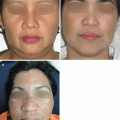
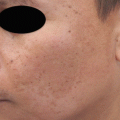
Topicals
Chemical peels
Lasers and lights
Transdermal drug delivery modes
Retinoids
Glycolic acid
ND:Yag (QS and long pulsed)
Skin needling
Hydroquinone
Salicylic acid
QS ruby
Microneedling
Corticosteroids
Jessner’s
QS alexandrite
Iontophoresis
Triple-combination cream
Mandelic acid
Non-ablative lasersa
Monopolar radiofrequency
Novel skin-lightening agents:
Botanicals
Vitamins
Alternative
Trichloroacetic acid
Intense pulse light:
Fractionated
Pulse-in-pulse mode
Tranexamic acidb
Retinol
Ablative lasersa
Kojic acid
Variable pulsed light
Azelaic acid
Pulsed dye laser
Studies have shown varied clinical benefits of TA. A double-blind, randomized, controlled, split-face clinical trial investigated topical TA 5 % compared to vehicle and found no significant difference in Melasma Area and Severity Index (MASI) score reduction on both sides, with TA also causing erythema [16]. Another study compared TA 3 % to HQ 3 % with dexamethasone 0.01 % in a double-blind, split-face trial and found that both decreased MASI scores, but there was no significant difference between the improvements in the two groups [17]. Topical liposomal tranexamic acid 5 % has been studied in comparison to HQ 4 % in a split-face trial in which both topicals were applied twice daily and both resulted in significantly reduced MASI scores [13].
Oral TA given as two pills, twice a day for 6 months in a Chinese study of 74 patients, resulted in nearly 64 % of the patients having a good to excellent response with no significant complications [18]. A larger study of oral TA, given as 250 mg twice daily for 3 months, found that patients who took the medication had a significant reduction in MASI [19]. Oral TA has also been studied in combination with topical TA. In one study, patients took oral TA (two tablets, three times daily) and used topical tranexamic acid twice daily for 8 weeks, and the subjects had decreased epidermal pigmentation and erythema, as well as a decrease in dermal melasma changes including vessel number and mast cell count [20].
TA has also been studied in a microinjection form and with microneedling. In a randomized, open-label study comparing these two treatment modalities, both were found to improve MASI scores with greater improvement in the microneedling group, which may have been due to deeper and more uniform delivery of the TA through the microchannels created by microneedling [21].
Side effects of TA can include nausea, diarrhea, orthostatic reactions, hypomenorrhea, and, rarely, skin reactions, acute renal cortical necrosis, and color vision disturbances [14, 18].
In the future, as further studies are done on TA, it may become a more commonly used treatment for those with melasma.
33.1.4.2 New Drug Delivery Vehicles
New vehicles for topical drug delivery are being investigated. Ethosomes® and Transferomes® have been studied in vitro for topical drug delivery of compounds such as linoleic acid which are used to treat disorders of hyperpigmentation [22]. By using these drug delivery carriers for topical medications, there may be increased skin penetration past the stratum corneum, which may lead to improved local and systemic delivery, as well as higher clinical efficacy.
New conjugates of traditional melasma medications may also be on the horizon. Treatments that typically cause skin irritability, such as salicylic acid (SA) and HQ, have been combined into new formulations which have higher lipophilicity, less aqueous solubility, and lower crystallinity than each medication separately [23]. These new conjugates may improve skin absorption and skin tolerance.
33.1.4.3 Novel Skin-Lightening Agents
Novel skin-lightening agents are also being developed, many of which have shown promising results. The number of studies done on these newer agents however is very limited, and therefore larger studies will be needed to truly determine their safety and efficacy. One of these agents, undecylenoyl phenylalanine, an antagonist to alpha-melanocyte-stimulating hormone and beta-adrenergic receptors, has been found to significantly lighten melasma with minimal side effects [24]. Given that hormones are thought to play a role in the pathogenesis of melasma, topical flutamide 1 %, an antiandrogenic agent previously used for hair loss, has been studied in an RCT and was found to be as effective as topical HQ 4 % in improving melasma by mexameter assessment and was superior to HQ when comparing MASI scores [25]. Cysteamine, an intrinsic antioxidant that protects against ionizing radiation and that can inhibit melanin synthesis, was used topically in melasma patients and in comparison to placebo was found to significantly reduce MASI scores at 2 and 4 months [26].
33.1.4.4 Alternative Topical Therapies
Natural ingredients have been gaining popularity in the treatment of pigmentary disorders including melasma. Topical lignin peroxidase, a purified active enzyme derived from the fermented fungus Phanerochaete chrysosporium, has been studied in a 9-week single-center, open-label, prospective study in 31 Chinese women and was found to improve melasma through decrease in skin pigmentation and improvement in skin luminance [27]. Rumex occidentalis, an Asian herb that inhibits tyrosinase activity and blocks melanin formation, was studied in a randomized double-blind placebo-controlled trial in which 45 subjects either received 3 % R. occidentalis cream or 4 % HQ cream, and both were found to be comparable in efficacy and safety [28]. Plant extracts containing catechins/polyphenols, which have inhibitory effects on tyrosinase, have resulted in a significant decrease in melanin levels when compared to placebo [29]. Another plant-derived ingredient, silymarin, derived from the milk thistle plant, contains silybin, a potent antioxidant with photoprotective properties and the ability to inhibit L-dopa oxidation activity of tyrosinase. When studied in 96 melasma patients, a significant improvement was seen in a dose-dependent manner along with prevention of UV-induced skin damage and no significant side effects [30]. Mulberry extract, which has flavonoids and antioxidant properties as well as tyrosinase inhibiting properties, was evaluated in a randomized, single-blind, placebo-controlled trial and was found to significantly improve MASI and quality of life scores [31]. Topical niacinamide (4 %), which has anti-inflammatory properties and can decrease melanosome transfer, has been studied against HQ 4 % in an 8-week, 27-subject study and was found to similarly improve pigmentation in melasma as HQ, but also had the added benefit of reducing mast cell infiltrate and solar elastosis [32].
Various skin-brightening ingredients are also being marketed into commercial products that are hydroquinone free. A topical cream with disodium glycerophosphate, L-leucine, phenylethyl resorcinol, and undecylenoyl phenylalanine was used to treat mild to moderate melasma twice daily for 12 weeks in 20 female patients and resulted in a significant reduction in melasma severity and appearance with high tolerability [33]. Methimazole, an antithyroid medication, was formulated into a 5 % topical cream and used once daily in two patients with HQ-resistant melasma and after 8 weeks led to significant melasma improvement with high tolerability and no significant side effects [34].
33.1.4.5 Chemical Peels, Lasers, and Lights
Various chemical peels, lasers, and lights continue to be studied as treatment modalities for improving melasma [35]. Chemical peels recently studied include glycolic acid (GA), salicylic acid (SA), Jessner’s, mandelic acid (MA), and trichloroacetic acid (TCA) [36, 37]. Chemical peels have also been used in combination with topical creams.
Lasers and lights recently studied include Q-switched (QS), ablative, and non-ablative lasers that are fractional and non-fractional, long pulsed ND:Yag, intense pulsed light (IPL), copper bromide, and pulse dye lasers (PDL) [38–42]. Q-switched lasers, including ND:Yag, ruby, and alexandrite lasers, are continuing to be studied at various settings in different skin types with varied success rates [43, 44]. Although some laser and light treatments have caused improvement in melasma, other studies have found worsening rebound melasma, postinflammatory hyperpigmentation (PIH), and/or hypopigmentation.
Ablative and non-ablative lasers including CO2 and Erbium Yag in fractional and non-fractional forms have also shown promise in melasma treatment particularly in targeting dermal melasma [45, 46]. The use of ablative lasers, however, has been more limited due to the increased risk of PIH and difficulty in maintaining long-term results [47].
A new type of IPL, called fractionated IPL, has recently been studied as a weekly treatment for melasma in Asian women and, when compared to biweekly conventional IPL, was found to result in decreased MASI scores with no melasma rebound [48]. Another new type of IPL uses a pulse-in-pulse mode, which releases multiple fractionated subpulses in one pulse width [49]. When compared to traditional IPL, both significantly decreased MASI scores, with the former being preferred by patients due to decreased discomfort during and after treatments. Another light treatment similar to IPL called variable pulsed light is also being investigated. Unlike IPL, it disperses energy as a few small, rapid microflashes instead of one flash of light. It offers the advantage of decreased pain and better absorption into select layers of the skin with decreased amounts of heat entering neighboring tissues [50].
Lasers targeting the vascular component of melasma are also garnering interest. Two recent studies examining the use of copper bromide laser which has wavelengths of 578 nm and 511 nm targeting vascular lesions and pigmentation, respectively, have found the laser to not be efficacious in treating melasma and when compared to Kligman’s combination cream formula , the latter was found to be more effective [51, 52]. In contrast, another report of a woman treated with PDL and Kligman’s formula found that the PDL treatment prevented recurrence of melasma in treated areas [2].
33.1.4.6 Laser Combination Treatments
Combination treatments for melasma continue to show promise in further improving melasma and helping those with refractory melasma. Traditional topical treatments are being studied in combination with other topical and procedural treatments and have been found to have various degrees of therapeutic efficacy.
QS ND:Yag 1064 nm laser has been studied in combination with several other treatments. When used with IPL, it has been found to be more effective, particularly for refractory melasma and then when each is used as a monotherapy [39, 53, 54]. When QS ND:Yag laser toning has been combined with vitamin C ultrasonic application, it has led to higher physician and patient assessments [55]. QS ND:Yag laser done after microdermabrasion and maintained with a topical regimen of tretinoin or vitamin C along with sunscreen has been shown to be efficacious in inducing melasma remission for up to 6 months with minimal risk and recovery time [56].
IPL and fractional erbium glass laser, each combined with TCC, have shown greater efficacy together than when each treatment is used alone [57, 58]. Another recent study found that patients treated with oral TA, IPL, and QS ND:Yag laser treatments compared to those who had the laser and light treatments without TA found that the patients in the former group had improved clinical efficacy without significant side effects [59]. This result was supported by another Korean study, which examined the use of oral TA in patients receiving low fluence (LF) 1064-nm QS ND:Yag and found that those in the combination group had overall greater clinical improvement than those who had the laser treatment alone [60].
33.1.4.7 Transdermal Drug Delivery: Needling, Iontophoresis, and Energy Devices
Additional treatments being investigated for melasma include modes of transdermal drug delivery including skin needling, microneedling, iontophoresis, and radiofrequency. Skin needling has been found to enhance the penetration of topical products, and in a study combining skin needling with a depigmenting serum (containing rucinol and sophora-alpha), the combination resulted in significant improvement of the melasma in comparison to the use of the serum alone [61]. Iontophoresis can increase drug penetration of a compound into the skin in a controlled fashion by applying current through the tissue [62]. Vitamin C iontophoresis has been used successfully in the treatment of melasma and when used in a nanosome formulation has even been found to have better clinical results than 70 % GA [63]. It has also shown efficacy in melasma treatment when followed by a home maintenance regimen consisting of MA [39].
Platelet-rich plasma (PRP) has recently been studied in a single patient who underwent three treatments and had regression of her melasma with greater than 80 % reduction in epidermal pigmentation [64]. The mechanism of action is not fully understood but may involve the effects of TGF-β1 on inhibition of melanin synthesis along with an increase in skin volume.
Energy devices, such as monopolar radiofrequency, have also been combined with transdermal drug delivery of a product containing kojic acid 1 % and were found to decrease MASI scores, average melanin, and erythema with no significant side effects [65].
33.2 Vitiligo
33.2.1 Overview
Vitiligo is a relatively common acquired, idiopathic pigmentary disorder characterized by one or more depigmented patches of the skin. The disorder can be cosmetically disfiguring and severely impacts quality of life. Patients with vitiligo often experience low self-esteem, depression, stigmatization, and isolation [66, 67]. The mechanisms involved in the destruction of melanocytes in vitiligo are complex and include genetic influences, self-destruction, oxidative stress, neural defects, and autoimmunity. Current research places autoimmune and oxidative stress at the forefront of mechanisms mediating the destruction of melanocytes in vitiligo.
33.2.2 Treatment Strategies
New approaches for vitiligo address stabilization of the disease as well as repigmentation. While no consensus is available on the concept of stabilization in vitiligo, the vitiligo disease activity (VIDA) score has been utilized in several publications. It is a 6-point scale in which the activity of the disease is evaluated based on the appearance of new vitiligo patches, or the enlargement of preexisting patches assessed during a period ranging from less than 6 weeks to 1 year [68]. New therapies for stabilization of the disease include minocycline, methotrexate, and narrowband UV phototherapy (NB-UVB) (Table 33.2). Emerging therapies for repigmentation include prostaglandin analogues, rituximab, afamelanotide, and JAK inhibitors.
Table 33.2
Pipeline therapies for vitiligo
Stabilization | Repigmentation |
|---|---|
Oral corticosteroids | Afamelanotide |
NB-UVB phototherapy | Prostaglandin F2α analogues |
Minocycline | Rituximab |
Methotrexate | Alternative phototherapy |
JAK inhibitors | |
Simvastatin |
33.2.2.1 Therapies for Stabilization
Minocycline
Minocycline is a broad-spectrum bacteriostatic antibiotic commonly prescribed for acne vulgaris. Studies document that it also has anti-inflammatory and immunomodulatory properties [69]. It inhibits cytokine production, free radical formation, and apoptosis. Recently, minocycline was compared to oral minipulse dexamethasone therapy for stabilization of active vitiligo [70]. Twenty-five patients were treated with minocycline 100 mg daily for 6 months, and 25 patients received dexamethasone 2.5 mg on 2 consecutive days weekly. Both drugs were significantly effective for stabilization of vitiligo. Side effects were minimal in each group. This study expands our therapeutic options for stabilization of vitiligo.
Methotrexate
Methotrexate is an antimetabolite and competitively inhibits dihydrofolate reductase. It inhibits DNA, RNA, thymidylates, and protein synthesis. In addition, it inhibits T-cell activation and intracellular adhesion molecule expression by T cells. Methotrexate also downregulates B cells [71]. In a recent study of 42 patients, methotrexate was compared to oral minipulse corticosteroid therapy for stabilization of vitiligo. Half were treated with methotrexate 10 mg weekly and half with dexamethasone OMP 2.5 mg on two consecutive days weekly for 6 months. Both were equally effective in controlling vitiligo disease activity [72].
Narrowband UVB Phototherapy
Recent studies document the beneficial effects of NB-UVB phototherapy for stabilization of vitiligo. Forty-two patients were evaluated in a prospective comparative 3-month trial. Half were treated with NB-UVB twice weekly and the remainder with minocycline 100 mg daily. Stabilization occurred in 76 % of the NB-UVB group compared to 33 % of patients treated with minocycline. These findings suggest that NB-UVB phototherapy is indeed effective in stabilizing active vitiligo [73]. In addition, NB-UVB has been shown to be effective in reversing oxidative stress in patients with vitiligo. This may be essential for stabilization of the disease [74]. This investigation is one of the very first to document the stabilizing effects of NB-UVB in a comparative trial.
33.2.2.2 Therapies for Stabilization
Afamelanotide
Afamelanotide is a potent synthetic analogue of alpha-melanocyte-stimulating hormone. It stimulates melanogenesis and melanocyte proliferation. When compared to α-MSH, afamelanotide exerts prolonged physiologic effects with stronger binding affinity to the melanocortin receptor (MC1R). Several studies have demonstrated defects in the melanocortin system in vitiligo patients [75, 76]. These defects include low physiologic plasma α-melanocyte-stimulating hormone, decreased α-MSH in lesional skin areas, and decreased expression of prohormone convertase.
Clinically, afamelanotide combined with NB-UVB was found to be significantly superior to NB-UVB monotherapy in a randomized multicenter study involving 55 patients with non-segmental vitiligo [76, 77]. Patients were treated for 6 months and observed 6 months thereafter. Twenty-eight patients were treated with afamelanotide and 27 with NB-UVB alone. The time to onset of repigmentation with combination group was significantly lessened, especially on the face and upper extremities, and more evident in skin types IV–VI. Moreover, there was significantly greater repigmentation in the combination group versus NB-UVB monotherapy. Notable side effects from combination treatment versus monotherapy were nausea, fatigue, and hyperpigmentation. Overall, the drug was well tolerated. However, the optimum dosage, administration frequency, and further side effects need to be determined.
Prostaglandin Analogues
Prostaglandin E2 (PGE2) is a novel and potentially beneficial treatment for localized stable vitiligo. PGE2 controls the proliferation of melanocytes through stimulant and immunomodulatory effects. In a consecutive series, repigmentation occurred in 40 of 56 patients with stable vitiligo treated with translucent PGE2 0.25 mg [78]. The patients applied the PGE2 gel twice daily for 6 months. The mean onset of repigmentation was 2 months. The response was excellent in 22 of 40 patients, with complete repigmentation observed in eight patients. The patients with disease duration of 6 months showed the most significant response. Repigmentation occurred the earliest on the face and scalp.
Bimatoprost, a synthetic prostamide (prostaglandin-ethanolamine) F2α analogue, is associated with hyperpigmentation of periocular skin caused by increased melanogenesis [79]. In a study by Narang et al. with ten patients, three had 100 % repigmentation, three had 75–99 %, and one had 50–75 %. The best responses were observed on the face [80]. A recent study documented efficacy of bimatoprost in 18 patients with non-facial vitiligo [81].
Latanoprost, also a prostaglandin F2α analogue, has also been evaluated for repigmentation of vitiligo in 22 patients. It was superior to placebo and has comparable efficacy of NB-UVB in achieving repigmentation of vitiliginous lesions [82].
Alternative Phototherapy
Natural sunlight emits a spectrum of accessible UV radiation, including sufficient amounts of NB-UVB beneficial for vitiligo. However, it also emits nontherapeutic wavelengths increasing the erythrogenic response. A topical cream was subsequently developed which filtered nontherapeutic radiation for solar exposure with a bias for 311 nm, allowing NB-UVB to pass through for treatment [83].
In a double-blind placebo-controlled study, 15 vitiligo patients exposed themselves to natural sunlight thrice weekly following the use of the selectively permeable topical cream allowing exposure to NB-UVB [84]. This was compared against a placebo sunscreen with SPF4. Only acrofacial vitiligo was treated. UV exposure was determined by an MED chart and measurement of solar ultraviolet light using a UV radiometer. Mean repigmentation at 12 weeks was 49 % for the topical cream and 3 % for the placebo group. Additional studies are warranted.
Rituximab
Rituximab is a chimeric monoclonal antibody against the protein CD20, primarily expressed on the surface of B cells. B cells are responsible for antibody formation. Rituximab destroys B cells and is used to treat diseases characterized by increased numbers of B cells or B-cell dysfunction. Anti-melanocyte antibodies have been shown to mediate the destruction of melanocytes in vitiligo [67, 85]. A 6-month pilot study was conducted in five vitiligo patients with active disease. They received intravenous rituximab 1 g in a single dose and were followed for 6 months. After 6 months, three of five patients showed marked repigmentation. One had slight improvement and the remaining patients showed no change. This repigmentation was correlated with biopsy findings of an increased number of melanocytes, decreased lymphoid infiltration, and decreased melanocyte apoptotic markers (Bcl-2, Apaf-1, caspase-9). As expected, circulating CD20 lymphocytes were decreased; however, melanocyte-specific antibodies and immunoglobulin levels showed no difference [86].
JAK Inhibitors
Tofacitinib is a JAK 1/3 inhibitor approved to treat severe rheumatoid arthritis. Tofacitinib inhibits the production of IL-17 and proliferation of CD4 cells. In addition, it inhibits ɣ-interferon signal transduction [87]. Current studies suggest that interferon ɣ-induced expression of the chemokine CXCL 10 mediates depigmentation in vitiligo [88, 89].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree