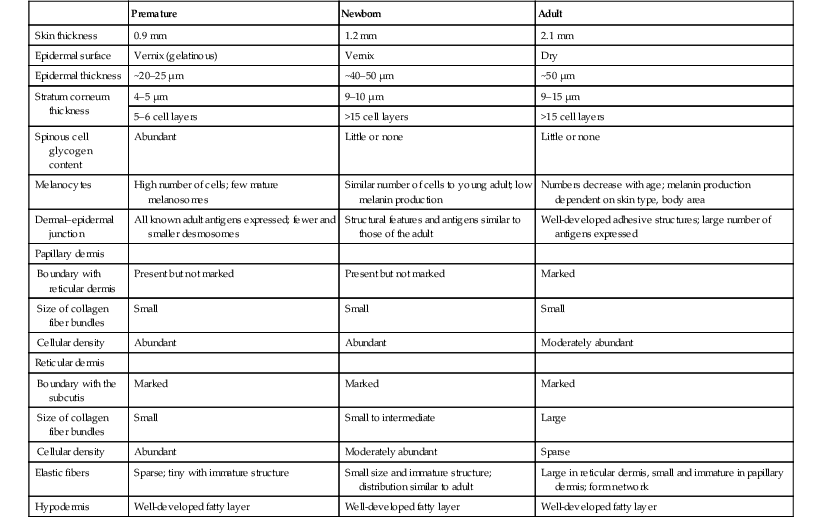
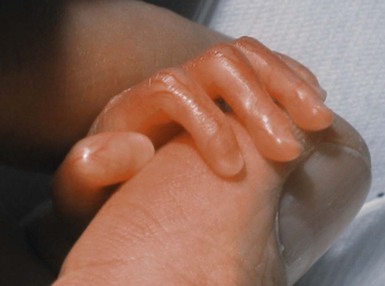
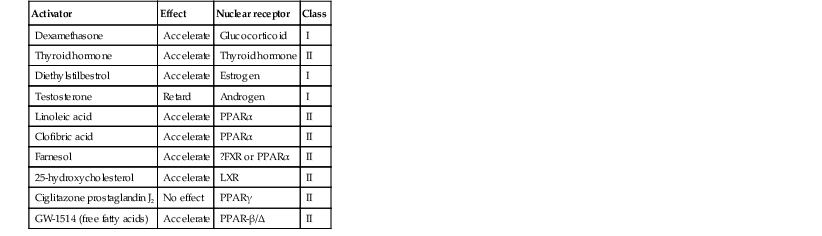
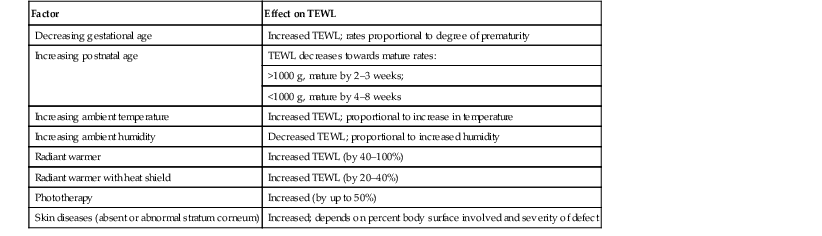
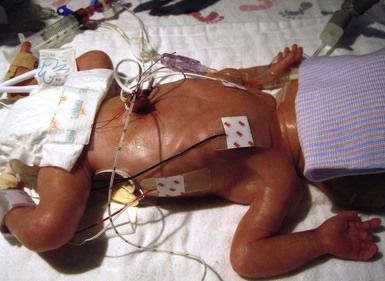
Erin F. Mathes, Mary L. Williams The premature infant assumes the challenge of postnatal life, despite the immaturity of essential functions. Skin functions are primarily protective, and immaturity of the skin contributes to the vulnerability of the preterm infant. The main function of the skin is to provide a permeability barrier that both protects the aqueous interior of the infant from desiccation in the xeric atmosphere and prevents massive influx of water when immersed in hypotonic solutions.1 Other important functions of skin include barriers to percutaneous absorption of exogenous xenobiotics, to injury from mechanical trauma, to colonization and penetration by microorganisms, and to injury from ultraviolet light. In addition to its barrier functions, skin also participates in the thermoregulatory, neurosensory, and immunologic systems. The consequences of skin immaturity for the premature infant depend on the infant’s position on the maturational timetable for each cutaneous function, which is in turn dependent on the infant’s gestational and postnatal ages. All skin layers (i.e. epidermis, dermis, and subcutaneous fat) are thinner in the preterm infant than at term (Table 4.1).2 Because the outermost layers of the epidermis (i.e. the stratum corneum) are the primary effectors of most of the barrier properties of skin, the timetable for maturation of the stratum corneum predicts the competence of many skin functions. Stratum corneum begins to form around hair follicles at about 14 weeks’ gestational age and spreads to include the epidermis between hair follicles by 22–24 weeks’ gestational age (see Table 1.2). During the ensuing weeks, the thickness of the stratum corneum increases from only a few to several cell layers,2 such that by term, it is actually thicker than adult stratum corneum. The ‘excess’ outermost layers of stratum corneum are then shed during the first days of life; this process of physiologic desquamation is accentuated in postmature babies. Another component of fetal skin, the vernix caseosa (a complex proteolipid material) is formed in part by sebaceous gland secretions beginning at about 28 weeks’ gestational age. The percentage surface area covered with vernix peaks at 33–37 weeks’ gestational age, then decreases in full-term and post-term infants. Its functions may include roles in temperature regulation, permeability barrier, and innate immunity.3 The histologic features described above underlie the clinical characteristics of skin maturation embodied in the Ballard scale (see Table 1.5) widely used for assessing gestational age.4 In the extremely premature infant (<24 weeks), the skin is sticky, friable, and transparent (Fig. 4.1); lanugo hairs are absent. As gestation progresses, the skin becomes less transparent, and peeling and surface cracking are increasingly seen, indicative of a thickening stratum corneum, and lanugo hair density peaks and then regresses. Despite definition of these milestones of gross and microscopic skin development, with the exception of the permeability barrier, little is known about the competency or developmental timetable of most skin functions in premature infants. The permeability barrier resides in the stratum corneum through its provision of a hydrophobic lipid shield over the underlying nucleated cell layers.1 Because of their plasma and intracellular membranes, most cells are hydrophobic relative to the vascular and extracellular compartments; however, in the stratum corneum this pattern is reversed. Instead, the extracellular compartment of the stratum corneum is filled with a highly organized series of hydrophobic lipid membranes in the extracellular spaces, whereas the anucleate corneocytes form an aqueous compartment as a result of loss of their plasma and organelle membranes. This interposition of hydrophobic lipid membranes in the extracellular compartment retards the movement of water inward or outward across the stratum corneum. The stacking of multiple layers of cornified cells surrounded by extracellular lipid bilayers further enhances this barrier to water movement, through the generation of a tortuous intercellular pathway for water movement. As a multilayered stratum corneum develops in the third trimester,2 the barrier to transepidermal water loss (TEWL) also matures, such that by 34 weeks’ gestation TEWL rates approximate adult values.5,6 Barrier competence can be measured non-invasively by a variety of direct and indirect means.7–9 By all of these measures, the extent of permeability barrier immaturity parallels the degree of prematurity. In addition to increasing stratum corneum thickness, the development of a competent permeability barrier in fetal rat skin is accompanied by (1) deposition of neutral lipid in the intercellular domains of stratum corneum; (2) increasing stratum corneum cholesterol and ceramide content; and (3) the organization of these lipids into mature lamellar membrane structures, as viewed by electron microscopy.10,11 Whether these same lipid biochemical and ultrastructural changes also underlie barrier formation during human skin development has not yet been determined, but a large body of evidence from other sources predicts that this will be the case. Permeability barrier maturation accelerates after birth, such that by 2–3 weeks’ postnatal age, most premature infants, regardless of gestational age, have competent barriers.6 Thus, maturation that may require approximately 10 weeks to complete in utero is accelerated following premature delivery. However, as the limits of viability have been lowered to include survivors of 25 weeks’ (<750 g) to even 22 weeks’ gestational age, barrier function may take as long as 8 weeks following birth, to mature.12,13 The gestational ages of these very immature infants directly coincide with the timetable for stratum corneum formation (see above). It may not be surprising, therefore, that extremely premature infants do not respond as rapidly to maturational signals initiated by birth. In fetal rat skin, it is air exposure with evaporation of water from the skin surface that stimulates accelerated barrier formation, because this acceleration can be prevented by covering the skin surface with a vapor-impermeable membrane.14 The accelerated postnatal development of human preterm skin may also be prevented if it is covered with occlusive materials or exposed to a humid environment.15,16 Permeability barrier ontogenesis is developmentally regulated; hence, small-for-dates infants exhibit a barrier function that is appropriate for their gestational age.17 In fetal rats, barrier maturation is regulated by glucocorticoids,18 thyroid hormone, and sex hormones, as well as by activators and ligands of the PPARα and LXR nuclear hormone receptors (Table 4.2).11 Some of these agents also regulate lung development; and glucocorticoids often are administered prepartum to mothers to accelerate fetal lung maturation when premature delivery is imminent.19 Whether barrier maturation is also stimulated by these interventions in humans is presently uncertain. One group demonstrated that preterm infants of mothers treated with glucocorticoids have reduced insensible water losses and lower serum sodium concentrations in the first 4 days of life, consistent with a maturational effect on the skin barrier.20 In contrast, Jain and colleagues21 reported that epidermal maturation and barrier function did not appear to be influenced by antenatal steroids. Another aspect of the permeability barrier that undergoes postnatal maturation is the ‘acid mantle’. An acidic pH within the stratum corneum is required for normal permeability barrier homeostasis and for stratum corneum integrity and cohesion. At birth, human stratum corneum has a near-neutral surface pH. This pH declines over the ensuing days to weeks to become acidic, comparable with that of adults. Complete acidification may not occur until 3–6 months of age in term infants.22 Whether this maturation is further delayed in preterm infants is unknown.23–25 The primary consequences of permeability barrier immaturity are: (1) increased evaporative loss of free water from the skin surface, placing the infant at risk for volume depletion, particularly hypernatremic dehydration; and (2) energy loss through heat of evaporation; that is, 0.58 kilocalories (kcal) are expended for each milliliter (mL) of water that evaporates.26 Therefore, optimal care of the premature infant requires both accurate compensation for cutaneous water losses to preserve fluid and electrolyte balance, and maintenance of the infant in a thermally neutral environment, such that caloric intake can be directed toward growth and not heat production.26 Although concurrent cutaneous water losses can be measured directly (i.e. by measuring TEWL), this procedure is not standard practice in most nurseries. Instead, cutaneous water losses and respiratory fluid losses are together considered insensible (i.e. not directly measured). In term infants, TEWL accounts for approximately two-thirds of insensible losses; but cutaneous water losses are much higher in preterm infants, whereas respiratory fluid losses remain relatively constant.27,28 Neonatal fluid requirements are estimated using complex formulas that take into account the following: • Measured losses in urine and feces • Estimates of insensible losses • Requirements to support growth (increasing with post-gestational age). Neonatal fluid requirements must be modified by postnatal age to compensate for fluid redistribution (i.e. requirements on the first extrauterine day are decreased as a result of contraction of the extracellular compartment). There is also considerable variability in TEWL among preterm infants of the same gestational and post-gestational ages; moreover, barrier maturation is often quite precipitous.7,12 Fluid replacements are also adjusted as needed to compensate for excessive weight loss or gain and/or for serologic parameters of fluid or electrolyte imbalance. This inevitably results in ‘chasing fluids’. While the foregoing describes usual nursery practices, it is important to bear in mind that cutaneous fluid losses are not inherently insensible (i.e. unmeasurable). Indeed, it has been shown that measurement of TEWL using rapid and noninvasive instrumentation from as few as three body sites permits accurate estimation of total cutaneous losses in preterm infants.29 Despite this, measurement of TEWL has not been adopted widely by intensive-care nurseries in the approach to fluid management. Fluid replacements must be adjusted for a number of environmental conditions (Table 4.3), because cutaneous losses are not merely a function of stratum corneum maturity. They are also modified by the ambient temperature and humidity, since both affect the vapor pressure of water at the skin surface.17 Phototherapy for hyperbilirubinemia also increases TEWL30 and fluid requirements, particularly when white light systems are used.31–33 Many skin disorders also adversely affect the competence of the permeability barrier (see below and Table 4.3). A humidified incubator (Fig. 4.2) can provide a thermally neutral environment with low rates of evaporative water loss, because at a relative humidity of 80% or more, skin surface evaporation effectively ceases.16,34,35 Scrupulous antisepsis, however, is required to prevent bacterial colonization of this environment, particularly with water-loving organisms such as Pseudomonas spp. Moreover, these devices obstruct access to extremely ill or unstable infants. Therefore, these infants are commonly cared for on an open bed, where a radiant warmer provides a thermally neutral environment at the expense of greatly increased rates of TEWL.34,36,37 Infants requiring care under radiant warmers are typically the youngest and most premature; that is, the population with the poorest skin barriers. Newer hybrid beds with a radiant warmer and humidifier allow for easy access to and observation of infants.35 Use of a plastic cover or plastic bubble blanket may increase the humidity and mitigate to some extent, the adverse effects of the radiant heating on TEWL.38–42 Although these plastic shields are widely employed, standards for thermal stability and transmission have not been established.43 Other strategies to reduce TEWL in the preterm infant include the use of protective skin dressings or ointments. Semipermeable dressings (e.g., Bioclusive®, Omniderm®, Opsite®, Tegaderm®) that permit some passage of water vapor and other gases, but are impervious to water and microorganisms, can reduce TEWL rates and also are protective against the trauma caused by adhesives from monitors.40,44–47 Moreover, barrier maturation is not inhibited by these dressings, and neither have increased rates of infection nor colonization by microorganisms been observed. Nonetheless, increased bacterial colonization under such dressings is observed in other clinical settings48 and remains a serious consideration with their use on preterm infants. In addition, many of these dressings contain adhesive materials, and even those without adhesives can cling to the moist skin surface of the preterm newborn and injure the epidermis, unless they are either removed carefully or allowed to detach spontaneously. Furthermore, partial body applications (i.e. trunk and abdomen) to very immature infants (<1000 g), may not be sufficient to reduce total fluid requirements.44 These limitations hinder the widespread adoption of artificial dressings in routine skin care. Because the benefits of these agents have only been shown in studies with small numbers of subjects, confirmation in larger cohorts is required before their routine use can be generally recommended.42 Topical ointments, such as petrolatum49 or Aquaphor®50,51 reduce TEWL, although the effect from a single application lasts only 4–8 h.50–52 Less frequent applications do not decrease fluid requirements, but they appear to improve the overall condition of the skin and protect against skin trauma.50 Although largely composed of nonphysiologic lipids (e.g., long-chain hydrocarbons and wax esters), these emollients have a long history of dermatologic use without associated toxicity or evidence of significant percutaneous absorption. Nonetheless, internal hydrocarbon accumulations (paraffinomas) are reported, albeit rarely.53,54 Hence, the possibility remains that these lipids may not be entirely innocuous when applied to the skin of very premature infants (see also Chapter 5). A systematic review of randomized controlled trials comparing prophylactic application of topical ointment in preterm infants to routine skin care55 reported that daily application of topical ointment increases the risk for coagulase-negative staphylococcal and nosocomial infections in these patients. Therefore, routine application of topical ointments is no longer recommended for premature infants. However, this should not necessarily be extrapolated to developing countries. Circumstances may be very different in these settings: survival of preterm infants is much lower, care practices differ, and the morbid risks divergent. Indeed, sunflower seed oil and Aquaphor® have been shown to reduce the incidence of nosocomial infections in Bangladesh and Egypt.56–58 An alternative approach to barrier fortification in these infants would be use of mixtures of lipids physiologic to the skin. The extracellular membranes of the stratum corneum that provide the barrier to TEWL comprise an approximately equimolar mixture of ceramides, long-chain free fatty acids, and cholesterol.59 The effect of various mixtures of these physiologic lipids on permeability barrier function has been examined in mature skin in experimental systems, as well as in aged skin and in some skin disorders.59–61 Recent studies in animal and adult human models suggest that in addition to improving epidermal hydration, vernix-based creams may improve the rate of barrier recovery from injury.3,62 Yet, despite the theoretic advantages of employing either physiologic lipid mixtures, or vernix-based creams, these formulations have not been examined for efficacy in treating the barrier immaturity of the preterm infant. Another direct consequence of skin barrier immaturity is the increased absorption of topically applied substances (Table 4.4), sometimes with tragic consequences (see Chapter 5).43,63–65 This vulnerability was first recognized historically when preterm infants developed methemoglobinemia through the absorption of aniline dyes in the laundry marks placed on diapers.66 Subsequently, the demonstration of neurotoxicity due to percutaneous absorption of hexachlorophene, a commonly used antibacterial cleanser,66 led to wider recognition of the vulnerability of the preterm infant to toxicity from topically applied agents. TABLE 4.4 Hazardous or potentially hazardous compounds that may be absorbed across the skin of preterm infants (see also Chapter 5).63–87 a Potentially hazardous compounds. The same factors that determine the movement of water from inside out also regulate the movement of low molecular weight substances from outside in.65 In mature skin, small (<800 Da) hydrophilic molecules are effectively excluded by the extracellular membrane system from penetration across a mature stratum corneum, whereas small hydrophobic molecules or those with amphipathic properties (containing both hydrophilic and hydrophobic parts) are able to penetrate.1,67 In the preterm infant, the thinner stratum corneum results in a reduction in the length (tortuosity) of the intercellular pathway, which would enhance percutaneous absorption of hydrophilic molecules. It is likely that there are also qualitative changes in the lipid composition and/or structural integrity of the lipid bilayers of immature stratum corneum that may further alter its permeability function. In addition to immaturity of the permeability barrier, several other factors in premature infants may contribute to toxicity from topical xenobiotics.64,66
Skin of the Premature Infant
Introduction
The permeability barrier in the preterm infant
Consequences of permeability barrier immaturity
Fluid and electrolyte imbalance and evaporative energy loss
Strategies for minimizing fluid and evaporative energy losses
Increased percutaneous absorption of xenobiotics
Compound
Toxicity
Sources
Alcohol (methylated spirits)
Skin necrosis, neurotoxic
Topical antiseptic, vehicle for topical medications/products
Aluminuma
Neurotoxicity
Metal containers for topical ointments
Aniline dyes
Methemoglobinemia
Laundry marks (historical)
Boric acid, borax
Shock, renal failure
Antifungals, talc powders
Benzocaine
Methemoglobinemia
Topical analgesics; teething products
Benzethonium chloridea
Carcinogen
Antiseptic soap
Benzyl benzoatea
Neurotoxicity
Scabicide
Bicarbonate
Metabolic alkalosis
Baking soda for diaper dermatitis
Camphora
Gastrointestinal toxin, neurotoxicity
Topical antipruritic; camphorated oils (VapoRub®; Campho-Phenique®)
Coal tarsa
Carcinogen
Topical anti-inflammatory products
Chlorhexidine gluconate
Skin necrosis
Topical antiseptic
Corticosteroid
Adrenal suppression, hyperadrenocorticism
Topical corticosteroids
Diphenhydramine
Neurotoxicity
Topical analgesics (Caladryl®)
Epinephrine
High output failure
Topical vasoconstriction
Gentian violet
Possibly carcinogenic
Antimicrobial
Glycerina
Hyperosmolarity
Emollients; cleansers (Aquanil®)
Hexachlorophene
Neurotoxicity
Antiseptic soaps (pHisoHex®) (historical)
Iodochlorhydroxyquin
Optic neuritis
Topical antibiotic (Vioform®)
Imidazoles
Drug interactions secondary to p450 inhibition
Topical antifungal medications: ketoconazole, miconazole, clotrimazole (Lotrimin®)
Isopropyl alcohol
Skin necrosis; neurotoxicity
Topical antiseptics
Lactic acida
Metabolic acidosis
Topical keratolytics (Lac-Hydrin®)
Lindane
Neurotoxicity
Scabicide (Kwell®)
Mercury
Neurotoxicity; acrodynia; nephrotic syndrome
Disinfectants; teething powder (historical)
Methylene blue
Methemoglobinemia
Vital stain (historical)
Neomycin
Ototoxicity
Topical antibiotic (Neosporin®)
Nystatina
Nephrotoxicity
Topical antifungal (Mycostatin®)
Phenol
Cardiac and neurotoxicity
Disinfectants (e.g., commercial laundries); local anesthetic/antimicrobials (e.g., Castellani’s paint)
Propylene glycola
Hyperosmolarity; neurotoxicity
Topical vehicles; emollients, cleansers (Cetaphil®)
Povidone–iodine
Skin necrosis; hypothyroidism
Topical antiseptic (Betadine®)
Prilocaine
Methemoglobinemia
Topical anesthetic (EMLA®)
Resorcinol
Methemoglobinemia
Topical antiseptic
Salicylic acid
Salicylism
Topical keratolytics
Silver sulfadiazine
Kernicterus; argyria
Topical antibiotic (Silvadene®)
Sulfura
Paralysis; death
Scabicide ointment
Triclosana
Neurotoxicity
Topical antiseptic
Urea
Elevated BUN
Topical keratolytics/emollients
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Skin of the Premature Infant
4