and Emanual Maverakis3
(1)
University of British Columbia, Vancouver, British Columbia, Canada
(2)
New York Medical College, Valhalla, NY, USA
(3)
University of California Davis, Sacramento, CA, USA
5.1 Laboratory Diagnosis of Zinc Deficiency
Presently, measurement of zinc levels in blood plasma or serum is the simplest and most common way of determining the body’s zinc status. However, infections, injuries, and other stress stimuli can alter blood zinc levels and confound the clinical picture when attempting to diagnose AE based on plasma zinc levels. The mechanism for the decreased serum zinc levels observed in the setting of inflammation has not been completely elucidated, but a recent discovery that interluekin-6 (IL-6) upregulates the expression of the ZIP14 zinc transporter in murine liver and may contribute to the hypozincemia seen in inflammatory states [1]. In addition, zinc is distributed to different parts of the body; as a result blood zinc levels may not be reflective of the total body stores. Normal levels of zinc in blood plasma range between 70 and 110 μg/dL, while zinc levels in blood serum range from 80 to 120 μg/dL. Under normal conditions, zinc excretion through urine varies but urinary zinc excretion is significantly reduced in individuals with zinc deficiency [2]. Hair zinc levels are also decreased in patients with AE and it was proposed to detect heterozygous carriers of SLC39A4 mutations [3].
There are several steps the clinician can take in order to maximize the accuracy of the serum zinc concentrations when assessing for potential zinc deficiency. Labs should be drawn in a fasting patient who has not taken any zinc supplements the day of the laboratory test. The sample should be collected in a trace element-free collection tube, and care should be taken to avoid hemolyzing the sample which may falsely elevate the serum zinc level [4]. Fasting zinc levels should be greater than 70 μg/dL, and will be lowered after meals. Serum zinc levels less than 50 μg/dL are suggestive of potential AE, but true diagnosis requires a supportive clinical picture [5].
In vitro or in vivo zinc absorption tests are performed using zinc radioisotopes: zinc-65 or zinc-69 may be performed as confirmatory measures [6]. It should be emphasized that laboratory techniques are very important in determining accurate test results, since contamination of containers and samples and flawed laboratory techniques can alter the patient’s lab results and cause a false diagnosis.
Hyperleukocytosis, anemia, hypertension, as well as low calcium levels, phosphate levels, gastric lipase levels and protease levels are seen in laboratory results of AE patients. In the stool test of these patients, fatty acid content is high (50 % of the cases) and sometimes candida albicans, trichocephalus, and lamblia are found [7].
For patients with borderline laboratory results, a therapeutic trial is recommended to determine if the abnormality is due to a zinc deficiency. This therapeutic trial, also known as a zinc tolerance test, presents an accurate representation of body zinc nutriture and is carried out in the following manner [8, 9]:
1.
After a fast, a baseline plasma level is drawn.
2.
220 mg zinc sulfate (50 mg elemental zinc) is orally administered.
3.
After 2 h, plasma level is redrawn
4.
If plasma zinc is increased by two or three times, then the result is indicative of zinc inadequacy
5.2 Histopathology
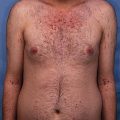
Confounding the diagnostic process, cutaneous lesions of AE do not have specific histopathology. The early skin lesions of AE are characterized by replacement of the granular layer by clear cells and parakeratosis. These skin lesions may have the appearance of eczema or psoriasis. As the condition progresses, the pallor of the upper part of the epidermis becomes more prominent and the parakeratosis is observed to become more confluent. Later on, the pallor is seen to disappear while the psoriasiform appearance still persists [13].
Notable characteristics of AE include the spreading of parakeratosis and psoriatic epidermal hyperkeratosis with large pale keratinocytes on top of lumbar spine and dyscrasia cells, flattened layer of malpighian cells, and absence of granular layer. In addition to its academic advantages, examination of these manifestations and lesions under the electron microscope can be helpful. Furthermore, biopsy examinations can also be conducted in order detect non-specialized changes in enterocytes.
5.3 Biological Diagnosis of Acrodermatitis Enteropathica
Measurement of plasma zinc levels is the most reliable method of determining the amount of zinc present in the patient’s body providing that the sample is not hemolyzed or contaminated. However, there have been cases of falsely low levels of zinc caused by acute stress, infections, and myocardial infarction due to zinc redistribution from blood plasma to body tissues making the assessment of zinc status in the body difficult and inaccurate [14]. On average, zinc concentration in serum is 16 % more than that of plasma due to zinc release in the process of blood clotting and from hemolyzed erythrocytes.
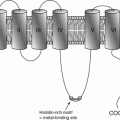
In general, plasma zinc levels do not depend on age or sex of the individuals. One exception is zinc levels in infants younger than 6 months that are approximately 25 % less than the normal levels seen in children and adults. Moreover, zinc levels also depend on albumin levels in plasma. As a result, elderly individuals in whom hypoalbuminemia is more common, have lower zinc levels [15]. Furthermore, leukocytes have high zinc content (56.8–168 μg Zn/1010 WBC), which is roughly 10 times the amount present in erythrocytes (9.3–15.5 μg Zn/1010 RBC). Nevertheless, zinc levels in erythrocytes and hair strands are often used to evaluate zinc status of the body; however, since these cells and tissues have a slow restoration process, measurement of their zinc levels does not properly resemble the body’s recent zinc status. The zinc level in neutrophils is a more precise and accurate resemblance of the body’s recent zinc status.
Among zinc-dependent metalloproteinase enzymes, qualitative assessment of the activity of alkaline phosphatase present in serum and neutrophils may be useful for evaluating zinc status in the body. In the absence of evidence of liver or bone disease, decrease in alkaline phosphatase activity may indicate zinc deficiency [16]. In 1985, this observation was further supported by the clinical research carried out by Weismann and Hoyer, in which the assessment of the activity of alkaline phosphatase was utilized to diagnose mild cases of zinc deficiency as well as cases of AE [17].
In patients diagnosed with AE, zinc levels in plasma, urine, hair, erythrocytes, and leukocytes, as well as the activity of alkaline phosphatase, ribonuclease, and serum LDH have been observed to decrease. Additionally, a strong correlation was seen between the level of plasma zinc and the activity of alkaline phosphatase. Conversely, the activity of erythrocyte carbonic anhydrase did not show any correlation with plasma zinc levels [16, 18].
In cases of zinc deficiency, plasma zinc levels decrease several weeks before clinical manifestations appear. Generally, when plasma zinc levels reach 60–70 μg/dL, the skin manifestations of the disease begin to appear as roughness and dryness. In most AE patients, zinc levels often reached 10–50 μg/dL before any clinical symptoms began to appear. As previously mentioned, urinary excretion of zinc diminishes in patients that have zinc deficiency disorder; therefore zinc excreted through urine, collected within the first 24 h of patient’s admission to hospital or medical clinic, can be useful in the diagnosis provided that the most common causes of second degree hypozincemia such as cirrhosis, sickle cell disease, and uremia are taken into account [19].
5.4 Clinical Diagnosis
Arriving at the diagnosis of AE relies on the ability of the clinician to recognize the constellation of distinct symptoms. However, the presenting signs other than skin lesions often vary with the age of the patient. Clinical findings such as psoriasis manifestations, symmetrical lesions, alopecia, anorexia, neurological disorders, diarrhea, and mood changes have been often reported in infants; while mental retardation, alopecia, weight loss, secondary infections, and growth problems have been reported in toddlers and school aged children.
Laboratory examinations are interpreted in support of the clinical symptoms, but cannot be a substitute in the absence of the appropriate clinical findings. Estimations of zinc levels in serum, urine, and hair are used to diagnose zinc deficiency in individuals. Nevertheless, interpretation of laboratory results is prone to some difficulty because of the potential overlap of the clinical symptoms of AE with coincident findings that may be observed in healthy individuals such as dermatitis. Additionally, low zinc levels in serum, urine, and hair are also observed in other disease and disorders. As a result, the age of the patient and albumin levels of serum are also considered for a more precise and accurate diagnosis. A more specific and accurate laboratory test is often necessary. In vitro or in vivo zinc absorption tests are performed using zinc radioisotopes zinc-65 or zinc-69.
In the absence of diagnostic hypozincemia the above laboratory tests, 3–30 μ mole of zinc per kilogram is prescribed for 5 days. This treatment is especially recommended for infants or children with one or more symptoms of AE. A positive response to this treatment would be a retrospective support of the clinical diagnosis [6, 20].
5.5 Molecular Diagnosis
Confirmation of the diagnosis of AE can be brought by a genetic testing for AE. True AE cases necessarily present either a homozygous or two compound heterozygous mutations of the SLC39A4 gene [21]. Patients who do not have such mutations are to be considered as presenting AE-like disorders, which can either be acquired or due to a genetic predisposition by a mutation in another gene of zinc homeostasis (zinc transporters or metallothioneins). Certain forms of transient zinc deficiencies may be due to mutations of the SLC30A2 gene [22–26].
5.6 Differential Diagnosis
AE-like symptoms of zinc deficiency, which can have numerous different causes, have been observed in infants who were diagnosed with acquired zinc deficiency [2]. This is because some zinc deficiency disorders like dermatosis are clinically indistinguishable from acrodermatitis enteropathica as a consequence of their overlapping symptoms. For instance, perioral and acral dermatitis are the shared symptoms of dermatosis and AE; and both are seen to markedly improve with the intake of oral zinc supplements [27]. Interestingly, there have been reported cases of patients with AE-like eruptions who were suffering from essential free fatty acid and protein deficiencies as well as mild zinc deficiency. In one particular case, the condition of the patient was improved by total parenteral nutrition including amino acids, albumin, lipid, and zinc. Hence it was inferred that all three elements in concert caused her dermatoses [28, 29]. Zinc and essential fatty acids (EFA) interact in a various ways inside the human body; this may be why their deficiencies have similar symptoms. It has been observed that essential fatty acids are important in zinc absorption. Zinc was found to be necessary for at least two stages in EFA metabolism: the conversion of linoleic acid to gamma-linolenic acid and the mobilisation of dihomo-gamma-linolenic acid (DGLA) for the synthesis of prostaglandins. Zinc may also be important in the conversion of DGLA to arachidonic acid and in arachidonic acid mobilisation [29].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree