascular malformations (either venous or lymphatic).
The correct diagnosis relies upon the typical natural history of the condition, particularly absence at birth or a premonitory mark followed by a period of rapid growth during the first month of life. IH are rarely present as a fully formed soft tissue mass. They typically complete growth by 1 year of age, though there are some exceptions [3]. The appearance of vascularity, particularly telangiectasias, should not be assumed to be definitive for IH as many rapidly expanding tumours, such as rhabdomyosarcoma, may also have overlying telangiectasias. Similarly, the imaging findings of high vascular flow should not be considered diagnostic, but the specific characteristics of IH have been well described in the radiology literature (see discussion below). Multifocal vascular-appearing lesions are often assumed to represent IH and the term ‘disseminated neonatal hemangiomatosis’ has been applied to this clinical scenario, particularly when visceral involvement is present. However, a number of other conditions including multifocal lymphangioendotheliomatosis and pyogenic granuloma-like growths can resemble multiple haemangiomas. Biopsy of these lesions for analysis with GLUT1 staining may be needed to help differentiate these conditions from multifocal IH [1].
References
1 Frieden IJ, Rogers M, Garzon MC. Conditions masquerading as infantile haemangioma: Part 1. Australas J Dermatol
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree