6 Spare-Part Surgery
Summary
The use of discarded tissue for reconstruction is referred to as “spare-part surgery.” It is an important strategy to utilize tissue otherwise wasted. Although there are no core principles or techniques, successful application can be made by understanding the basic concepts of flaps and grafts.
Keywords: spare-part surgery, reconstruction, trauma, microsurgery
6.1 Introduction
Limb-mutilating injuries or oncologic radical amputations are a challenge for the surgeon to restore effective function and appearance of the upper or lower extremities. When tissue needs to be harvested from donor sites for reconstruction, this may be in the form of single tissue grafts such as skin, bone, nerve, or veins, or vascularized composite flaps. These procedures invariably require violation of various sites in the body with additional donor site morbidity. The surgeon needs to weigh cost-effectiveness between the gain of function from reconstruction of the injured limb and the morbidity of harvesting from an uninjured donor site. An alternative strategy is to scavenge tissue for reconstruction from parts that are nonsalvageable amputated sites.1
The “spare parts” concept involves using parts of amputated or otherwise nonsalvageable limbs, allowing the harvest of flaps for reconstruction without additional donor-site morbidity. These undamaged and potentially usable elements include skin, bone, nerves, tendons, vessels, or portions of composite functional units. Fillet flaps, a component of the spare parts concept, can be used as pedicled or free flaps. In severe traumatic injury, pedicled fillet flap is a good option for reconstruction. But in the case of cancer reconstruction, tumor involvement of the proximal limb and trunk often precludes the use of the limb fillet flap as a pedicled flap. Thus, free fillet flap coverage is required.2
Spare parts represent those components that may be overlooked in a pile of presumed unusable and mutilated tissues. Every traumatic injury of the extremity is different; the surgeon must weigh the value of the remains of a mutilated extremity to carefully distinguish the viable from the nonviable. Of what is available, one must then determine the prospective use and contribution of the remaining constituents to the overall function and form of the limb.
The unique aspect of spare-part surgery in trauma is that the surgeon has to assess and create a strategy on the spot to maximize the use of tissues that otherwise would be discarded. This requires surgical expertise, experience, and planning at the time of primary surgery. Certain factors should be taken into consideration in the decision-making, the first being prerequisites that must be satisfied when the surgeon contemplates spare-part surgery.
6.2 Debridement
Debridement is the first step in any limb amputation and it is the only way to reduce bacterial burden for early reconstructive procedure. Debridement aims to convert a dirty contaminated wound into a clean surgical wound by removing all tissue that is devitalized, including the zone of injury, that is of marginal viability, or that is contused and likely to be nonfunctional. The approach is similar in concept to tumor surgery. Especially, there is little role of staged debridement for spare-part surgery. There is only one chance of debridement before doing spare-part surgery for urgent reconstruction. According to Büchler, a conservative approach with preservation of contused and partially devascularized structures can result in fibrosis and scarring, which compromise the ultimate functional result.3
After all life-threatening emergencies are addressed and the patient is medically stabilized, operative surgical debridement and irrigation can be initiated. Thorough removal of all nonviable skin, soft tissue, muscle, bone, and foreign bodies is crucial in obtaining a clean wound bed, reducing bacterial contamination, and preventing subsequent infection.
In the amputated part, however, consistency, color, contractility, and circulation are uncheckable to determine muscle viability. It is also difficult to assess the amount of excision in skin because there is no fresh-bleeding skin edge. Spare-part surgery is usually performed in severely crushed limb, not in clearly cut amputation, so debridement amount can be only dependent on surgeon’s experience.
At the end of the procedure, the wound should be washed with copious quantities of saline, preferably as pulsed lavage or Versajet. The Versajet hydrosurgery system (Smith & Nephew, Key Largo, FL) offers a unique way of performing debridement: a high-pressure fluid jet running parallel to the surface draws devitalized soft tissues into a cutting chamber for excision and evacuation.
6.3 Extracorporeal Membrane Oxygenation Circuit
Spare-part surgery can be a “hot potato,” in the case of marginally crushed amputated limbs in which it is difficult to make a clear decision on whether urgent replantation, spare-part surgery, or stump revision would be the best choice for the patient. Even in the cases of clear-cut amputation with marginal ischemic time, decision-making between urgent replantation and spare-part surgery is still difficult because there is only one chance of utilizing an amputated part. Even in a clear-cut, and not crushed, amputated part with short ischemic time, it requires a couple of days for the patient’s condition to be stabilized after having undergone prolonged general anesthesia.
Greaney et al reported that utilization of the extracorporeal membrane oxygenation (ECMO) circuit allows for preservation of the extremity for several days, thus allowing an extended period of time for patient stabilization.4 The abilities to optimize oxygenation, induce cooling, and titrate the concentration of hemoglobin in the circuit are the additional benefits of isolating the limb on an ECMO circuit. Due to the complex nature of ECMO and the many variables that must be taken into account when implementing this technology, an in-depth discussion of this technology is warranted herein. Among the factors that were carefully evaluated in conjunction with the cardiac perfusion team to optimize tissue perfusion in this patient’s extremity were circuit size, oxygen delivery, anticoagulation, perfusion temperature, and the ECMO’s laminar blood flow.
The concept of isolating the circulation of the limb to prevent harm to the body is not new. However, it is more commonly applied in the setting of isolated limb perfusion for oncologic purposes. Isolated limb perfusion is primarily utilized in the setting of treatment of melanomas and sarcomas that cannot be surgically excised.5 It can also be used to deliver thrombolytic medications in the setting of extensive arterial or venous thrombosis.6
When ECMO is used after cleansing the amputation part, during the couple of days while waiting for stabilization of the patient, debridement can be done to remove all devitalized tissue, as described earlier.
Even though the ultimate limit of viable limb preservation is not defined, it may help extend perfusion time, slow down metabolism, and/or help regenerate damaged tissue during the time of perfusion.7 These also help enhance the survival of each tissue graft such as arterial or vein graft, nerve graft, or bone graft if availability of fillet flap or replantation is decreased.
6.4 Tissue Graft
Tissue graft is the most common form of spare-part surgery. Amputated or nonsalvageable components can be used as sources of tissue grafts. These include skin grafts, nerve grafts, arterial or vein grafts, and bone grafts for reconstruction of the remaining limb. These components fall within the zone of injury and a careful assessment is necessary, especially for vascular and nerve grafts where there is a high chance of surgical failure if injured segments are used.
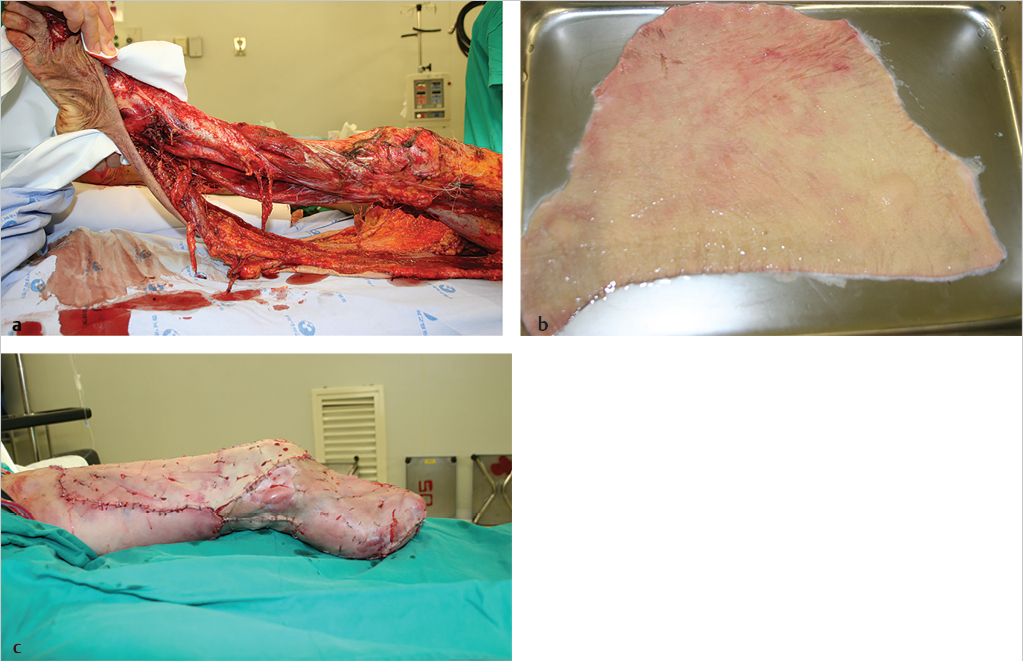
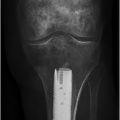
Skin graft from an amputated part can be stored at most for 14 days from the time of injury. This graft can be used not only in emergency operation setting, but also in elective setting after amputation stump is stabilized (▶ Fig. 6.1a–c). Bone is also resistant to ischemia for up to 48 hours.8 Short segment of the bone chip can be useful as a nonvascularized bone graft. However, nerve graft should be carefully chosen because it has limited threshold of ischemia (only 6 hours) and can easily be damaged by traction injury more than any other type of tissue graft.
Arterial or vein graft is also suitable for reconstruction of amputation stumps with flaps. They can contribute to preventing shortening of stump because of vascular crushing injury. Based on the length of ischemia, vigorous irrigation with heparin or urokinase is necessary to ensure washing out of all thrombosis along the graft. Although there are no certain data that demonstrate the time of ischemic threshold of vessels, thrombosis inside of the graft lumen is a major limiting factor of availability of the graft.
Fig. 6.1 (a) A 51-year-old man is shown to have a mangled lower extremity without the chance for limb salvage. (b) In the case of below-knee amputation, there will not be adequate skin coverage. Thus, all the skin was harvested from the amputated part to be used as a graft. (c) After below-knee amputation, the skin was used as a graft.
6.5 Fillet Flaps
By definition, fillet flaps are axial-pattern flaps that can function as composite-tissue transfers. They can be used as pedicled or free flaps and are a beneficial reconstruction strategy for major defects, provided there is tissue available adjacent to these defects.9 The use of fillet flaps can be extended to the reconstruction of major defects caused by tumor resection or to the treatment of recurrent pressure sores.2,10
Fillets of the lower limb have been used to salvage amputation stumps in lower-leg amputations. Length of preservation is of the utmost importance in lower-leg amputations because the performance and quality of life of the patient correlate significantly with the level of amputation.11,12,13,14,15 Amputation level has a significant impact on patient rehabilitation with below-knee amputation (BKA) well recognized as being functionally and aesthetically superior to above-knee amputation (AKA). Functionally, unilateral amputation, distal amputation level, and younger age have been shown to be predictive of better walking ability.16 Comparison of metabolic requirements of walking within a prosthesis also demonstrates energy expenditure of transtibial amputees to be the lowest and that of transfemoral amputees to be the highest.17 Hence, when possible, BKA remains the amputation level of choice. This requires a functional knee articulation, sufficient tibial length, and adequate soft-tissue coverage to provide a well-padded, robust, and ideally sensate stump able to withstand the rigors of ambulating within a prosthesis. If the soft-tissue requirements cannot be met, then further tibial shortening, through knee amputation, AKA, or free tissue transfer, becomes the only remaining option.
6.5.1 Pedicled Fillet Flap
The fillet plantar flap can be easily raised on the posterior tibial neurovascular pedicle and provides adequate size, soft-tissue bulk, stability, and strength to withstand the shear forces associated with walking. Use of the “spare part” provides a single-stage reconstruction, relative ease of flap dissection, and no further donor site morbidity in a patient already undergoing major surgery. With a two-team approach, operative time can be further shortened with harvesting of the flap performed on a side table after amputation of the limb.18
While this flap could be raised as a pedicled flap, preserving continuity of the tibial nerve, this would introduce the difficulties of length dissection within an inflamed field, pedicle redundancy, and risk of kinking during flap inset. Sensory recovery facilitates early weight bearing, better proprioceptive feedback during gait training, and reduction of prosthesis-related complications such as ulcers, neuroma, and pain.
These flaps are reliable. Technical difficulties include placement of the redundant neurovascular bundle in the distal thigh or under the flaps.
Case Example
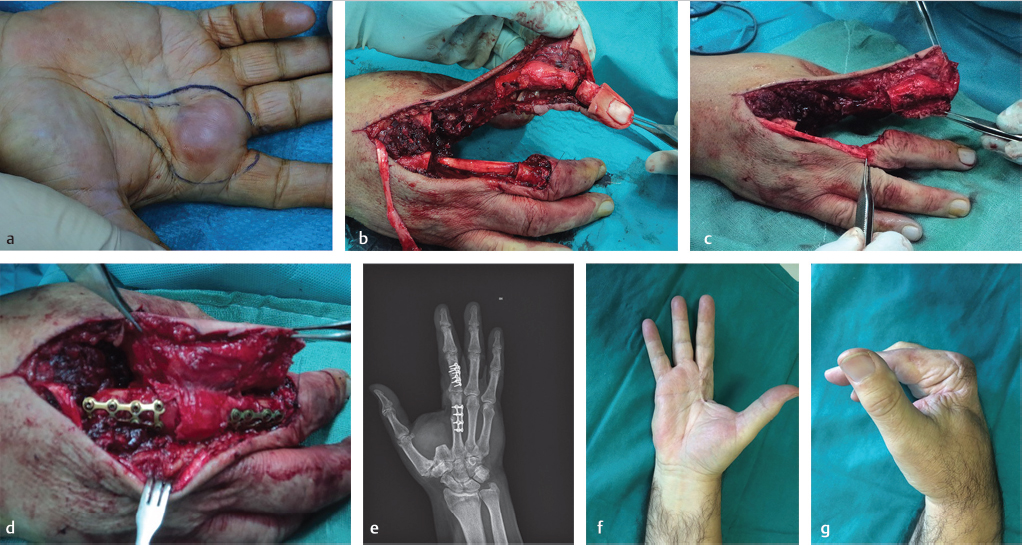
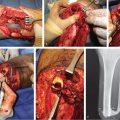
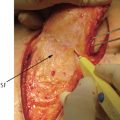
A 64-year-old man had recurrent rhabdomyosarcoma on his right hand. The tumor extended to both the second and third metacarpal heads (▶ Fig. 6.2a). Radical resection of the metacarpal joints was performed including skin, margins free of disease. Pedicled fillet flap based on radial neurovascular bundle was performed including skin and proximal interphalangeal joint of the second ray, which transferred to the metacarpal bone of the third ray (▶ Fig. 6.2b, c). Bony fixation was performed with metallic plate and screw (▶ Fig. 6.2d, e). The reconstructed hand achieved a range of motion of 0 to 80 degrees at 6 months postoperative (▶ Fig. 6.2f, g).