Anatomic implants were introduced in the late 1980s, gaining popularity through the 1990s and beyond. The moratorium on silicone meant that the use of theses devices was less diffuse in the US and has remained so. Fifth-generation devices had improved form stability with more aggressive texturization to enhance soft tissue interaction. The ability to vary height, width and projection independently creates much versatility in their use for complex anatomical situations producing natural results. The impact of BIA-ALCL has had a significant impact on their use.
Key points
- •
Manufacturing quality, standards, and design of implants continue to improve over time.
- •
The greatest impact on implant selection and indications has been the increasing awareness of breast implant-associated anaplastic large cell lymphoma and its association with textured surfaces.
- •
For anatomic devices, the form stability of the gel is arguably more important than for round devices.
Introduction
The evolution and development of breast implants has continued to grow since their inception in the early 1960s. Manufacturing quality, standards, and design all continue to improve over time. This development history is often referred to as the implant generations ( Table 1 ). It describes the technological advances and improvements over this period. Over time, our understanding of the interaction between these devices and the body has also continued to develop. Hence, our knowledge of phenomena such as capsular contracture, the importance of the biofilm, tissue integration, movement within the breast, form stability, and, importantly, implications of breast implant-associated anaplastic large cell lymphoma (BIA-ALCL), all continue to evolve with emerging data and research.
| Implant Generations | ||
|---|---|---|
| First generation | 1960s | Thick shell Viscous gel Dacron patch |
| Second generation | 1970s–1980s | Thin shell No patch |
| Third generation | 1980s–1992 | Thicker shells 1st barrier coats |
| Fourth generation | 1992–present | Better manufacturing barrier coat/texture |
| Fifth generation | 1993–present | Cohesive gel/shape form stable implants |
| Sixth generation? | Present | BLite air filled microspheres bound to silicone |
Although the implant started as a simple structure—a liquid silicone-filled sphere with an outer silicone shell—over time it has developed into a much more sophisticated device. The gel content has changed significantly transitioning from non–form-stable liquidity to much more solid form-stable silicone gel. Differential cross-linking of silicone polymers has allowed for endless permutations of these differing gel properties. The latest technological advances have seen the development of the B-lite implant, which incorporates air-filled borosilicate microspheres bound to silicone polymers to render the implants 30% lighter than standard silicone. , In addition, the shell structure and the surface have changed dramatically over the years. Improved barrier layers to minimize gel bleed, and differing surface texturization from smooth, to nano surfaces, different depth texturization and the continued use of polyurethane in many parts of the world. Sophisticated implant Matrices have been developed by all the manufacturers especially for their anatomic devices, allowing for a virtually custom-made approach for individual patients.
Impact of breast implant-associated anaplastic large cell lymphoma on the use of anatomic devices
The greatest impact in recent times on implant selection and indications has undoubtedly been the increasing awareness of BIA-ALCL and its association with textured surfaces. All anatomic implants have textured surfaces to varying degrees. Therefore, in simple terms, the elimination of texture would mean the elimination of anatomic devices. In Europe, culturally and historically virtually all devices have been textured, including round ones. Until now, there has been virtually no experience with the use of round smooth devices on the European continent, a practice that is the polar opposite to that in the majority of the United States.
As a result, for the first time in Europe there has been a shift in implant use from anatomic to round smooth or less textured devices. The question is, does this change matter? The answer depends on 2 issues: First, the belief that anatomic implants confer sufficient advantage over round implants in certain situations, and secondly that the risk of ALCL from these devices is acceptably low to justify their continued use.
To address the second point first, the true risk of BIA-ALCL is unclear, and as with many contentious subjects it depends on literature selection and interpretation, as well as subjective experience and perspectives. An in-depth analysis of ALCL risk is beyond the scope of this article, but the subject cannot be ignored completely when considering the selection of anatomic implants. The surface with the greatest association with BIA-ALCL was that of the Allergan BIOCELL textured surface. , The reasons for this association are poorly understood, and despite hypotheses around the role of biofilm in the causation of the disease, this link is not proven as yet. , In 2019 in recognition of this risk association, Allergan voluntarily withdrew and stopped production of the BIocell surface on the advice of the US Food and Drug Administration. According to figures from the US Food and Drug Administration, 84% of all cases of ALCL are derived from BIocell. The remaining 16% are accounted for by the remaining textured contingent. In addition, another culprit often quoted is polyurethane. The 2 main manufacturers for this material were Silimed and Polytech. Silimed is now defunct and accounted for more than 90% of all polyurethane-related ALCL cases. In summary, although BIA-ALCL risk pertains to any device, there is a strong argument to suggest that today the risk is significantly lower when grouping the remaining textured devices together, now that Allergan Biocell and Silimed polyurethane are no longer part of the statistics. This issue is clearly important and has to be carefully discussed with patients when considering the pros and cons of implant selection and all the associated risks.
Perception of risk is something that affects individuals differently, both clinicians and patients. The work of Sieber and Adams, looking at the micromort analysis of BIA-ALCL as compared with many mundane daily events such as driving a car, drinking a glass of wine, living for 2 days in New York, and many others, was designed to put into perspective the actual risk of ALCL as compared with such other activities. At the point of writing this article, it seems that our true understanding of ALCL and its significance is still some way off.
Differences between round and anatomic implants
There are many differences between round and anatomic devices. Clearly, they are shaped differently, but it is important to understand the significance of this shape difference; this factor is something that will be expanded on in this article. In addition, there are other differences worthy of mention: surface, gel, dimensionality, and projection point and volume distribution
Surface
As mentioned elsewhere in this article, for anatomic implants, by definition, a degree of texturization is required to minimize rotation risk. For round implants, although the same texturization is also available, it is perhaps less necessary than for the shaped devices, because rotation is not a risk, and therefore the option of a smooth surface is available for users of round implants who seek to avoid texture.
Gel
Gel consistency may vary for the 2 devices. For anatomic devices, the form stability of the gel is arguably more important than for round devices. Form stability maintains the asymmetric shape of the anatomic device and allows it to impart its shape to the breast tissue; this is a key feature of the anatomic implant (see Projection Point and Volume Distribution). For a round device, the imposition of the shape of the device is less important and, therefore, the gel is often able to be softer. For some patients, this factor may also be an advantage.
Dimensionality
A major advantage of anatomic devices is that they can be varied in 3 dimensions as opposed to 2 dimensions, as in round implants. For the latter, because the height and the width are always the same, the only other independent variable is the projection of the implant. For anatomic implants, the height can be varied independent of the width, with the projection also being an independent variable. This combination gives great versatility in being able to select an implant more precisely for certain anatomic situations and for complex asymmetries ( Fig. 1 ).
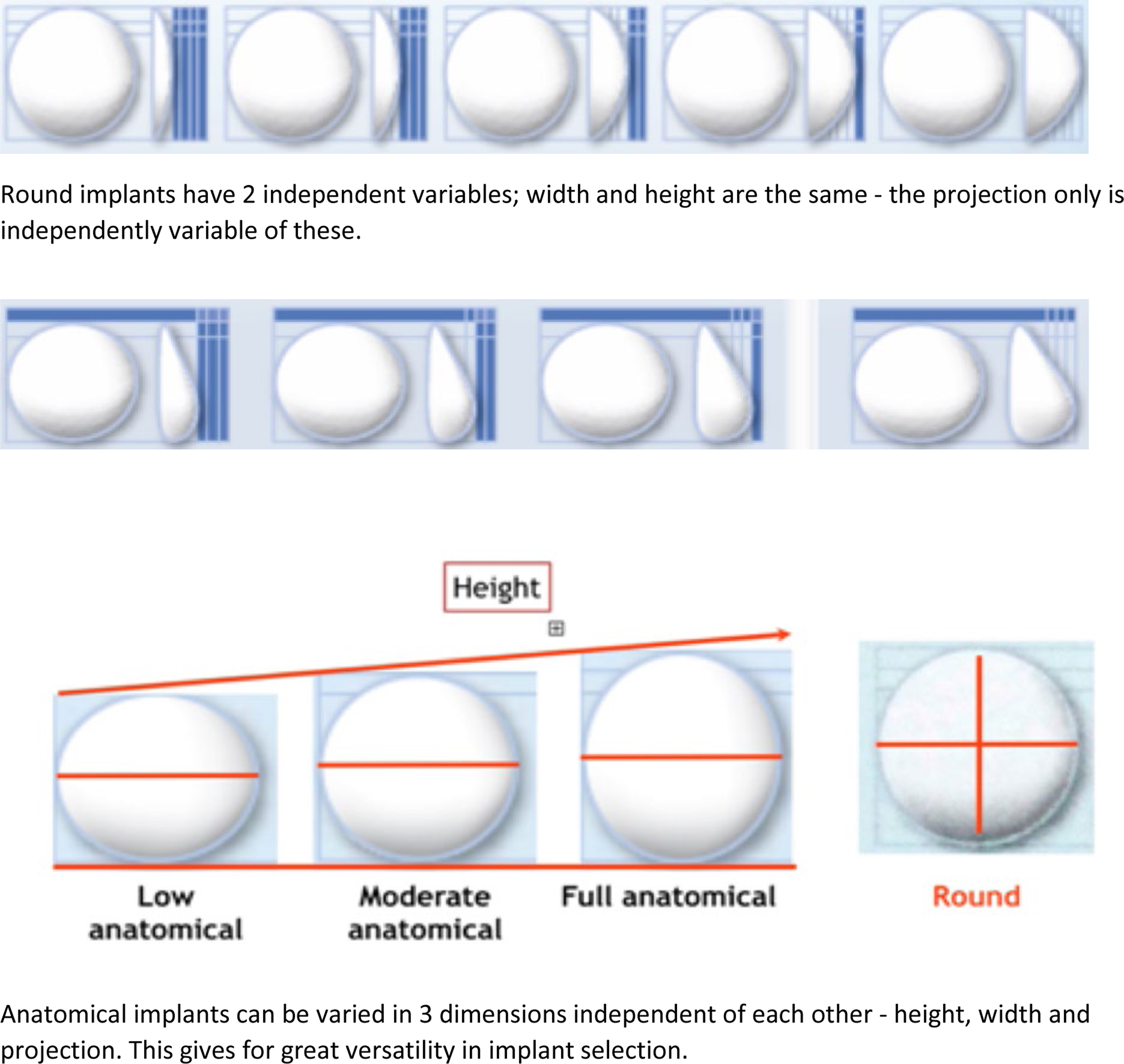
The Y number is a well-established formula for calculating implant height selection according to the morphologic features of the chest wall to optimize selection for any particular individual.
Projection Point and Volume Distribution
The difference in volume distribution between the 2 devices results in a significant difference in the position of the point of maximum projection. This element is perhaps the most important in being able to select a device that is able to achieve optimal volume distribution above and below the nipple–areola complex (NAC) especially in difficult anatomic situations. Anatomic implants have low projection points—short height devices have even lower projection points than full height devices. Round implants have relatively higher projection points ( Fig. 2 ).
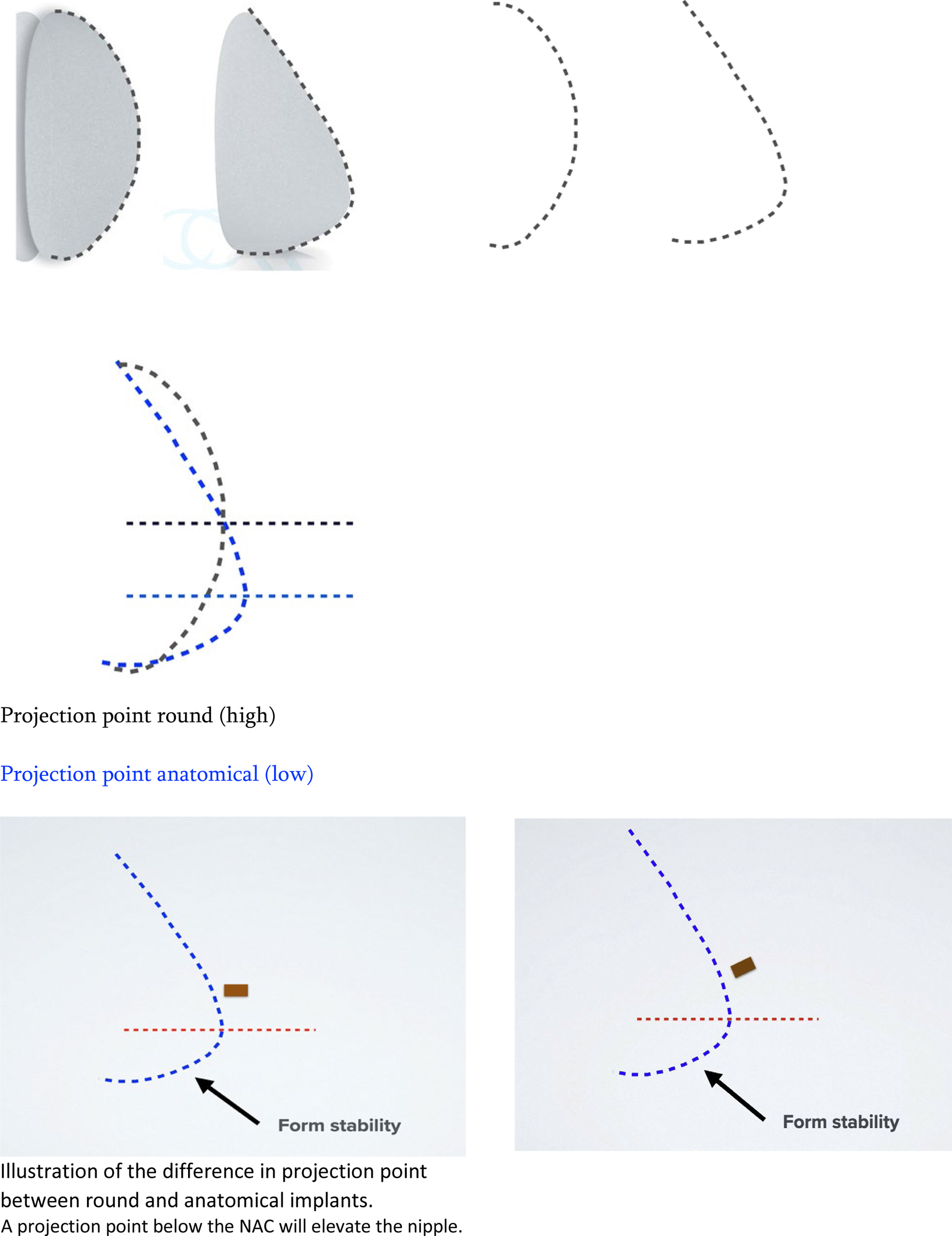
The significance of the projection point is that, ideally, the maximum point of projection should be just below the NAC. This configuration allows a forward and upward projection of the NAC on the breast mound as well as a harmonious volume distribution in keeping with the authors’ 45:55 ratio. If the projection point lies above the NAC, the nipple will be pushed in a downward direction, which is highly undesirable ( Fig. 3 ). In addition, a high projection point with a downward pointing nipple often results in an inadequately filled lower pole and hence a waterfall deformity. The low projection point of the anatomic device as well as the volume distribution means that adequate filling of the lower pole can be achieved with a harmonious volume distribution. , The understanding of this last point is perhaps the most critical element in understanding the difference between round and anatomic devices and the beneficial effects they can have on breast shape.
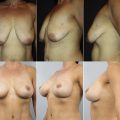
Why are we told there is no difference between the devices?
Various publications in the literature suggest that, as plastic surgeons, we are not able to discern a round implant form an anatomic implant when looking at postoperative images of women with implants. The implication of this assertion is that if there is no difference between the 2 devices, why use an anatomic implant at all? The major flaw of these studies is a lack of control imagery. In other words, in a postoperative image where the result seems as if it might be round, but in fact it is anatomic, the image might have seemed to be even more round with a round implant; however, this comparison is never presented. The fact that an anatomic implant can look round and vice versa is simply a factor of starting anatomy and tissue-based implant selection. All anatomic implants look round if they are too large, and almost all round implants look natural when they are small enough and the starting anatomy is favorable. This similarity in appearance absolutely does not mean that there is no difference between the devices. It is important to understand, based on the volume distribution and projection point discussed earlier, when and how to use anatomic implants.
Indications for anatomic implants
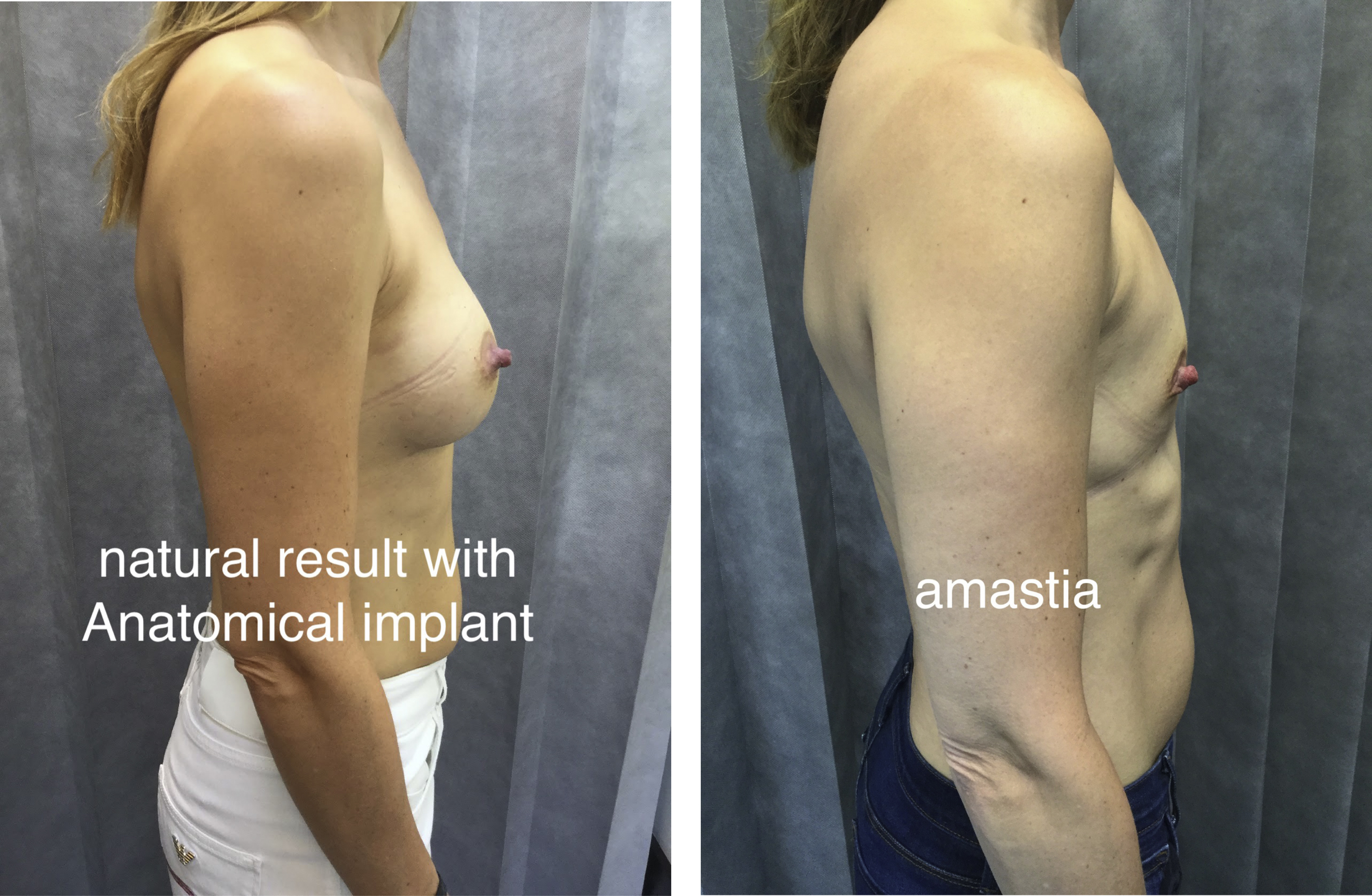
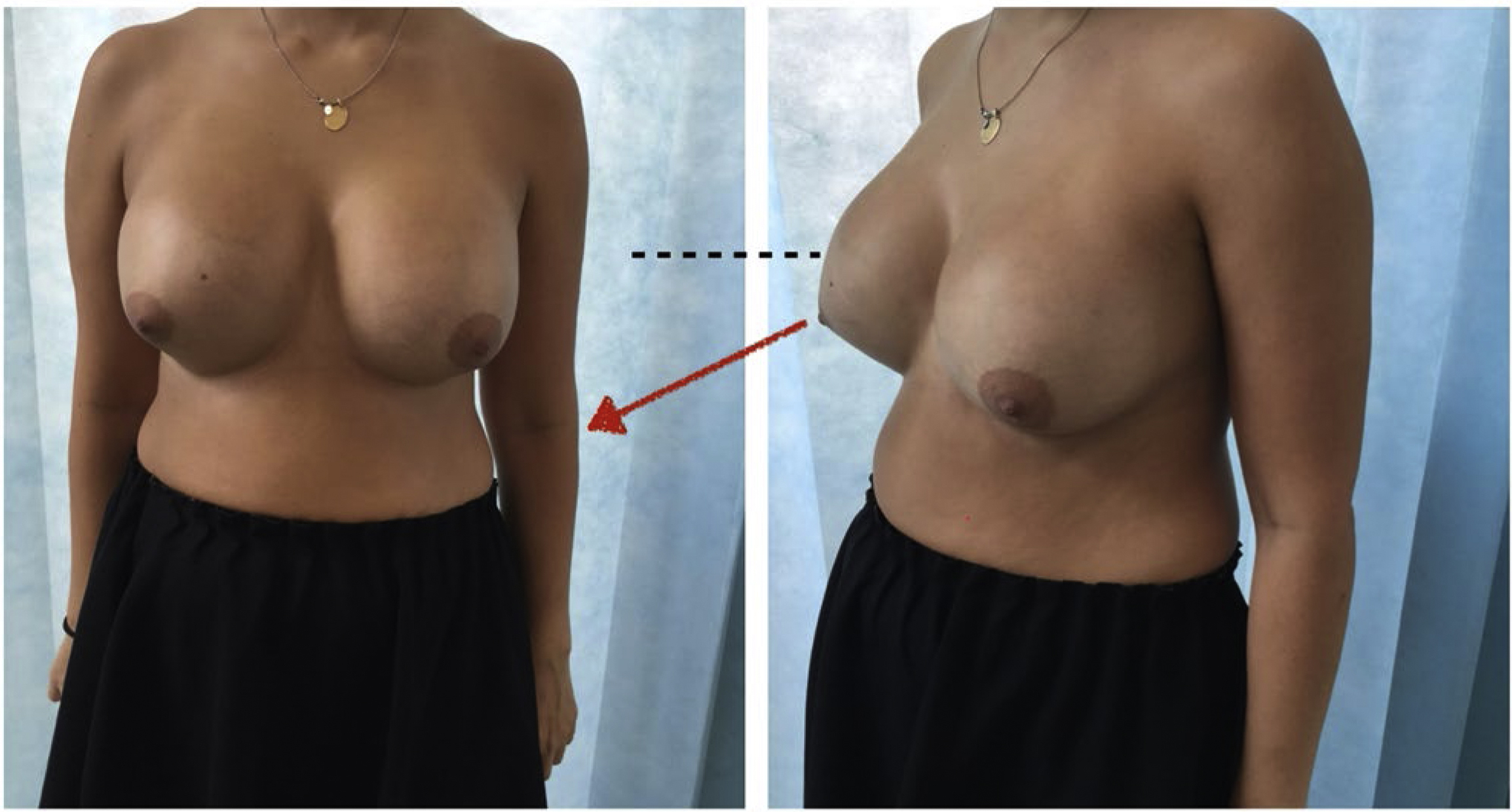
Natural Look
Anatomic implants are classically suited to those seeking a natural look. The volume distribution of the device lends it self to recreation of the 45:55 volume distribution previously defined by the author as being indicative of ideal natural breast beauty. The ICE principle was devised to plan for anatomic implant placement to replicate this distribution predictably. This is especially the case in those who have amastia or very little breast development ( Fig. 4 ).