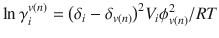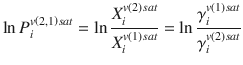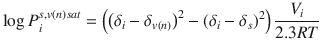,
where R is the ideal gas constant, T and P are temperature and pressure, x i is the mole fraction of component i, N j are the numbers of molecules of each component j, 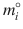 is the chemical potential of pure component i at a reference state (
is the chemical potential of pure component i at a reference state (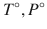 ), and γ i is the activity coefficient. Rearranging the equation leads to an expression of the change in the partial Gibbs energy of mixing (ΔG i ) as a function of concentration:
), and γ i is the activity coefficient. Rearranging the equation leads to an expression of the change in the partial Gibbs energy of mixing (ΔG i ) as a function of concentration:
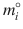 is the chemical potential of pure component i at a reference state (
is the chemical potential of pure component i at a reference state (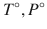 ), and γ i is the activity coefficient. Rearranging the equation leads to an expression of the change in the partial Gibbs energy of mixing (ΔG i ) as a function of concentration:
), and γ i is the activity coefficient. Rearranging the equation leads to an expression of the change in the partial Gibbs energy of mixing (ΔG i ) as a function of concentration: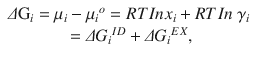
where ΔG i ID and ΔG i EX are the ideal and excess Gibbs mixing energies for component i. The activity coefficient of component i can then be taken as:
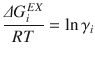 Chemical potential is the driving force behind solubility, phase equilibrium, and diffusion. The activity coefficient captures the departure from ideality for real mixtures and provides a mechanism to link that departure to the mixture composition. Activity coefficient models, which incorporate parameters that describe the interactions between mixture components, provide a way to include mixture composition effects in chemical property predictions.
Chemical potential is the driving force behind solubility, phase equilibrium, and diffusion. The activity coefficient captures the departure from ideality for real mixtures and provides a mechanism to link that departure to the mixture composition. Activity coefficient models, which incorporate parameters that describe the interactions between mixture components, provide a way to include mixture composition effects in chemical property predictions.
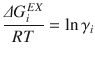
6.2.1 Solubility
Predicting the solubility of a compound in various vehicles is important both in manufacturing processes and in the formulation of drug products. The solubility of a compound in a solvent is the equilibrium expressed as:
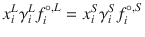 If the solid is pure, then this reduced to
If the solid is pure, then this reduced to
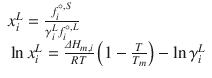 where ΔH m,i is the enthalpy of fusion, T m is the melting point, x i L is the solubility, γ i L is the liquid activity at saturation, and
where ΔH m,i is the enthalpy of fusion, T m is the melting point, x i L is the solubility, γ i L is the liquid activity at saturation, and 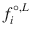 and
and 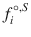 are the fugacity of the pure liquid and solid phases. So, prediction of solubility can be based on accessible physical properties, and a model for liquid phase activity, of the solute.
are the fugacity of the pure liquid and solid phases. So, prediction of solubility can be based on accessible physical properties, and a model for liquid phase activity, of the solute.
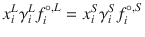
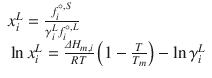
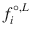 and
and 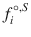 are the fugacity of the pure liquid and solid phases. So, prediction of solubility can be based on accessible physical properties, and a model for liquid phase activity, of the solute.
are the fugacity of the pure liquid and solid phases. So, prediction of solubility can be based on accessible physical properties, and a model for liquid phase activity, of the solute.Many activity coefficient models have been developed and applied to solubility in drug-like compounds. These range from simple models to complex computational chemistry-based simulations. Many are incremental improvements on already existing models such as the Non-Random Two-Liquid (NRTL) and UNIversal QUAsiChemical (UNIQUAC).
Mirmehrabi proposed a simple activity coefficient model, similar to the Margules model but extended to mixtures, and used it to predict vapor-liquid equilibria in 41 systems and solubility of stearic acid in a solvent mixture with better results than UNIFAQ (UNIQUAC Functional Activity Coefficients) (Mirmehrabi et al. 2006a). The same proposed model was tested with three drugs in pure and mixed solvents showing that while it performed well for binary systems, UNIQUAC and NRTL outperformed in mixture systems (Mirmehrabi et al. 2006b). A simple one-parameter Wilson model has been used to predict the solubility of four drugs in water plus cosolvents of either ethanol, methanol, or 1,4-dioxane (Matsuda et al. 2010). Computations of the infinite dilution activity coefficient of salicylic acid in water and ethanol, performed with the one-parameter Wilson model of Matsuda et al. (2010), highlight the large change in activity due to variation of the mixture composition from pure water to pure ethanol (Fig. 6.1).
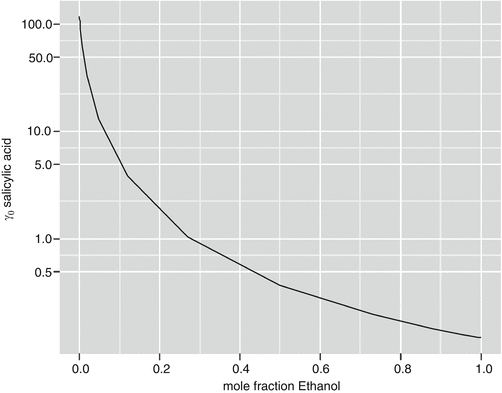

Fig. 6.1
Activity coefficient of salicylic acid in water and ethanol mixture, by one parameter (Wilson model with parameters from Matsuda et al. 2010)
The basic NRTL model was successfully applied to correlate simvastatin solubility in 15 solvents, with the goal of predicting solubility in mixtures (Nti-Gyabaah et al. 2009). The polymer NRTL model of Chen (1993) was extended by Chen and Song (2004) by introducing the concept of a segment activity coefficient (SAC) to create the NRTL-SAC model. The NRTL-SAC model decomposes molecules into recognized segments and adjusts the activity prediction based on the segments’ hydrophilicity, hydrophobicity, and polarity. The NRTL-SAC model has been used to model 14 drug compounds’ solubility in up to 14 solvents (Chen and Song 2004). Application of the NRTL-SAC model to vehicle mixtures showed good agreement with experimental data (Chen and Crafts 2006), even in associating systems such as ethanol/water, and captured the solubility peak at minimum activity.
An adapted version of the UNIFAC model, A-UNIFAC, was developed by Mengarelli to extend its capabilities to associating systems (Mengarelli et al. 1999). The A-UNIFAC model was extended and compared with NRTL-SAC in predicting solubility for seven drug-like compounds in up to 28 pure solvents and was found to be adequate, though the NRTL-SAC model gave generally better results (Mota et al. 2012). A comparison of UNIFAC, Conductor-like Screening MOdel for Realistic Solvents (COSMO-RS), NRTL-SAC, and a pharma-modified UNIFAC was made, using up to 17 drugs in 15 pure solvents and a few mixtures, in which NRTL-SAC and the modified UNIFAC fared best (Diedrichs and Gmehling 2010). Tung compared a SAC version of COSMO and NRTL-SAC for four drugs in 13 solvents and found that while NRTL-SAC predicted better overall, including in solvent mixtures, COSMO-SAC had the advantage of being an ab initio method (Tung et al. 2008).
Gross and Sadowski developed the perturbed chain statistical associating fluid theory (PC-SAFT) (Gross and Sadowski 2001) as an equation of state for modeling vapor-liquid and liquid-liquid systems. It was extended to associating systems such as ethanol/water (Gross and Sadowski 2002), and has been applied to the prediction of solubility of six drugs in up to 17 pure solvents and several mixtures (Spyriouni et al. 2011).
6.2.2 Partition Coefficient
The partition coefficient is the ratio of concentrations of a solute in two different liquid phases. At a minimum, this represents a ternary system in equilibrium. The equilibrium between two liquid phases α and β at a constant temperature (T) and pressure (P) is governed by:
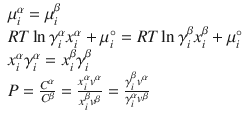 where C α and C β are the concentration of solute in the two phases, P is the partition coefficient, and v α and v β are the molar densities of the two phases. The equilibrium concentrations are determined by the activity coefficients (or vice versa to compute activity from observed data). The NRTL and UNIQUAC? models have been used to correlate partition coefficient data for atenolol and propranolol in a water/n-octanol system, with both showing the ability to predict P as a function of temperature (Mohsen-Nia et al. 2012). The SPARC performs automated reasoning in chemistry (SPARC) system has been used to predict solubility (N = 707, RMS = 0.487), activity coefficient (N = 2647, RMS = .272), and distribution coefficient (N = 698, RMS = 0.44) (Hilal et al. 2004), where N is the number of compounds in the dataset and RMS is the root mean square error in the prediction in log units. Ingram used COSMO-RS in the prediction of partition and distribution coefficients of 19 ionizable drugs over a range of pH and with various counterions (Ingram et al. 2011).
where C α and C β are the concentration of solute in the two phases, P is the partition coefficient, and v α and v β are the molar densities of the two phases. The equilibrium concentrations are determined by the activity coefficients (or vice versa to compute activity from observed data). The NRTL and UNIQUAC? models have been used to correlate partition coefficient data for atenolol and propranolol in a water/n-octanol system, with both showing the ability to predict P as a function of temperature (Mohsen-Nia et al. 2012). The SPARC performs automated reasoning in chemistry (SPARC) system has been used to predict solubility (N = 707, RMS = 0.487), activity coefficient (N = 2647, RMS = .272), and distribution coefficient (N = 698, RMS = 0.44) (Hilal et al. 2004), where N is the number of compounds in the dataset and RMS is the root mean square error in the prediction in log units. Ingram used COSMO-RS in the prediction of partition and distribution coefficients of 19 ionizable drugs over a range of pH and with various counterions (Ingram et al. 2011).
6.2.3 Diffusion
Chemical potential plays a role in diffusion as well. The generalized Maxwell-Stefan equation relates the diffusional flux of components of a mixture to the driving force for diffusion (Taylor and Kooijman 1991):
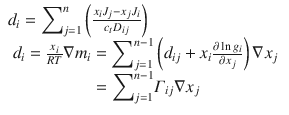 In a binary system (or perhaps one with only one diffusing solute), the equation simplifies to:
In a binary system (or perhaps one with only one diffusing solute), the equation simplifies to:
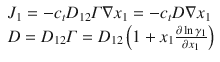 The diffusion coefficient usually estimated from experimental data is the Fickian diffusion coefficient D, which is the product of the Maxwell diffusion coefficient D 12 and the thermodynamic factor Γ. Note that the mixture molar density c t is taken to be constant here. The thermodynamic factor can be computed from any activity coefficient model.
The diffusion coefficient usually estimated from experimental data is the Fickian diffusion coefficient D, which is the product of the Maxwell diffusion coefficient D 12 and the thermodynamic factor Γ. Note that the mixture molar density c t is taken to be constant here. The thermodynamic factor can be computed from any activity coefficient model.
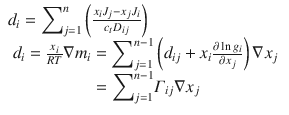
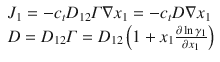
The activity-adjusted diffusion coefficient D 12 has been computed from observed Fickian diffusion coefficients D for several binary systems (Vignes 1966). The results showed that D could vary by an order of magnitude with the concentration of the solute and that the Maxwellian diffusivity D 12 as a function of concentration of solute was log-linear, being described by: 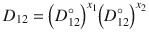 , where
, where 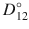 and
and 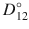 are the infinite dilution diffusivities. The implication is that with an activity coefficient model for a solvent/solvent system, and a prediction of dilute diffusivity, that diffusivity can be predicted over the entire range of concentrations.
are the infinite dilution diffusivities. The implication is that with an activity coefficient model for a solvent/solvent system, and a prediction of dilute diffusivity, that diffusivity can be predicted over the entire range of concentrations.
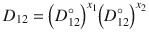 , where
, where 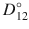 and
and 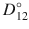 are the infinite dilution diffusivities. The implication is that with an activity coefficient model for a solvent/solvent system, and a prediction of dilute diffusivity, that diffusivity can be predicted over the entire range of concentrations.
are the infinite dilution diffusivities. The implication is that with an activity coefficient model for a solvent/solvent system, and a prediction of dilute diffusivity, that diffusivity can be predicted over the entire range of concentrations.6.3 Potential Mechanisms of Interaction
Thermodynamics provides a mechanism for understanding how components in a mixture can interact to modulate key properties related to transdermal penetration. How these properties interact, however, at different levels of the penetration process is the key driver of the rate and extent of absorption. As the penetrant moves from the dosing vehicle (if there is one), into solution at the skin surface, then into the stratum corneum and dermis, it encounters different environments at each level. The vehicle can then have synergistic effects by acting in multiple levels. In addition, mechanisms that change the composition of the solvent and/or absorption environment can have dynamic effects on the chemical properties related to penetration and absorption.
6.3.1 Skin Surface
Several mechanisms at the skin’s surface affect the dissolution, diffusion, and penetration of drug from the dosing vehicle into the stratum corneum. Transport of the penetrant from the dosing vehicle to the surface of the skin may be limited by the solubility of the penetrant in the dosing vehicle or the rate of diffusion across a boundary layer. If the penetrant is dosed as a suspension, then dissolution may also become a rate-limiting factor. In the case of a neutral solute (penetrant) in a pure solvent, the thermodynamics of the system are relatively simple, even if not ideal (in the sense of “ideal” thermodynamic behavior). For binary or higher mixtures of solvents, however, the combined effects on the solute properties such as solubility and diffusion coefficient are not necessarily simple combinations of the properties in the pure solvent systems, and as such may exhibit synergistic effects.
Consider a boundary layer of the dosing vehicle sandwiched between the skin surface and some bulk fluid or dosing apparatus (e.g., a drug eluting patch). The flux of penetrant through the boundary layer at steady state is determined by Fick’s law as: 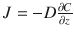 , where J is flux of penetrant, C is concentration of penetrant, D is the diffusion coefficient, z is the distance through the boundary layer. Assuming a constant gradient
, where J is flux of penetrant, C is concentration of penetrant, D is the diffusion coefficient, z is the distance through the boundary layer. Assuming a constant gradient 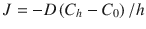 , where C h is the bulk concentration at the top of the layer and C 0 is the penetrant concentration at the skin surface (note, not in SC membrane itself). If the bulk concentration of penetrant is near saturation, then the mixture effects that increase solubility can increase flux as long as diffusion to the skin surface is the rate-limiting step.
, where C h is the bulk concentration at the top of the layer and C 0 is the penetrant concentration at the skin surface (note, not in SC membrane itself). If the bulk concentration of penetrant is near saturation, then the mixture effects that increase solubility can increase flux as long as diffusion to the skin surface is the rate-limiting step.
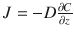 , where J is flux of penetrant, C is concentration of penetrant, D is the diffusion coefficient, z is the distance through the boundary layer. Assuming a constant gradient
, where J is flux of penetrant, C is concentration of penetrant, D is the diffusion coefficient, z is the distance through the boundary layer. Assuming a constant gradient 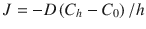 , where C h is the bulk concentration at the top of the layer and C 0 is the penetrant concentration at the skin surface (note, not in SC membrane itself). If the bulk concentration of penetrant is near saturation, then the mixture effects that increase solubility can increase flux as long as diffusion to the skin surface is the rate-limiting step.
, where C h is the bulk concentration at the top of the layer and C 0 is the penetrant concentration at the skin surface (note, not in SC membrane itself). If the bulk concentration of penetrant is near saturation, then the mixture effects that increase solubility can increase flux as long as diffusion to the skin surface is the rate-limiting step.One way to increase solubility is to add surfactants, which can aid in the solvation of hydrophobic molecules. Solvation imposes increased order around the solute molecule, however, and reduces rate of diffusion, as the solute and surrounding surfactant molecules have an increased tendency to move as a unit. In essence, the effective radius of the solute increases. This implies a trade-off between increased solubility increasing flux and reduced diffusivity reducing flux. When surfactant concentrations are above the critical micelle concentration (CMC), phase separation into micelles will occur, further complicating the system. Micelles diffuse much more slowly than the single solute molecule, but due to their size, can deliver multiple solute molecules instantaneously when they disassemble at the stratum corneum interface. Nonetheless, with the introduction of micelles, penetration is generally reduced (Wiechers 1989).
If volatile solvents are used, evaporation can cause an increase in solute concentration as the effective volume of solvent decreases (Stinchcomb et al. 1999). This disappearance of solvent volume can also reduce the depth of the boundary layer, which also increases flux. When a solvent mixture is used, the evaporation of one of the solvents results in a change in the solvent composition over time (except in the case of an azeotrope), which can drastically change the physical properties of solubility, diffusivity, and partition coefficient. As the solute concentrates due to solvent evaporation, it may begin to precipitate. If the solvent evaporates completely before the solute is fully absorbed into the stratum corneum, rapid intial flux will give way to slower flux and crystallization of penetrant on the skin surface, with the end result of incomplete absorption (Oliveira et al. 2012b). If, however, the solvent evaporates more slowly than the absorption rate, the effect will be to increase the overall absorption rate. One other possibility is that the solvent does not evaporate completely, which is more likely in a solvent mixture where one of the components is not volatile.
In the case of incomplete evaporation, the impact on absorption is more complex. Up to the point of precipitation, the absorption rate may be increased, presuming no other effects on solubility or diffusion coefficient. Supersaturation of lipophilic solutes in the donor well has been shown to increase their penetration by increasing thermodynamic activity while not affecting the stratum corneum barrier function (Moser et al. 2001). When the evaporation stops, C h will be at (or possibly above) the saturation concentration C sat and should remain at the saturation concentration, until all the precipitated drug redissolves. If redissolution is slow, the net effect may be an initial increase in flux followed by lowering of flux to the dissolution rate, which may be overall slower than if the system were occluded and evaporation could not occur. These same effects may occur if, instead of evaporation, loss of solvent is through its penetration into the skin.
6.3.2 Stratum Corneum
The transit and accumulation of a penetrant through the stratum corneum is governed primarily by partitioning and diffusion. The interaction of these phenomena and the potential for mixture components to influence one or both of them amplifies the impact of the mixture. The “brick and mortar” model of the stratum corneum (Michaels et al. 1975) suggests that the main pathway for diffusion is the intracellular lipid lamellae, which provide a hydrophobic barrier to water loss from the underlying tissue. The mechanisms by which vehicle constituents could affect the stratum corneum barrier function are extraction of lipids from the lamellae; alteration of the partition coefficient between the vehicle and stratum corneum lipids; disruption of the lipid bilayers; displacement of water domains; or loosening of the keratin structure in corneocytes (Daniels and Knie 2007).
The stratum corneum is composed of flattened corneocytes stacked in layers with an intracellular matrix of lipid lamellae, corneocyte envelops, corneocyte lipid envelops, enzymes, and structural proteins (Elias 2012). The barrier function of the skin is primarily due to the stratum corneum (Baroni et al. 2012; Bouwstra 1997), with the lipid lamellae contributing the majority of that function (Squier et al. 1991). The effectiveness of the barrier to water transit is modulated by the total lipid content of the stratum corneum (Grubauer et al. 1989; Squier et al. 1991). The only continuous pathway through the stratum corneum is via the lipid multilayer, and any molecules diffusing through the stratum corneum must traverse the lipid multilayer or move within it (Bouwstra 1997). In addition, the overlapping arrangement of the corneocytes in the “brick and mortar” model of the stratum corneum creates a tortuous path that effectively increases the length of the lipid pathway by about an order of magnitude (factor of 12.7 in human skin samples) (Michaels et al. 1975; Talreja et al. 2001).
The lipid lamellae are arranged in a multilayer, which is generally oriented parallel to the stratum corneum surface (Bouwstra 1997). A lipid monolayer is chemically bonded to the keratinized cell membranes of the corneocytes (Swartzendruber et al. 1987; Wertz et al. 1989) with various ceramides, free fatty acids, and cholesterol (Bouwstra 1997) comprising the lamellae between them. The lipids are organized primarily in crystalline phases, though some liquid phase may be present, and the presence of free fatty acids results in a denser packing structure (Bouwstra et al. 2003). The presence of the denser, orthorhombic packing structure, as opposed to the hexagonal or liquid-crystal phases, correlates with the effectiveness of the stratum corneum barrier (Damien and Boncheva 2009). It has been determined that the presence of endogenous proteins does not affect the lipid organization (Bouwstra et al. 2003).
As previously stated, the diffusional barrier is the intercellular lamellae of lipids. These are primarily neutral lipids and sphingolipids (largely ceramides) (Lampe et al. 1983) with some polar lipid and cholesterol sulfate. These lipids are formed into bilayers that span the gap between corneoctyes in several layers, with the lipid head groups oriented at the corneocyte membranes. The lamellar layers consist of a crystalline regime on both sides where the lipid head groups of adjacent layers meet, and a more disordered fluid phase between them with embedded head groups of the shorter chain lipids and cholesterol filling in. The lamellae are stacked multiple times over to fill in the intercellular pathway. The tight packing of the head groups in the crystalline regime and the orderliness of the fluid regime provide a strong barrier to diffusion of large and hydrophilic compounds. The lateral diffusion of lipophilic solutes in the stratum corneum lipids is dependent on the weight and size of the molecule (Johnson et al. 1996).
Disruptions to the packing structure in the crystalline regime or introduction of moieties that displace the small chain lipids and cholesterol in the fluid phase can weaken the diffusional barrier by disrupting the ordered structure of the layer and increasing fluidity. Some vehicle components may completely disrupt the lipid layers, resulting in their extraction. Many permeation-enhancing compounds, such as alcohols and alkanes, can extract lipids from the intercellular lipid domains of the stratum corneum (Walker and Smith 1996). At high concentrations, ethanol has been shown to extract lipids, while at lower concentrations it disrupts the lipid head groups resulting in disruption of the crystal structure and increased permeability (Krill et al. 1992; Suhonen et al. 1999). Fatty acids (Aungst 1989), various ceramides (Vávrová et al. 2003), a variety of other compounds including ethers and alcohols (Ibrahim and Li 2010), and numerous terpenes (Williams and Barry 1991) have been shown to increase permeation by fluidization of the lipid lamellae. Acidic pH may also contribute to fluidization (Kitagawa et al. 1995).
Modulation of the partition coefficient can occur by modification of the mixture properties on the skin surface or through changes induced in the stratum corneum lipid structure. The partition coefficient between the stratum corneum lipids and the vehicle at the skin surface is 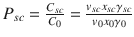 , which is a function of the mixture composition through the activity coefficients. So, vehicle components that raise γ sc or lower γ 0 of the penetrant increase the partitioning into the stratum corneum. A study of the effect of vehicle mixtures of SLS, water, ethanol, and propylene glycol demonstrated that partitioning into the stratum corneum was determined by the relative solubility of the solute in the vehicle and stratum corneum lipids (Van der Merwe and Riviere 2005b). Experiments in model membranes and skin support a model of vehicle interactions where high solvent uptake promotes drug partitioning by enabling the solute to exist within the solvent fraction/solvent-rich areas inside the membrane or skin by increasing solubility in the membrane (Oliveira et al. 2012a). Conversely, lowering the solubility in the vehicle has been found to also contribute to greater partitioning into the stratum corneum (Hilton et al. 1994).
, which is a function of the mixture composition through the activity coefficients. So, vehicle components that raise γ sc or lower γ 0 of the penetrant increase the partitioning into the stratum corneum. A study of the effect of vehicle mixtures of SLS, water, ethanol, and propylene glycol demonstrated that partitioning into the stratum corneum was determined by the relative solubility of the solute in the vehicle and stratum corneum lipids (Van der Merwe and Riviere 2005b). Experiments in model membranes and skin support a model of vehicle interactions where high solvent uptake promotes drug partitioning by enabling the solute to exist within the solvent fraction/solvent-rich areas inside the membrane or skin by increasing solubility in the membrane (Oliveira et al. 2012a). Conversely, lowering the solubility in the vehicle has been found to also contribute to greater partitioning into the stratum corneum (Hilton et al. 1994).
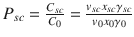 , which is a function of the mixture composition through the activity coefficients. So, vehicle components that raise γ sc or lower γ 0 of the penetrant increase the partitioning into the stratum corneum. A study of the effect of vehicle mixtures of SLS, water, ethanol, and propylene glycol demonstrated that partitioning into the stratum corneum was determined by the relative solubility of the solute in the vehicle and stratum corneum lipids (Van der Merwe and Riviere 2005b). Experiments in model membranes and skin support a model of vehicle interactions where high solvent uptake promotes drug partitioning by enabling the solute to exist within the solvent fraction/solvent-rich areas inside the membrane or skin by increasing solubility in the membrane (Oliveira et al. 2012a). Conversely, lowering the solubility in the vehicle has been found to also contribute to greater partitioning into the stratum corneum (Hilton et al. 1994).
, which is a function of the mixture composition through the activity coefficients. So, vehicle components that raise γ sc or lower γ 0 of the penetrant increase the partitioning into the stratum corneum. A study of the effect of vehicle mixtures of SLS, water, ethanol, and propylene glycol demonstrated that partitioning into the stratum corneum was determined by the relative solubility of the solute in the vehicle and stratum corneum lipids (Van der Merwe and Riviere 2005b). Experiments in model membranes and skin support a model of vehicle interactions where high solvent uptake promotes drug partitioning by enabling the solute to exist within the solvent fraction/solvent-rich areas inside the membrane or skin by increasing solubility in the membrane (Oliveira et al. 2012a). Conversely, lowering the solubility in the vehicle has been found to also contribute to greater partitioning into the stratum corneum (Hilton et al. 1994).Another potential interaction in the stratum corneum is binding of the drug to proteins in the keratinocytes (Frasch et al. 2011), which may cause a depot effect of the drug. That is, penetrant may move rapidly into the stratum corneum but accumulate there rather than enter the epidermis. For example, partition coefficient in delipidized stratum corneum increased for some compounds, possibly due to increased exposure to a protein domain that is normally shielded by the lipid domain (Surber et al. 1990). Depending upon the desorption rate, this could result in an extended time of absorption or even reduced absorption, if desorption is so slow that the penetrant is eliminated by desquamation. For penetrants with local action or toxicity, modulation of this accumulation may be crucial.
Hydration of the stratum corneum results in the inclusion of bulk water that may pool in corneocytes or within the intercellular lipid matrix (Pieper et al. 2003). Inclusion of water in the stratum corneum, as a function of relative humidity (RH), was found to sharply increase the diffusivity of water in the membrane at values over 80 % RH (Wiechers 1989). These results may indicate the formation of a hydrophilic pathway, when the stratum corneum is in a highly hydrated state.
6.3.3 Epidermis and Dermis
As the penetrant moves through the epidermis, it must cross cell membranes and diffuse through cells (Monteiro-Riviere 2010). Alternatively, it is possible that small polar molecules could follow an aqueous paracellular pathway through tight junctions to the basal lamina (Brandner 2009; O’Neill and Garrod 2011). By the time the penetrant arrives in the epidermis, it is likely to be separated from the hydrophilic penetration enhancers and other vehicle components. Remaining components can interact in a few interesting ways to enhance absorption rate. Increasing the partitioning into the cell membranes or the fluidity of the membranes will increase diffusion of the penetrant. Metabolizing enzymes in the epidermis may reduce overall bioavailability of the penetrant (Stinchcomb 2003; Storm et al. 1990), though a co-penetrant that inhibits or downregulates expression of the enzymes will diminish that effect.
In the dermis, the vasculature acts as the final barrier between the skin and the systemic availability of the penetrant. Systemic uptake is a function of the perfusion rate and the partitioning between the tissue and plasma. Penetration enhancers that increase blood flow would create a stronger sink condition for diffusion, increasing permeation rate (Riviere and Williams 1992; Wiechers 1989). Co-penetrants that affect protein binding of the penetrant in the extracellular spaces of the dermis or blood plasma will affect the equilibration into the blood and the eventual venous uptake of the penetrant. Finally, like many tissues in the body, the skin expresses several drug transporter systems. The P-glycoprotein (P-gp) transporter system is one of the most important in presenting barriers to drug uptake by tissues. In the dermis, P-gp is concentrated at the vasculature and presents a barrier to drug uptake from the systemic circulation. For penetrants, P-gp may provide a boost to penetration rate that co-penetrants could inhibit (Skazik et al. 2011).
6.4 Experimental Assessment of Interactions
The interaction of vehicle constituents to modify the transport properties of a penetrant can be assessed in several ways. In vivo experiments offer the most direct assessment of effects via endpoints such as C max (maximum plasma concentration) and T max (time of C max), which indicate rate of exposure, and AUC (area under the concentration profile) which indicates extent of exposure. But, in vivo experiments can be expensive, time consuming, unethical (in the case of toxicants), and may not provide much information on the mechanism of the interaction. In vitro and ex vivo experiments provide a way to isolate mechanisms of interaction as well as increase the throughput of experiments. In trying to identify molecular properties of vehicle constituents that contribute to changes in penetrant transport properties, collection of data over several penetrants and vehicle mixture combinations is required and only feasible in a high-throughput system.
6.4.1 Diffusion Cells
Diffusion cells, whether static or flow-through, presents a conceptually simple system for determining skin permeation rate. A donor well above the fixated membrane sample is dosed with a penetrant and vehicle mixture. The penetrant (and perhaps some of the vehicle constituents) partition into and diffuse through the membrane to the receptor well. In the Franz-type static cells, the receptor well is a closed system which is periodically sampled, while in the flow-through cell, fresh perfusate is continually introduced and collected (Bronaugh and Stewart 1985; Franz 1975). In both cases, it is common to include a binding agent such as albumin in the perfusate to create a sink condition on the receptor side. If the boundary layer in the donor and receptor wells is kept small, by stirring or turbulence, or if diffusion is rapid, then the rate-limiting barrier to permeation will be diffusion through the membrane.
The use of different membranes such as silastic and porcine skin allows the combination of the obtained data to factor out the contributions from the membrane and account for effects in the donor and receptor wells, such as solubility, evaporation, binding, and if the penetrant partitioning/binding to silicone membrane is negligible or otherwise accounted for – diffusivity.
6.4.2 Isolated Perfused Porcine Skin Flap
A deficiency with in vitro systems is the inability to assess effects due to perfusion or metabolism which only manifest in viable, integrated tissues. The isolated perfused porcine skin flap (IPPSF) addresses these deficiencies by providing an ex vivo system for percutaneous penetration studies. The IPPSF is surgically prepared in situ on weanling swine and excised after closure of the incisions. The flap is cannulated and perfused with a modified Krebs-Ringer solution containing glucose, antimicrobial agents, and serum albumin to maintain viability. The perfusate effluent can be collected or sampled for analysis to determine the absorptive flux of penetrants placed on the surface of the flap. The integral, viable nature of the IPPSF allows the investigation of effects on permeation and partitioning due to changes in perfusion, metabolism, inflammation, and cytokine signaling (Riviere et al. 1986; Riviere and Monteiro-Riviere 1991).
6.4.3 Model Membrane Systems
Model membrane systems have been developed and used to understand diffusion and partitioning of compounds as well as the effects of vehicles on membrane structure. These systems lack the complex structure and interactions encountered with in vitro and in vivo stratum corneum, such as corneocyte envelop boundaries and metabolic interactions. They do, however, allow for a finer focus on the structural changes induced by vehicle components and potential correlations to transport properties. Lipids extracted from the stratum corneum (SC) can be studied as liposomes or lamellar sheets (Kitagawa et al. 1995; Suhonen et al. 2008; Wertz et al. 1986). Incorporation of phospholipids into SC lipid liposomes increases fluidity of the membranes and partitioning of estradiol, progesterone, and propranolol into the liposomes (Kirjavainen et al. 1999). A study of the interaction of sodium dodecyl sulfate with SC lipid-like liposomes found that increasing cholesterol sulfate content in the membrane increased the resistance of the membrane to solubilization (López et al. 2000). Partitioning of absorption enhancers into SC lipid liposomes has been correlated to increased permeability through fluidization of the membrane (Ibrahim and Li 2010).
Fixation of model membranes to an ancillary structure is an effective approach for enabling high-throughput experiments. The Parallel Artificial Membrane Permeability Assay (PAMPA) has been used for years to assess intestinal permeability (Avdeef 2012). PAMPA consists of membrane constituents fixed on a filter material on a 96-well microtiter plate and placed in a 96-well receptor plate containing buffer. Half of the wells are used with the membrane and half are used as controls (Kansy et al. 1998). Permeant solutions are introduced on the donor side, and flux is measured after a suitable incubation time. Recent developments have led to a modified PAMPA system that shows promise for predicting transdermal permeability (Sinkó et al. 2012). The use of a PAMPA system to predict the effects of vehicles on dermal absorption has been recently reported (Karadzovska and Riviere 2013).
Another high-throughput technique for permeability assay is the membrane-coated fiber (MCF) array, wherein model membranes are immobilized on fibers. The fibers are exposed to the donor solution for a fixed incubation time, after which the entire fiber can be processed by GC/MS. Samples taken well before equilibrium can be used to assess permeability, while at equilibrium they will assess partitioning. Silicone-MCF partition coefficient correlated well (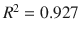 ) with the octanol/water partition coefficient (Xia et al. 2003). The MCF technique is bolstered by a descriptive mathematical model that can be used to regress data and obtain partition coefficient estimates when equilibration time is inconveniently long (Xia et al. 2004). The composite approach (MCF with regression) was used to estimate partitioning and absorption of jet fuel components in polydimethylsiloxane (PDMS-MCF) and polyacrylate (PA-MCF) membranes, showing excellent correlations between partitioning and observed log K o : w (Xia et al. 2005). Extending the idea of using multiple MCFs in an array has led to a unique assay for transdermal permeation using PDMS-MCF, PA-MCF, and Wax-MCF (CarboWaxcoated) partition coefficients jointly to predict porcine skin diffusion cell log K p with high fidelity (
) with the octanol/water partition coefficient (Xia et al. 2003). The MCF technique is bolstered by a descriptive mathematical model that can be used to regress data and obtain partition coefficient estimates when equilibration time is inconveniently long (Xia et al. 2004). The composite approach (MCF with regression) was used to estimate partitioning and absorption of jet fuel components in polydimethylsiloxane (PDMS-MCF) and polyacrylate (PA-MCF) membranes, showing excellent correlations between partitioning and observed log K o : w (Xia et al. 2005). Extending the idea of using multiple MCFs in an array has led to a unique assay for transdermal permeation using PDMS-MCF, PA-MCF, and Wax-MCF (CarboWaxcoated) partition coefficients jointly to predict porcine skin diffusion cell log K p with high fidelity (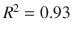 ) in 32 compounds (Xia et al. 2007). The multiple MCF approach has also been applied to the study of vehicle mixture interactions (Baynes et al. 2008; Riviere et al. 2007, 2010). The major limitation of this method is the need for study compounds to be amenable to assay by gas chromatography.
) in 32 compounds (Xia et al. 2007). The multiple MCF approach has also been applied to the study of vehicle mixture interactions (Baynes et al. 2008; Riviere et al. 2007, 2010). The major limitation of this method is the need for study compounds to be amenable to assay by gas chromatography.
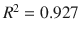 ) with the octanol/water partition coefficient (Xia et al. 2003). The MCF technique is bolstered by a descriptive mathematical model that can be used to regress data and obtain partition coefficient estimates when equilibration time is inconveniently long (Xia et al. 2004). The composite approach (MCF with regression) was used to estimate partitioning and absorption of jet fuel components in polydimethylsiloxane (PDMS-MCF) and polyacrylate (PA-MCF) membranes, showing excellent correlations between partitioning and observed log K o : w (Xia et al. 2005). Extending the idea of using multiple MCFs in an array has led to a unique assay for transdermal permeation using PDMS-MCF, PA-MCF, and Wax-MCF (CarboWaxcoated) partition coefficients jointly to predict porcine skin diffusion cell log K p with high fidelity (
) with the octanol/water partition coefficient (Xia et al. 2003). The MCF technique is bolstered by a descriptive mathematical model that can be used to regress data and obtain partition coefficient estimates when equilibration time is inconveniently long (Xia et al. 2004). The composite approach (MCF with regression) was used to estimate partitioning and absorption of jet fuel components in polydimethylsiloxane (PDMS-MCF) and polyacrylate (PA-MCF) membranes, showing excellent correlations between partitioning and observed log K o : w (Xia et al. 2005). Extending the idea of using multiple MCFs in an array has led to a unique assay for transdermal permeation using PDMS-MCF, PA-MCF, and Wax-MCF (CarboWaxcoated) partition coefficients jointly to predict porcine skin diffusion cell log K p with high fidelity (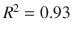 ) in 32 compounds (Xia et al. 2007). The multiple MCF approach has also been applied to the study of vehicle mixture interactions (Baynes et al. 2008; Riviere et al. 2007, 2010). The major limitation of this method is the need for study compounds to be amenable to assay by gas chromatography.
) in 32 compounds (Xia et al. 2007). The multiple MCF approach has also been applied to the study of vehicle mixture interactions (Baynes et al. 2008; Riviere et al. 2007, 2010). The major limitation of this method is the need for study compounds to be amenable to assay by gas chromatography.6.5 Predicting Absorption from Complex Vehicle Mixtures
A quantitative structure to permeability relationship (QSPR) is a mathematical equation that relates a property of permeability, such as diffusion or partition coefficients, to chemical descriptors of the permeant (and possibly vehicle) such as molecular weight, K o : w , dipolarity, etc. An early relationship that could be considered a QSPR related partition coefficient in skin to solubility parameters, which in turn is a method for computing activity coefficients (Barton 1975; Sloan et al. 1986). Sloan reasoned that for small molecules of a similar size, diffusivity would be approximately constant, and therefore the permeability rate constant k p would depend only on partition coefficient. Using a relationship for the activity coefficient of the solute i in solution n:
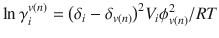 where V is the molar volume, δ i and δ v(n) are the solubility parameters of the solute i and vehicle n, ϕ v(n) is the volume fraction of solvent n (which approaches unity for dilute solutions), R is the gas contant, and T is temperature. Substituting into the definition of partition coefficient:
where V is the molar volume, δ i and δ v(n) are the solubility parameters of the solute i and vehicle n, ϕ v(n) is the volume fraction of solvent n (which approaches unity for dilute solutions), R is the gas contant, and T is temperature. Substituting into the definition of partition coefficient:
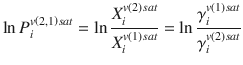 for vehicle n and skin s, gives:
for vehicle n and skin s, gives: