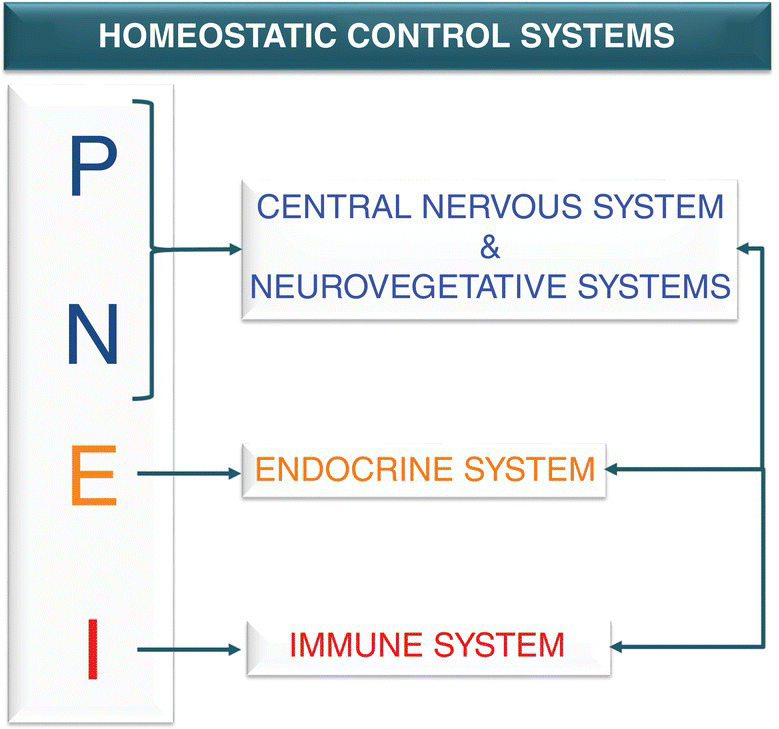
2 Torello Lotti1 and Katlein França1,2 1 Centro Studi per la Ricerca Multidisciplinare e Rigenerativa , Università degli Studi Guglielmo Marconi, Rome, Italy 2 Department of Dermatology & Cutaneous Surgery, Department of Psychiatry & Behavioral Sciences, Institute for Bioethics & Health Policy, University of Miami Miller School of Medicine, Miami, FL, USA In the field of experimental psychology applied to medicine, the second half of the 1980s is characterized by Dr. Rober Ader and his colleagues. They theorized and divulged the principles of Psycho‐Neuro‐Endocrine‐Immunology (PNEI), the scientific field of study that investigates the link between bidirectional communications among the nervous system, the endocrine system, and the immune system, and the correlations of this cross‐talk with physical health. The sentence “For this is the great error of our day that the physicians separate the soul from the body” (Hippocrates, sixth century BCE) clearly represents the primum movens of Dr. Ader’s studies against the traditional scotomized medical view which is described, for example, by the assertion that the immune system is autonomous, with its self‐regulatory and functions separate and independent from the rest of the body. Dr. Ader’s initial research in the 1970s on the conditioning of the immune system by psychosocial factors become a cornerstone for studies that described the vast communications network among immune cells, hormones, and neurotransmitters; Ader’s early observations were also confirmed by Ader himself and other researchers at Harvard University during the 1980s. Thanks to Dr. Ader’s work devoted to the postulation and the development of the new science of PNEI, these old views become less legitimate and, nowadays, the PNEI concepts guide the scientific community to a unified vision of the biological functions of the body [1–4]. The PNEI innovative medical approach represents a paradigm shift from a strictly biomedical view of health and disease taken as hermetically sealed compartments to a more interdisciplinary one. After years of ostracism and diffidence, mind–body interactions are now well recognized, deeply studied in the medical literature, and taught at most important medical schools. In 1983 Dr. Ader wrote: “Converging data from a variety of disciplines suggest that the immune system is integrated with other physiological systems and, like all such systems operating in the interests of homeostasis, is sensitive to regulation or modulation by the brain. Thus, the immune system stands as a potential mediator of a variety of psychophysiologic effects” [5]. The concept of cross‐talk between PNEI system components and the pivotal role of the immune system clearly appear in these sentences. More than 30 years have passed since Ader’s pioneering observations and the PNEI concept is now well established and accepted, despite the initial resistances. An example of a modern, full, integrated medical approach to reduce job‐related distress symptoms (e.g. in healthcare workers) and adrenocortical activity is represented by Psycho‐Neuro‐Endocrino‐Immunology‐based meditation (PNEIMED) an innovative approach that combines the teaching of philosophy and meditation practice with biomedical analysis from a systemic and integrative perspective [6]. Moreover, from the biochemical point of view, the advances in the fields of molecular biology and physiopathology identified hormones, neuropeptides, cytokines, and growth factors as the signaling molecules involved in both physiological and pathological biological processes, are in clear accordance with the principles of PNEI The key element of PNEI approach is represented by the concept of bidirectional cross‐talk [7] between the psychoneuroendocrine and immune systems (Figure 2.1). Figure 2.1 Schematic representation of homeostatic control systems within the PNEI network. The psychoneuroendocrine system can influence the immune response and, therefore, the capacity of the organism to react against diseases; conversely, the immune system can influence the neuroendocrine functions of the whole body. Such cross‐talk among systems is carefully trimmed by feedback loops that simultaneously act in order to maintain the homeostatic equilibrium. This complex interplay is mediated by a wide network of cytokines, hormones, growth factors, neuropeptides, and other intermediate molecules collectively named signaling (or messenger) molecules. These molecules are the “ABC”, the fundamental language of physiological cross‐talk which efficiently regulates cellular responses to both endogenous and exogenous stimuli. The state of health or disease of a whole body can be depicted by the fluctuations of signaling molecules circulating levels: if the fluctuations are outside the homeostatic range (upper or lower than the physiological limits) we consider this status as a pathologic one. Gut and skin roles and relations with other organs and tissues are paradigmatic examples of the PNEI logic. Gut and skin are crucial contact organs through which the mammalian body communicates with the environment. They share some important characteristics: they are richly vascularized and innervated, and they are also heavily colonized by specific microbial strains [8, 9]. Gut and skin can be considered as complex immune and neuroendocrine organs integrated into the whole immune–endocrine system and their correct functioning is crucial to guarantee the homeostasis and, consequently, the survival of the entire organism [10]. All the PNEI axes, such as the Gut–Brain Axis and the Gut–Skin Axis, are multi‐level networks; they are continuously physiologically modulated by the cellular signaling exchange driven by cytokines, neuropeptides, neurohormones, and other messenger molecules. In physiological conditions, this continuous cross‐talk maintains the PNEI homeostasis of the axes. Recently, the concept of Gut–Brain–Skin Axis has been discussed by Petra Arck and colleagues [11]. In 2009 the researchers observed for the first time the connection between the well‐known Gut–Brain Axis and Gut–Skin Axis and, through experimental data (in vivo mice model), described the effectiveness of a probiotic‐based treatment for the reduction of stress‐induced neurogenic skin inflammation and hair growth inhibition. These evidences are in line with the observations of John H. Stokes and Donald M. Pillsbury who first theorized the Gut–Brain–Skin unifying vision in 1930 [12] (a clear example of PNEI approach application ante litteram). Arck and colleagues validated the unifying model Gut–Brain–Skin Axis in order to highlight the idea that beneficial effects on skin homoeostasis and skin inflammation can be achieved by the consumption of the right kind of probiotics. The complexity of the Gut–Brain–Skin Axis induces a deep reflection on its regulation, with particular emphasis on the role of the signaling molecule involved in this network. The imbalance of the signaling molecule at skin level is linked with the majority of inflammation‐related and autoimmune skin diseases (Figure 2.2). Figure 2.2 Schematic representation of the action of proinflammatory triggers on Gut‐Brain‐Skin‐Axis. The Gut–Brain–Skin Axis is a PNEI microcosm that acts as a homeostatic controller not only of its own systems but of the whole organism. Both the intestinal mucosa and the skin have in fact nervous (are able to secrete neuropeptides and neurohormones), endocrine (are able to secrete hormones), and immune (are able to secrete cytokines) competence, and they are in intimate connection with other organs, systems and apparatuses. By virtue of these interactions it appears that the presence of a state of physiological inflammation represents a normal phenomenon both in the intestine and at skin level. The intestinal mucosa and skin are constantly exposed to a heavy antigenic charge, mainly represented by bacterial flora. The tolerance of the microbiota is the key physiological inflammation. These PNEI concepts also offer the opportunity and the tools to study the inflammatory phenomenon in all its complexity, and to identify the homeostatic mechanisms governing all stages of the inflammatory phenomenon, from its onset to its resolution. From a PNEI point of view, inflammation is such an essential physiological process, and it can be homeostatically controlled to trigger, develop, and stop. The healthy status of an organism coincides with the condition of homeostasis, in which the vital parameters (pH, temperature, glycemia, and oxygen’s partial pressure) are maintained within a precise and defined range, and whose deviation up or down is identified with the pathological state. Inflammation is fully embedded in the physiological functions of homeostatic control; there is thus a level of physiological inflammation that falls within the parameters of “normality”[13]. In the intestine, some physiologically controlled inflammation is necessary for immunological function, as regulatory immune cells are triggered by intestinal microbiota and food constituents in order to regulate pro‐inflammatory pathways and maintain the correct immunocompetence. Phlogogenic events such as epithelial barrier disruptions, sudden changes of microbiota composition, altered immune balance, and, finally, homeostatic balance disruption can however contribute to disease onset. Physiological inflammation is overcome by a low‐intensity chronic inflammatory condition called Low Grade Chronic Inflammation (LGCI) [14–16]. LGCI and quali/quantitative alterations of the microbiota may contribute to the onset of local diseases characterized by alterations in the permeability of the intestinal mucosa (IBD [Inflammatory Bowel Disease], IBS [Irritable Bowel Syndrome], gluten sensitivity, and leaky gut syndrome). Systemic diseases such as autistic spectrum disorders, the anxious‐depressive syndromes, Alzheimer’s disease, type II diabetes, obesity, psoriasis, rheumatoid arthritis, BPCO (Bronco‐Pulmonary‐Chronic‐Obstructive pulmonary disease) and RRI (Recurrent Respiratory Infections) are also linked with inflammatory conditions and PNEI homeostasis alterations. Interestingly, skin microbial changes and loss of physiological immunocompetence are also related to some local and systemic diseases such as acne vulgaris, vitiligo, and atopic dermatitis (AD) [9, 12, 17]. Focusing the attention on skin compartment, it is important to remember that the skin’s defense system is composed of three main levels: the skin’s mechanical barrier, innate immunity, and acquired immunity [18, 19]. These levels play specific roles to react against external and internal inflammatory triggers. An example of the intercellular cross‐talk at the cutaneous level is the complex of signaling pathways that regulate the functional interactions between keratinocytes and melanocytes, fundamental for skin pigmentation. Keratinocytes produce growth factors and other signaling molecules which can drive melanocytes’ migration, differentiation, and melanin synthesis. Keratinocyte‐melanocyte cross‐talk represents a small PNEI network at the epidermal level: the psychoneuro component is guaranteed by the embryologic origin of melanocytes which derive from the same embryonic layer that originates some neuronal cell lines, the neural crest [20]. The intercellular cross‐talk between keratinocytes and melanocytes is homeostatically regulated by growth factors and cytokines of endocrine origin. The immune function is linked with and represented by the involvement of melanocytes in the anti‐oxidative stress protective mechanisms mediated by keratinocyte‐derived b‐FGF (basic‐Fibroblast Growth Factor) (Figure 2.3). Figure 2.3 Schematic representation of the keratinocytes‐melanocytes cross‐talk mediated by keratinocytes‐derived signaling molecules (ET; SCF; b‐FGF). These observations highlight the pivotal role of PNEI homeostatic mechanisms in the maintenance of healthy skin conditions. An alteration of skin structure (due to infection or mechanical/chemical injuries) and/or the loss of immune skin homeostasis contributes to the pathogenesis of inflammatory skin diseases that are characterized by the breakdown of the homeostatic cross‐talk. The role played by the immune system in the context of the PNEI network within the “skin system” is crucial for the maintenance of physiological inflammation. The physiological inflammatory process is supported by a panel of Th1‐related cytokines which comprise IL‐1, TNF‐α, and IL‐6 that exert their role in a precise chronobiology. Within 72–96 hours after the proinflammatory stimulus, the response is managed by a sequence of cytokine activation and deactivation: IL‐1 and TNF‐α (primary inflammatory cytokines) induce the production of adhesive molecules, chemokines, growth factors, and lipid mediators such as prostaglandins and nitric oxide (NO). These mediators stimulate leukocyte recruitment at the site of inflammation by amplifying the innate immune mechanisms. Then, IL‐6 acts as secondary mediator, responsible for maintaining the inflammatory response, and stimulates the production of acute phase proteins in the liver. This chronobiology reflects the temporal scan triggering mechanisms and maintenance of the acute inflammatory phenomenon, which is followed by the progressive decrease in the levels of IL‐1, TNF‐α, and IL‐6 and increased levels of IL‐10, the most important Th2 anti‐inflammatory cytokine, typical of the phase of inflammation resolution [21, 22]. In the presence of LGCI, the two phases of inflammation maintenance and resolution coexist. The inflammation is continuously enhanced without an effective restitutio ad integrum; the phases of sequential release of cytokines are altered, and IL‐1, TNF‐α, and IL‐6 levels are about three to four times higher than baseline. Contextually, we do not assist the up‐regulation of IL‐10 anti‐inflammatory. Inflammation persists over time, like a fire smoldering under the ashes. The persistence of an altered immune response to pro‐inflammatory triggers leads to the instauration of a chronic inflammatory process characterized by the absence of the typical signs and symptoms, the LGCI. A relevant number of dermatologic diseases include within their etiologic factors the presence of a shift of the immunological balance, which reflects an imbalance between the cytokines expressed by Th1/Th17 and Treg/Th2 lymphocyte subpopulations [23, 24]. The so called “Th1/Th2 shift” paradigm is supported by the evidence that Th1 cytokine hyper‐production is strictly linked with inflammatory and autoimmune skin diseases such as psoriasis, vitiligo, and alopecia areata. An example of the complexity of the skin PNEI cross‐talk is given by the deep analysis of the inflammatory mechanism at skin level. In 1999, Caroline Robert and Thomas S. Kupper published on The New England of Medicine[25] an exhaustive review on immune imbalance related to inflammatory skin diseases. The authors highlighted the fundamental role of T cell‐mediated immune surveillance in both physiological and pathological skin conditions, pointing out the central role of a class of memory T cells characterized by the presence of the Cutaneous Lymphocyte Antigen (CLA) on their surface and responsible for skin‐homing T cell. CLA‐positive T cells are generated in the draining lymph nodes and recruited back to the skin during inflammation. The presence of LGCI is a potent trigger for CLA+ T cells and their continuous activation is linked with the inappropriate immune surveillance which characterizes, for example, psoriasis, allergic contact dermatitis, and AD. Also, in vitiligo, CLA+ T cells contribute to the massive death of melanocytes driving the skin‐homing (mainly near disappearing melanocytes) of CD8+ T cells at perilesional level. The increased in situ presence of a CLA+/CD8+ T cells is responsible of the destruction of melanocytes, with consequent skin depigmentation [26]. LGCI is one of the most important etiopathogenetic factors of the most dramatic dermatologic chronic inflammatory autoimmune diseases, and consequently a therapeutic target. At present, there are no classical therapeutic opportunities to treat LGCI because the chronic use of anti‐inflammatory active principles studied for the management of acute phenomena shows an unfavorable efficacy/adverse effects balance; in particular, chronic nonsteroidal anti‐inflammatory drug (NSAID) use is connected to an increased incidence of chronic diseases such as heart failure and hypertension [27]. In the 1990s, anti‐cytokine therapy was proposed and tested for the treatment of inflammatory and autoimmune diseases mainly counteracting the expression of Th1 proinflammatory cytokines such IL‐1 and TNF‐α. Moreover, the therapeutic use of Th2 cytokines (e.g. IL‐10) and specific antibodies was applied for alopecia areata, psoriasis, and AD treatment. However, side effects due to high dosages normally used for these molecules have slowed down the development of possible new drugs [28]
Psycho‐Neuro‐Endocrine‐Immunology: A Psychobiological Concept
Bidirectional PNEI Cross‐talk in Dermatology: The Gut–Brain–Skin Axis
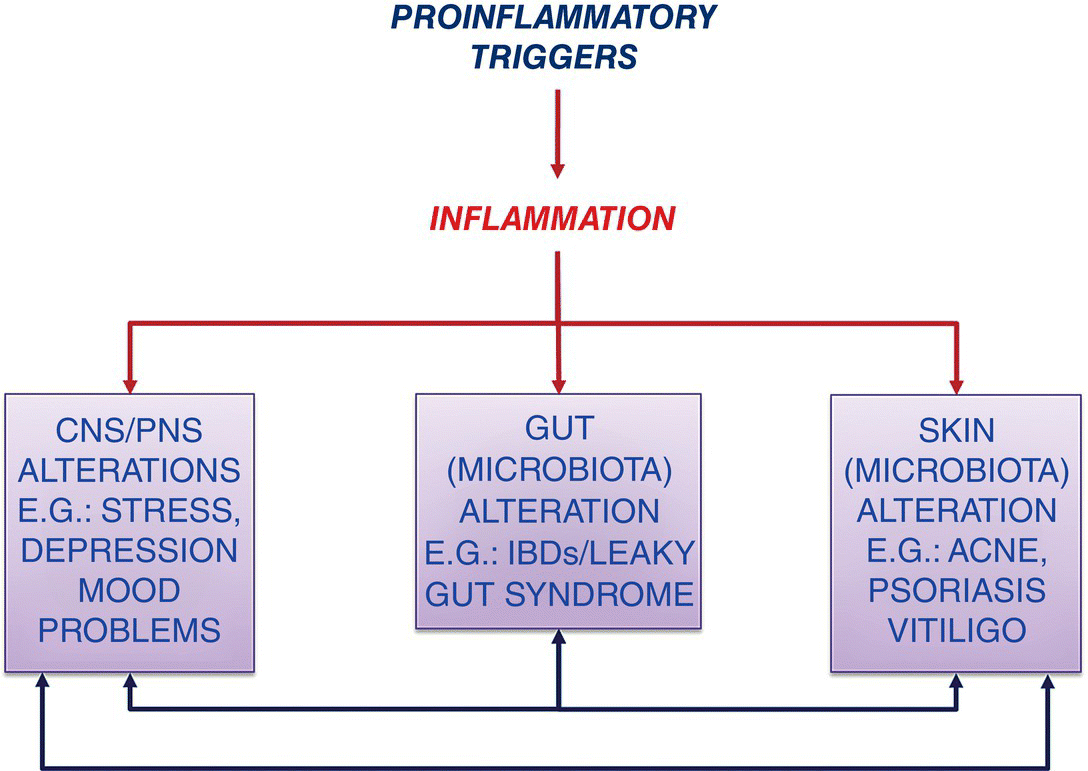
Alteration of PNEI Homeostasis, Inflammation, and Dermatologic Diseases
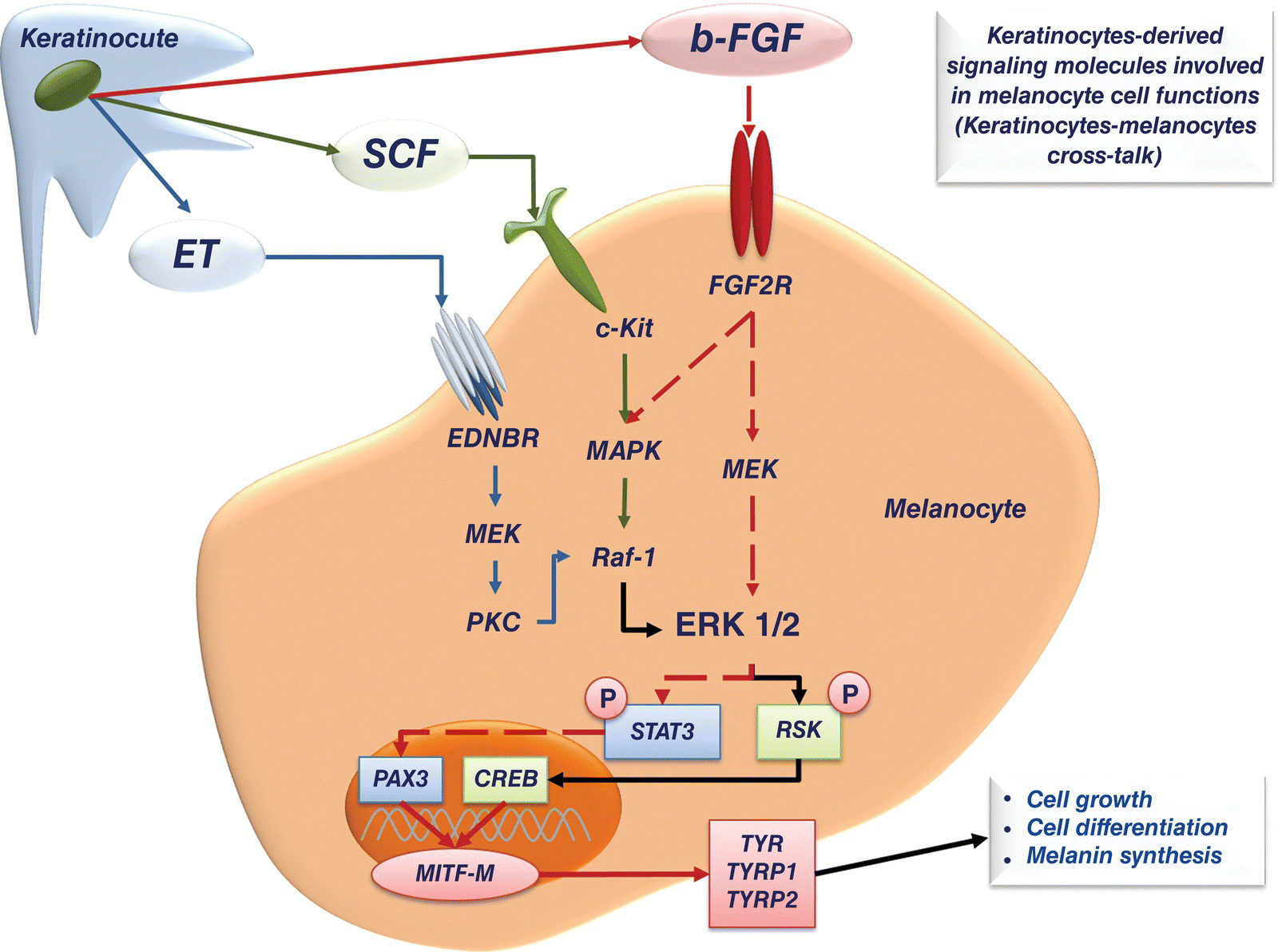
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree