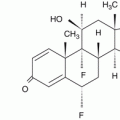
Fig. 32.1
The number of ICSRs due to steroid substances
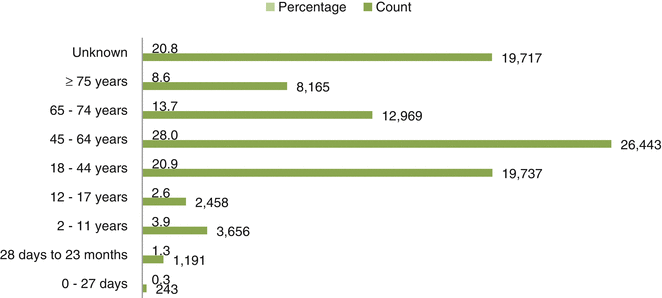
Fig. 32.2
Age-wise distribution of ICSRs due to steroids
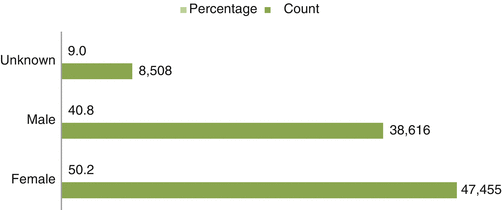
Fig. 32.3
Gender-wise distribution of ICSRs
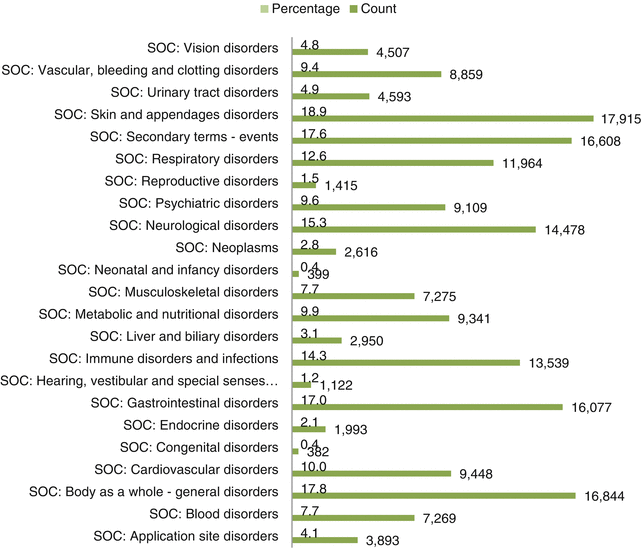
Fig. 32.4
System organ class-wise distribution of ICSRs
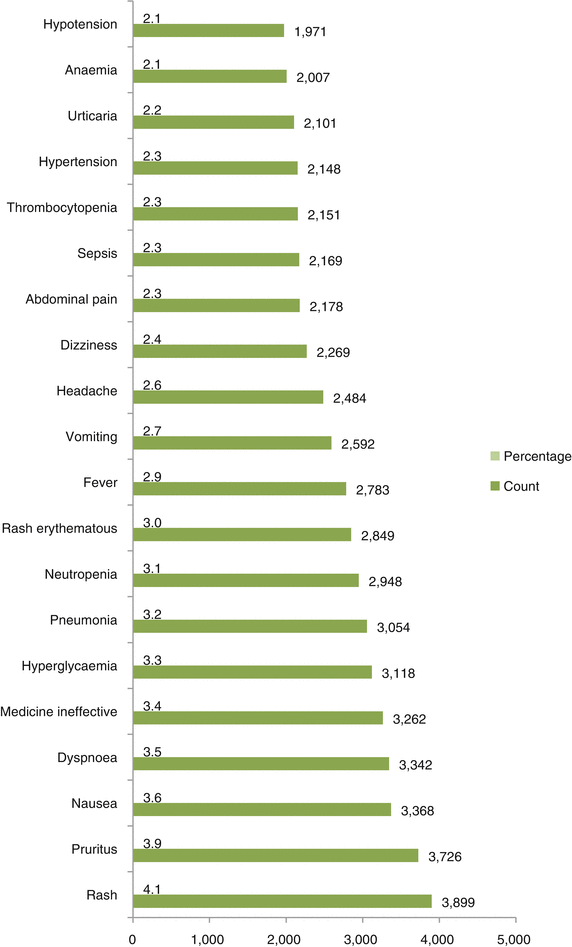
Fig. 32.5
Top 20 preferred terms (adverse drug reaction term) used during reporting of ICSRs due to steroids substances

Fig. 32.6
The year-wise number of ICSRs received at UMC-Sweden due to steroids substances
However, UMC states that (1) regarding the source of the information (2) that the information comes from a variety of sources, and the likelihood that the suspected adverse reaction is drug-related is not the same in all cases, (3) that the information does not represent the opinion of the World Health Organization [11].
Conclusion
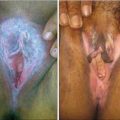
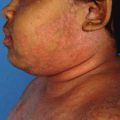
The present study revealed that topical steroids are associated with various serious and non-serious adverse events including rash, pruritis, atrophy, telangiectasia, periorificial dermatitis, etc. However, the present study could not assess the causal relationship because the drug may be abruptly stopped or the use of topical steroids may be irrational. Therefore, the author concludes that the steroids need to be prescribed rationally. If such adverse events occurred or came to the notice of healthcare professionals, they are encouraged to report known and unknown adverse events due to steroids to their respective countries pharmacovigilance system.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree