36 Urethral Diverticula
ETIOLOGY
The female urethra is a short tubular structure that is surrounded by multiple periurethral ducts and glands, the largest being the Skene’s glands; these are adjacent to the distal urethra and drain into the meatus. Congenital anomalies of the female urethra are quite rare. Infrequently, obstructing urethral valves have been identified; more often an ectopic ureter is identified within the urethra, which may masquerade as a urethral diverticular communication site. Boyd and Raz (1993) reported a patient with an ectopic ureter that drained into a urethral diverticulum.
Female urethral diverticula are diagnosed most frequently in the third to fifth decades. Most diverticula are acquired, and a favored hypothesis regarding the etiology of female urethral diverticula begins in the paraurethral glands. Most diverticula of the female urethra are located dorsally or laterally and distally. Physicians believe that repeated infections and subsequent destruction of the paraurethral glands lead to abscess formation within the periurethral and urethral glands. These obstructed glands then rupture into the urethral lumen and remain as outpouchings off the urethra, which eventually epithelialize, becoming a true urethral diverticulum as opposed to a urethrocele or pseudodiverticulum.
The location, number, and extent of urethral diverticula have an impact on the choice of treatment. A classification system for female urethral diverticula—location, number, size, configuration, communication, and continence (LNSC3)—has been described by Leach et al. (1993). Providing an accurate description of the diverticula under evaluation will, in turn, facilitate therapy.
PRESENTATION
Women present to their physicians with a host of symptoms, and, unfortunately, the patient’s description of each complaint is not always textbook clear. Therefore, the task is left up to the physician to identify, evaluate, and treat the pathology. A history of recurrent urinary tract infections, stress urinary incontinence, and incomplete voiding are some of the most common presenting symptoms in women with urethral diverticula (Table 36-1). According to Hoffman and Adams (1965), the single most important complaint is postmicturition-dribbling. The addition of dysuria and dyspareunia complete the classic triad “3 D’s.” These are all nonspecific complaints, however. If the symptoms are also accompanied by urgency, urge incontinence, frequency, and/or even a protruding vaginal mass, they are more highly suggestive of a urethral diverticulum. If pus can be expressed from the meatus with manual compression of the anterior vaginal wall, this strongly indicates the presence of a urethral diverticulum. Romanzi et al. (2000) reviewed their experience with diverse presentations of urethral diverticula and decided that when symptoms mimic other disorders, and especially when they do not improve and respond with standard therapy, entertaining the possibility that the source of the pathology is a urethral diverticulum is important.
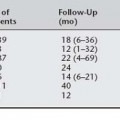
Table 36-1 Most Common Initial Complaints in Women Who Present for Evaluation and Are Ultimately Found to Have Urethral Diverticula From 1964 to 2000
| Mean (%) | Range (%) | |
|---|---|---|
| Recurrent urinary tract infections | 47 | 9–83 |
| Stress urinary incontinence | 46 | 28–100 |
| Incomplete voiding | 33 | 28–38 |
| Dysuria | 29 | 4–58 |
| Urgency | 28 | 18–47 |
| Urge incontinence | 27 | 11–35 |
| Frequency | 26 | 16–38 |
| Postvoid dribbling | 21 | 4–65 |
| Lower abdominal pain | 20 | 1–50 |
| Pus per urethra | 18 | 3–50 |
| Protruding vaginal mass | 18 | 7–27 |
| Dyspareunia | 13 | 1–24 |
| Hematuria | 10 | 5–18 |
| Urine retention | 10 | 3–21 |
| Difficulty voiding | 8 | 2–14 |
Many patients receive various treatments that include antibiotics, anticholinergic and antidepressant medications, bladder hydrodistention, and urethral dilations for suspected pelvic disorders. Some of the more common presumed diagnoses are listed in Table 36-2. In summary, in any case of persistent lower urinary tract symptoms unresponsive to therapy, one should exclude a urethral diverticulum.
Table 36-2 Initial Diagnoses First Given to Patients and Their Subsequent Treatments Before the Diagnosis of Female Urethral Diverticulum
| Diagnosis | Treatments |
|---|---|
| Chronic cystitis, trigonitis, cystitis cystica | Antibiotics |
| Stress urinary incontinence | Anti-incontinence surgery |
| Urgency, frequency, urge incontinence (overactive bladder, detrusor overactivity) | Anticholinergic therapy |
| Interstitial cystitis, idiopathic pain syndrome | |
| Urethral syndrome | Urethral dilation |
| Vulvodynia | |
| Cystocele | Surgery |
| Psychosomatic disorder |
DMSO, Dimethylsulfoxide.
DIAGNOSIS
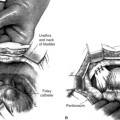
Not all patients will present with a suburethral mass, and not all suburethral masses are urethral diverticula. The differential diagnosis of periurethral or suburethral masses is extensive and include urethral diverticulum, urethrocele, Skene’s gland abscess, Gartner’s duct cyst, ectopic ureterocele, vaginal wall inclusion cyst, and other less frequent diagnoses (Box 36-1). The urethra may be tender, and, on occasion, a large diverticulum is evident as an anterior wall mass that may express pus and debris from the urethral meatus when compressed. Suspicions of a urethral carcinoma or calculi arise if a firm mass is palpated along the vaginal wall.
BOX 36-1 DIFFERENTIAL DIAGNOSIS OF SUBURETHRAL MASS
Modified with permission from Blaivas JG, Flisser AJ, Bleustein CB, Panagopolous G. Periurethral masses: etiology and diagnosis in a large series of woman. Am J Obstet Gynecol 2004;103:842.
One must clinically suspect a urethral diverticulum to select the most appropriate procedures and imaging studies. Many patients with urethral diverticula undergo urodynamic testing to evaluate their complaints of voiding dysfunction. Urodynamics provide information on bladder function, during both the storage and voiding phases. Certainly, not all patients will require urodynamic testing; however, it should be used in patients who have had previous pelvic surgery, recurrent stress urinary incontinence after bladder surgery, and urinary retention without any other known reason.