and Emanual Maverakis3
(1)
University of British Columbia, Vancouver, British Columbia, Canada
(2)
New York Medical College, Valhalla, NY, USA
(3)
University of California Davis, Sacramento, CA, USA
Zinc plays multiple roles in metabolism, which can be classified into three major categories: catalytic, structural and regulatory functions [1, 2]. Zinc functions as a component of the catalytic site of various enzymes (termed metalloenzymes), which was first described by Keilin and Mann in 1939 after demonstrating that the enzyme carbonic anhydrase (an essential enzyme involved in the metabolism of carbon dioxide) requires zinc for proper catalytic function [3]. In the catalytic site of carbonic anhydrase, zinc was found to function as a Lewis acid by accepting a pair of electrons [3]. In a similar manner, zinc also is essential for the catalytic function of multiple other enzymes including alcohol dehydrogenase, matrix metalloproteinases, alkaline phosphatase and various RNA polymerases [4–6].
Zinc also serves a structural role in the setting of zinc finger proteins, in which the zinc ions stabilize the unique secondary folded structure known as the zinc finger motif and contribute to the function of these proteins [7]. Zinc finger proteins are among the most abundant proteins in eukaryotes and have a diverse array of functions including DNA binding, transcriptional activation and regulation of cell processes such as apoptosis [7]. Retinoic acid receptors and vitamin D receptors also belong to the zinc finger family of proteins [8]. Finally, zinc ions serve an important regulatory function in that they are able to bind zinc-dependant proteins, as occurs with zinc binding to the metal response element transcription factor (MTF) which regulates gene expression in response to the presence of metal ions and oxidative stress [9]. The importance of this transcription factor in eukaryotes is highlighted by the discovery that that a loss of function mutation of MTF in mice was shown to be lethal [10].
Some individuals obtain high levels of zinc through their dietary sources. Interestingly, if a patient with zinc deficiency disorder obtained adequate dietary zinc, general clinical symptoms could be prevented. According to observations, individuals with hereditary zinc disorders, such as AE, that obtain high zinc content through their daily diet are often in less danger of having any severe problems caused by the disorder.
In order to study the clinical effects of deficiency, animal studies are often carried out due to the rarity of zinc deficiency in the modern varied human diet. One of the notable symptoms of AE observed in both children and animals is thymic atrophy and an increased susceptibility to infections: production of thymocytes and T-cells is reduced significantly [11].
It is important to note that zinc requirements depend on the physiological status of individuals. Compared a regular adult, a pregnant woman would have higher zinc requirements. Given the role of zinc in protein synthesis and other transcriptional processes, it would be logical for events that are associated with high metabolic activity, such as growth, pregnancy, and lactation, to require increased amounts of zinc [12].
Neutrophils, peripheral blood monocytes, tissue macrophages and mast cells require a minimum amount of zinc to function properly. In addition, zinc plays an important role in the metabolism of essential fatty acids. In regards to wound healing, zinc is a required nutrient. Zinc deficient patients have a slow and ineffective wound healing process, which is seen to improve quickly when zinc supplements are added to their diet. It is believed that high consumption of zinc through the diet has no pharmacological effect unless the body’s zinc levels are abnormal. Furthermore, role of zinc in treating dermatological disorders is controversial and the effect of this mineral on acne is being studied and some results have been obtained.
7.1 Biochemistry of Zinc Metabolism
Since 1934, zinc has been considered an essential mineral required for growth of mice and rats, while it was still not known as a required nutrient for humans [13]. Finally in 1974, zinc consumption of up to 15 mg per day for normal adults and up to 20–25 mg per day during pregnancy was recommended by nutritional counselors. It is important to note that the average daily diet consumed in the United States supplies approximately 12–15 mg of zinc for an individual. On average the human body contains 2–3 mg of zinc. This amount is almost half of the amount of iron present in the body and 10–20 times more than other elements such as copper, magnesium, and nickel [14].
In solution, zinc often becomes a cation with a charge of 2+ and it is very rare for the Zn2+ ion to oxidize further, which implies its stability. This stability is an important characteristic of zinc.
After its absorption by the intestine, zinc is transferred into blood vessels in different forms: albumin-zinc complex (60–70 %), globulin-zinc complex (10–20 %), transferrin (1–5 %), amino acid (5–10 %), and amino-globulin (less than 1 %). All body tissues contain zinc, but the highest levels are present in muscles, epidermis of skin, liver, kidney, bones, and prostate glands.
Zinc is also present in hair; however, after long term zinc deficiency levels measureably decline and increases in zinc consumption can significantly influence the zinc concentration of the hair. In patients who have been zinc deficient for a only a short time, the concentration of zinc in hair does not decline appreciably and thus is not reliable for accurate evaluation of zinc status of the body [15].
The presence of zinc in body is very important for proper function of many enzymes, such as metalloenzymes [16]. In 1940, Keilin and Mann found that zinc exists in carbonic anhydrase [3]. Moreover, roughly twenty metalloproteinase enzymes are associated with zinc [16].
Zinc also plays a role in bone physiology, especially during the growth and development of young individuals. It was observed that the concentration of zinc in bone is significantly higher than that of other tissues and zinc is considered to be an essential component of the calcified matrix [17].
7.2 Skin and Hair Physiology
Zinc is involved in essential functions of epidermal physiology, especially the keratinization process. Zinc is often found in the granular layer of the epidermis and its concentration in epidermis is six times greater than that in other dermal tissues. The three main functions of zinc in the keratinization process are catalysis, structural and regulatory. The catalytic roles of zinc involve its activation of metalloenzymes, such as RNA nucleotide transferases, RNA polymerase, alcohol dehydrogenase, and carbonic anhydrase. Zinc also has a key role in the formation of structural proteins. The regulatory role of zinc during keratinization includes regulating calmodulin, thyroid hormone binding, protein kinase C, and inositol phosphate synthesis. Calmodulin is responsible for the release of Ca2+ into the cytosol, where the presence of calcium is essential for the activation of epidermal transglutaminase, which is important for development of the keratinocyte. The protein kinase C, also calcium dependent, carries out the phosphorylation of proteins during keratinization. The regulation of calmodulin and protein kinase C is done by the thyroid hormone, and the inositol phosphate increases the level of calcium [18, 19].
Alopecia, dermatitis, and secondary skin infections are the major clinical manifestations of zinc deficiency disorder in humans and animals. Secondary infections are commonly caused by Candida albicans and gram-positive bacteria; while gram-negative infections are caused by Pseudomonas aeruginosa [20]. For example, the activity of the enzymes such as glutamate dehydrogenase and aminotransferase is significantly affected by zinc deficiency. Consequently, amino acid metabolism and protein synthesis, which is required for epidermal regeneration, is considerably diminished in individuals with zinc deficiency. Zinc supplements have been effective in the treatment of many of these zinc deficiency disorders. Furthermore, researchers have observed that the consumption of dietary zinc results in a measurable increase of zinc levels in the epidermal tissue within 72 h of ingestion.
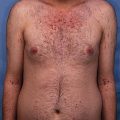
The primary skin lesions, such as bullous pustular dermatitis and polymorphic bulbous vesiculobullous eruptions have been observed as one of the main symptoms that is common within patients who are diagnosed with severe zinc deficiency disorder [21]. A visible feature of these epidermal eruptions is that they have hollow-centered erythematous surfaces. The outbreaks are usually symmetric and often located around the mouth, nose, eyelids, and external ears. The hands, head, elbows, knees, thighs, and hips are also at risk of getting infected [22]. These outbreaks appear in the form of psoriasis on the hips and around the mouth, often in a mask-shaped distribution. Infected lesions may be covered with purulent crusts and have the appearance of impetigo. In addition, lesions on the fingers may appear similar to Acrodermatitis Hallopeau. Cases of stomatitis, glossitis, and vulvitis have also been reported in patients. Losses of hair, eyebrows, and eyelashes have also been noted: strands of hair appear pale and thin and are readily detached without any resistance. As a result, zinc deficiency has been linked with alopecia [23].
7.3 Gastrointestinal System and Other Organs
Gastrointestinal symptoms often appear early in the disease course, coinciding with the onset of the epidermal eruptions. Patients have reported to have had diarrhea three to six times per day with colorless stools and sometimes brown, frothy, greasy and foul-smelling. These symptoms could be as a result of impaired absorption of water and electrolytes in the large intestine. Interestingly, the severity of the epidermal outbreaks and gastrointestinal symptoms are often similar. In addition, gastric and peptic ulcers have been reported as well as diabetes, stomatitis, bleeding of the gums, and nosebleeds have also been reported. In addition, zinc deficiency patients who suffer from stomach ulcers have also reported ulcer healing problems, which can be a fatal condition [24, 25].
7.4 Neurological Development, Growth, and Mental Status
The concentration and distribution of zinc in the brain varies with the stage of development the brain is in. Moderate deficiency of zinc has been recognized as one of the causes of growth failure and hypogonadism in males. Zinc deficiency greatly affects growth and development in children and adolescents. Furthermore, there have been reports of mental retardation, growth failure, mental lethargy, loss of appetite, emotional disorders, weight loss, continuous fatigue, and depression in patients who have been diagnosed with zinc deficiency. There have been reports of permanent mental retardation, schizophrenia, permanent skin damage, dwarfism, and even fatality in untreated patients [26].
7.5 Central Nervous System
There are two pathways through which zinc can reach the brain: brain barrier system and cerebrospinal fluid (CSF) that is formed by plexus choroid [27]. It is interesting to note that zinc primarily reaches the brain through the blood-brain barrier. The influx mechanism of zinc in the brain still remains unclear; however, the high zinc concentration in the parenchymal cells compared to the extracellular fluid is indicative of existence of an energy-dependent absorption in the neurons and glial cells. Yet no specific transporter involved in this process has been identified to date [27].
The distribution of zinc in the central nervous system is uneven, with the most abundant quantity being in the forebrain, more specifically the hippocampus and cerebral neocortex. The male infant hippocampal zinc concentration has been observed to reach an equilibrium within the first 5 years of life [28]. The cerebral zinc concentration has been seen to gradually increase in young individuals while being constant in adults [27]. Quantitatively speaking, the hippocampal zinc concentration is much greater than those of other elements, including calcium and iron [29]. Other parts of the brain, such as the amygdala, substantia nigra, lenticular nucleus, and the thalamus, contain significant quantities of zinc [29].
7.6 Enzymes
The primary catalytic function of zinc involves interaction with metalloenzymes as a cofactor in order to activate them, which often occurs when the zinc cofactor binds and becomes part of the enzyme’s active site. The presence of zinc is essential for the activation of these metalloenzymes [16]. These metalloenzymes, which include DNA polymerases, RNA nucleotide transferases, RNA polymerases, alcohol dehydrogenase, and carbonic anhydrase, have many important biochemical functions throughout the body; such as DNA replication and transcription, metabolism and catabolism of proteins, fats, and carbohydrates.
It has been discovered that the activity of zinc-dependent metalloenzymes is critically influenced by the amount of zinc present. For example, the activity of alkaline phosphatase, which is an active enzyme in the testes, intestine, bones, esophagus, stomach, and kidneys, is greatly affected by the reduction of zinc concentration in tissues; which in turn affects the functionality and performance of the associated tissues and/or organs [30].
7.7 Endocrine System
It appears that the presence of zinc may influence the function of hormones such insulin, ACTH, glucocorticoids, gastrin, growth hormone (GH), testosterone, androgen, prolactin, luteinizing hormone (LH), and follicle stimulating hormone (FSH). For instance, the plasma levels of GH and testosterone were significantly lower in zinc deficient rats compared to normal rats. Zinc deficiency often leads to gonadal growth impairment in sexually immature rat males. Moreover, the administration of GH in zinc deficient mice that have no pituitary glands had no effect on their growth rate; whereas growth rate was increased after the administration of zinc [31]. The effect of zinc on LH and FSH production and secretion will be discussed in Sect. 7.11.
7.8 Nucleic Acids
Zinc plays a fundamental role in the catabolism and biosynthesis of DNA and RNA, as it acts as a cofactor for DNA and RNA polymerases. In addition, zinc may be involved in sustaining the structure of polynucleotides [32].
7.9 Cell Development and Cell Cycles
Zinc deficiency can result in growth disorder and birth defect in newborns, which is due to the regulatory role zinc has in cell division and proliferation. Research has shown that zinc deficiency has adverse effects on majority of the stages of cell cycle. Zinc is considered essential for the progress of biochemical reactions of cells and progression of the cell cycle: passing from G1 phase to S (synthesis phase), from S phase to G2 phase, from the G2 phase to mitosis, and accurate chromosomal disjunction. This is because of the presence of zinc dependent molecules that are significantly involved in the phenomena of replication and/or transcription of DNA; these molecules include chromatin replication enzymes, transcription factors, and regulatory hormones [17, 33].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree