18
Integrative Treatment for Chronic Wounds
Evan Darwin1, Alexandra R. Vaughn2, and Hadar Lev‐Tov1
1 Department of Dermatology and Cutaneous Surgery, University of Miami Miller School of Medicine, Miami, FL, USA
2 Department of Dermatology, University of California – Davis, Sacramento, CA, USA
Introduction
Wounds are defined as a disruption of the skin that results in a loss of function and/or skin architecture [1]. Normal wound healing is a carefully orchestrated biological process in which a series of key phases occur, resulting in epidermal closure and scar formation. If any one of these stages is interrupted, leading to prolonged healing, the wound is defined as a chronic wound. There is no specific timeframe or healing rate under which a wound is considered chronic. However, due to demands of clinical practice and research, arbitrary definitions were made. For example, some suggest in the case of diabetic foot ulcers (DFUs) that a wound not reducing by at least 15% per week, or greater than 50% in a month, can be considered chronic [2]. Other clinicians have defined chronic wounds as those not responding to standard treatment in four weeks, at which point advanced therapies may be used [3].
Phases of Acute Wound Healing
Normal healing proceeds in four distinct phases that overlap considerably in vivo. However, for the sake of didactic clarity, a linear progression is described herein.
Hemostasis
Immediately following an injury, the body utilizes several mechanisms to ensure hemostasis. Exposure of the subendothelial components of the vasculature activates platelets, causing adhesion of platelets onto the subendothelial matrix, platelet–platelet adhesion, and the activation of the coagulation cascade to form a clot [4]. Platelet activation also releases growth factors and cytokines, such as platelet‐derived growth factor (PDGF), that help to begin the healing process [5]. As the healing process progresses, the clot will be removed by the proteolytic enzyme plasmin to allow for blood flow to the healing tissue [6].
Inflammation
The inflammatory stage follows hemostasis. It is defined by the accumulation of neutrophils, macrophages, T‐lymphocytes, and other inflammatory mediators at the wound site [7, 8]. Neutrophils are the first cell type to infiltrate the wound. They destroy invading micro‐organisms through the release of reactive oxygen species (ROS). They also release elastases and collagenases which remove damaged structural proteins, and produce cytokines such as tumor necrosis factor (TNF)‐alpha, and IL(interleukin)‐1 to recruit fibroblasts and epithelial cells [7, 8]. Neutrophils are eventually removed from the wound bed by a combination of apoptosis, necrosis, phagocytosis, and reverse migration [9]. Macrophages infiltrate after neutrophils, and help to remove neutrophils from the wound site [8, 10]. They participate in phagocytosis and also release growth factors and cytokines that help transition the wound from the inflammatory stage to the proliferative phase [8]. It is thought that abnormally activated macrophages are a key factor leading to chronic wounds, and it is the inflammatory stage of wound healing that is most often disrupted in chronic wounds [10, 11]. Lymphocytes are the last to infiltrate wounds and produce IL‐2 which recruits fibroblasts [8].
Proliferation
This stage is characterized by fibroplasia, angiogenesis, and epithelial cell migration [8, 12]. Fibroplasia is the deposition of collagen and structural proteins by fibroblasts which migrate to the wound in response to PDGF released from macrophages and platelets. These fibroblasts act to form the scar tissue and also to draw the wound closed by forming contractile actin bundles [7, 8]. New vasculature must form to supply the wound bed. Angiogenesis is induced during this stage by cytokines such as tumor growth factor (TGF)‐β, vascular endothelial growth factor (VEGF), and β‐fibroblast growth factor (βFGF) [8, 12]. Finally, epidermal cells proliferate and migrate collectively onto the provisional wound matrix toward the center of the wound [7, 8]. The cells release proteolytic matrix metalloproteinases (MMPs) that help to bore a pathway between the scab and the viable tissue [7].
Maturation
The tissue of a newly healed wound is very weak prior to the maturation stage. In the maturation stage, the wound undergoes remodeling and collagen cross‐linking to form stronger tissue. Other important components of the epidermis and dermis, such as hair and pigment cells, re‐form at the wound site [7, 8]. Following remodeling, a well‐healed wound can achieve up to 80% of the tensile strength of normal tissue [8].
Categories of Chronic Wounds
Venous Leg Ulcer
Venous leg ulcers (VLUs) are wounds that form on the lower legs in patients with chronic venous insufficiency (CVI). In the US, 1.5–3 per 1000 adults have leg ulcers, and about 70% of these are due to CVI [13]. Risk factors for the disease include increased age, family history/genetics, elevated BMI, history of superficial or deep venous thrombosis (DVT), and physical inactivity [14, 15].
In patients with CVI, venous hypertension typically results from a varied combination of outflow obstruction (from such sources as chronic DVTs), calf muscle pump failure, arteriovenous malformation or from reflux from faulty venous valves [13]. This results in high pressure in the venous system and extravasation of inflammatory mediators, iron, and fluid. Some theories suggest VLUs develop as a result of trapped inflammatory cells, an imbalance in MMPs, sequestration of growth factors and cytokines, and/or formation of hypoxia from a “fibrin cuff.” However, none of these theories adequately explain the pathogenesis of the disease [15].
The typical patient with venous disease will have a variety of presenting symptoms, often including varicose veins, peripheral edema, hemosiderin staining, venous dermatitis, white scars, (known as atrophy blanche), lipodermatosclerosis, venous starburst of veins radiating from the medial malleolus (corona phlebectatica paraplantaris) and/or ulcers (Figure 18.1) [13, 16]. The ulcers are typically found in an area extending from mid‐calf to approximately 1 in. below the medial malleolus [13]. Typically the wound has an irregular shape, and the wound base has yellow fibrinous tissue and pink granulation tissue [13]. VLUs are often painful, drain significant amounts of exudate, and not uncommonly become infected. Therefore, VLUs represent a significant burden to patients, healthcare systems, and society as a whole.
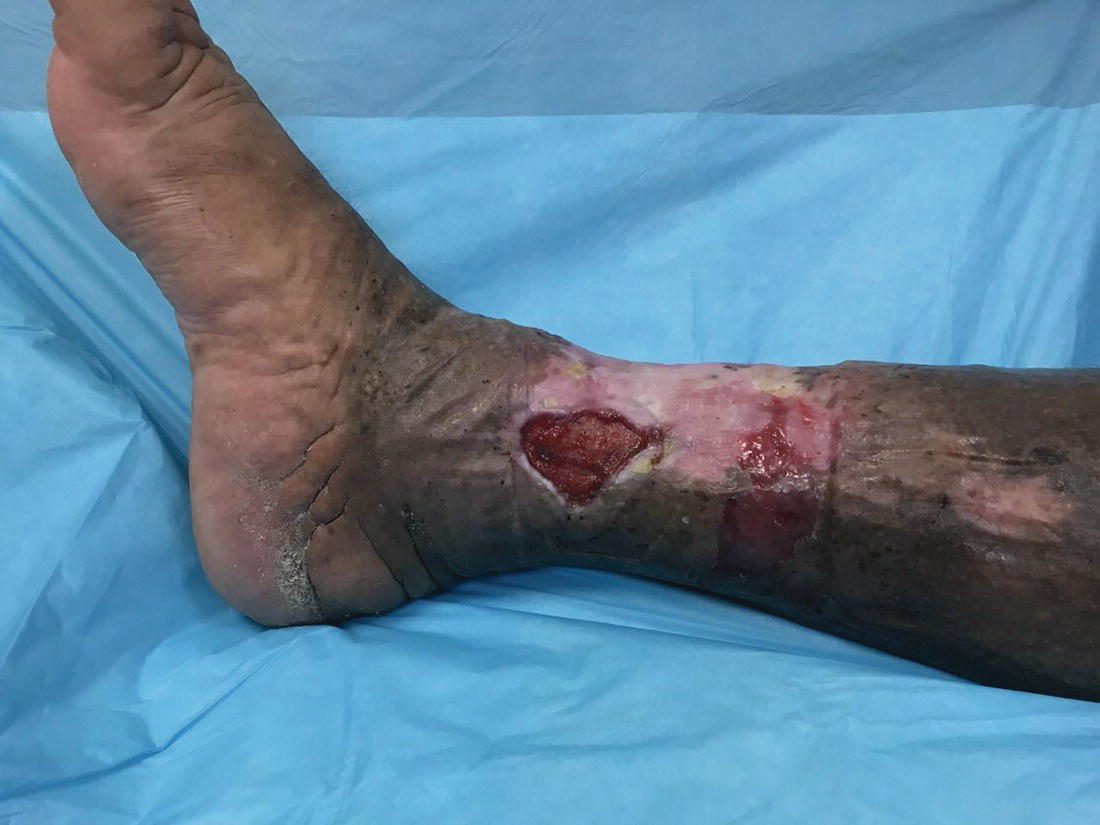
Figure 18.1 Venous leg ulcer.
Diabetic Foot Ulcer
DFUs are extremely common, with an annual incidence of 6.3% in the global population of diabetic patients, and with a lifetime incidence of 19–34% in people with diabetes [17]. Complications of DFUs include wound infection, amputation, and an increased 10‐year mortality risk [17, 18]. DFUs are the leading cause of nontraumatic amputation in Europe and the US [19, 20].
DFUs are primarily caused by diabetes‐induced peripheral neuropathy, leading to skin breakdown due to excess pressure on the foot and gait abnormalities [19, 21]. There is a high association of diabetes with peripheral artery disease (PAD), which may also contribute to ulcer formation [22]. Compounding the problem, the lack of pain sensation in these patients often leads to a delayed diagnosis of the disease [19]. Beyond the peripheral neuropathy, patients with DFUs often have impaired wound healing due to changes in cytokines and growth factors due to longstanding hyperglycemia [23].
DFUs are primarily found on bony prominences on the plantar foot because these areas are at a high risk of having excessive pressure. When evaluating a DFU, the most important signs are those of local inflammation (redness, warmth, pain, swelling, or exudate), which help to establish if the wound is infected. Other signs of infection could include the presence of necrosis, discoloration, friable tissue, fetid odor, or an inability to heal. Osteomyelitis is common in DFU infections, affecting 10–15% of moderate and 50% of severe infections [24]. Additionally, physicians should examine for fever, hypotension, and elevated WBC, although these factors may be absent in up to 2/3 of patients even with severe infections [19].
Pressure Ulcer
Pressure ulcers are a common occurrence in the acute care setting. The International Pressure Ulcer Prevalence Survey in 2015 demonstrated that in acute care facilities, post‐acute care facilities, and rehabilitation facilities, overall prevalence rates of pressure injury was 9.3% [25].
Pressure ulcers are caused by unrelenting pressure on soft tissue, typically over a bony prominence, that is greater than the pressure in the blood vessels supplying the area [26]. This causes ischemia and edema, which eventually can cause injury and potentially irreversible ischemic damage. Other factors that may contribute to pressure ulcer formation include friction, shear force, moisture, and malnutrition [26]. The largest risk factor for the disease is immobility, and pressure ulcers are highly associated with hip fractures and spinal cord injuries [26].
The most common place to develop a pressure ulcer is on the sacrum, followed by the heels and the ischium. Ulcer severity can be graded according to the National Pressure Ulcer Advisory Panel Staging System for pressure injuries, which ranges from non‐blanching erythema to full thickness tissue loss with exposure of tendon, bone, or muscle.
Arterial Ulcers
PAD affects more than 200 million people worldwide [27]. The most severe form, critical limb ischemia, presents as resting pain in the limb, necrosis, ulcers, or gangrene [27, 28]. The prevalence of critical limb ischemia is approximately 1% of the adult population, and up to 10% in patients with PAD [27].
Risk factors for arterial ulcers are the risk factors for atherosclerosis. These include hypertension, hyperlipidemia, smoking, and a Western diet high in saturated fats and highly refined carbohydrates. Less frequent causes of critical limb ischemia include vasculitis, thromboembolism, trauma, adventitial cystic disease, popliteal entrapment, and Buerger’s disease [27].
The basic pathophysiology of the disease is occlusion of macro and microvascular architecture causing downstream ischemia. The tissue exceeds its nutrient supply, and angiogenesis occurs to improve distal perfusion, but may consequently destabilize more proximal blood flow. Typically, the patient will present with absent hair, thin shiny skin, distal extremity‐dependent rubor, and diminished dorsalis pedis and posterior tibial pulses [27, 29]. While often used, capillary refill time is not a well‐validated indicator of peripheral vascular disease [29]. The wounds typically involve the toes first, as they are the most distal part of the vasculature and most prone to have poor blood flow. Ankle brachial index (ABI) and pulse volume recording (PVR) waveform analysis are commonly used to provide the diagnosis of PAD, with an ABI less than 0.9 indicating PAD, and an ABI less than 0.4 indicating a high likelihood of critical limb ischemia [27, 28].
Infectious Wounds
Wounds can also be of an infectious etiology. Bacteria known to cause ulcers include atypical mycobacteria, tuberculosis, diphtheria, anthrax, tularemia, chancroid, granuloma inguinale, and syphilis [30]. Fungi, such as blastomycosis, histoplasmosis, sporotrichosis, and aspergillosis among others can cause ulcers [30]. Viruses such as herpes can also cause ulcers of the skin [30]. While it may be difficult to determine by the wound appearance, the causative organism can often be identified by biopsies, cultures, and special staining in order to treat the wound appropriately [30].
Atypical Wounds
While the most common chronic wounds are described above, less common sources of wounds should also be considered in the case of non‐healing ulcers.
Hidradenitis Suppurativa
Hidradenitis suppurativa is a chronic inflammatory skin disease with unclear etiology, characterized by nodules, abscesses, and scarring of apocrine gland‐bearing skin areas [31]. This disease can be considered when a patient presents with lesions in the axillary, inguinal, or anogenital regions where apocrine glands are present [31]. The lesions present as tender subcutaneous nodules similar to large pimples, evolve into overt nodules, and eventually rupture resulting in exudative abscesses [31]. Over time, the disease can develop draining sinus tracts, fibrosis, and scarring in the area [31].
Vasculitic Ulcers
Vasculitic ulcers are uncommon. There are different types of vasculitis, including granulomatosis with polyangiitis [32]. In patients with multiple purpuric, hemorrhagic, or necrotic ulcers on the lower extremity, a diagnosis of vasculitis should be considered, and an appropriate workup ordered.
Neoplasms
Neoplasms should also be considered in the differential for chronic wounds. Common causes of neoplastic wounds include squamous cell carcinoma of the skin, mycosis fungoides (cutaneous T cell lymphoma), Kaposi sarcoma, or metastatic carcinomas in widespread cancers [30]. The patient presents with individual or multiple cutaneous nodules that become ulcerative. Biopsy should be undertaken in most chronic wounds to rule out a neoplastic pathology.
Radiation Ulcers
Radiation can be an effective treatment for cancer, but it damages skin and surrounding tissues. Skin ulceration can be difficult to heal due to impaired cellular repair and decreased angiogenesis [33]. All radiation ulcers should be biopsied to rule out a recurrence of the underlying malignancy [33].
Congenital Conditions
Several congenital conditions are known to cause chronic wounds. Sickle cell disease is a hematologic disorder found primarily in African Americans and often encountered in clinical settings. It may cause ulcerations of the lower extremity, and is particularly known to form ulcers in the lower third of the leg above the ankle in this population [30]. Another congenital condition known to cause chronic wounds is epidermolysis bullosa (EB). It affects all races, sexes, and ethnic groups. The disease is characterized by skin fragility and blistering that results in painful wounds, infections, scarring, and functional impairment [34]. This disease should be considered in a patient presenting with any kind of family history of EB, and diagnosis can be confirmed with biopsies and genetic studies [34].
Medication‐induced Wounds
Several medications can either induce chronic wounds, or cause traumatic wounds to become chronic. Several commonly implicated drugs include methotrexate, bleomycin, warfarin, heparin, bromine, and hydroxyurea [30]. Hydroxyurea‐induced ulcers are particularly worth consideration in patients with sickle cell anemia. Leg ulcers can be seen in around 9% of patients receiving hydroxyurea for myeloproliferative syndromes, and in 29% of patients with sickle cell anemia [32].
Pyoderma Gangrenosum
Pyoderma gangrenosum is a diagnosis of exclusion in skin ulcers. It can be either drug induced or autoimmune, and is associated with inflammatory bowel disease, connective tissue disorders, and hematologic disorders [35, 36]. The classic appearance is a painful, full‐thickness ulcer with violaceous undermined borders [36]. Histology is usually nonspecific, but may show neutrophilic infiltration [36].
Psychosocial Impact of Chronic Wounds
Chronic wounds significantly impact the social and emotional wellness of patients [37]. In addition to constant pain and discomfort, it is common for people with chronic wounds to feel resentful and frustrated [38–41]. In one study that looked at quality of life in patients with chronic wounds, over 58% reported that having a chronic ulcer was a burden to their life. In addition, the time required to care for chronic wounds showed a statistically significant correlation with feelings of resentment and anger [38]. Pain associated with chronic wounds often disrupts sleep, leading to fatigue and a diminished sense of well‐being throughout the day [41, 42]. In addition, lack of proper sleep can heighten anxiety about the wound [40]. In a study based in Brazil, over 40% of patients with chronic VLUs reported depression and over 30% noted feelings of anxiety [43]. Chronic wounds are notoriously difficult to treat, and understandably many patients feel as though they have lost hope and control; many patients even report losing trust in their healthcare providers [40].
The Role of Stress in Wound Healing
Management of chronic wounds requires frequent dressing changes and constant attention to promote proper healing. This can become very burdensome for patients and lead to immense psychological stress. There is evidence that chronic stress is associated with delayed wound healing [44]. A study in the UK of 43 patients with chronic wounds found a significant association between dressing changes and both acute and chronic stress [45]. In a recent study of patients with DFUs, those with the largest stress response (as measured by heart rate variability) had a significantly slower rate of healing; this study supports the notion that psychological stress may impair wound healing [46].
Physical and Environmental Aspects of Chronic Wounds
Patients with chronic wounds face a number of challenges. In addition to greatly diminished quality of life, the cost of caring for chronic wounds can be an immense financial burden. In a study in Australia, participants with chronic wounds reported an average of 10% of income spent on wound care supply products such as bandages [47]. Another difficulty is the avoidance of physical activity. One study found that there was a large discrepancy in the patient’s perception versus the physician’s perception of chronic pain, demonstrating that physicians tend to significantly underestimate the level of pain caused by chronic wounds [38]. Although healthcare providers recommend increased physical activity to help heal wounds, many patients conversely avoid physical activity and movement for fear of increased pain or making the wound worse [41, 48, 49]. It is important for healthcare providers to recognize both the financial and physical hardships that chronic wounds can impose and thoughtfully create treatment plans that will best enhance wound care compliance for each individual while addressing underlying causes [50].
Conventional Treatment of Ulcers
Local Care
With all wounds, it is important to address common complaints such as pain, odor, bleeding, itch, and exudate. The main goals in local wound care include removal of devitalized tissue, exudate control to achieve a moist wound environment, control of any infection, and supporting a healing wound edge. Choice of appropriate dressing depends on etiology and wound status and may require frequent changes in dressing type to match the evolving nature of the wound. Available wound dressings are examined and compared in Table 18.1 [51].
Table 18.1 Types of dressings.
| Type of dressing | Benefits | Potential adverse effects and limitations |
| Gauze | Inexpensive | Drying; may cause injury on dressing change |
| Films | Occlusive; retains moisture; allows air and water vapor exchange | Should only be used in minimally exudative wounds |
| Hydrocolloids | Occlusive | Should not be used in infected wounds |
| Hydrogels | Rehydrates dry wounds | May overhydrate |
| Foams | Moderately absorbent; minimizes trauma at dressing change | Opaque, can’t visualize wound bed |
| Alginates | Highly absorbent, hemostatic | Not for use on dry wounds |
| Hydrofibers/gelling fiber dressings | Highly absorbent, injury‐free dressing changes. | Not for use on dry wounds, may require secondary dressing for protection. |
| Super absorbent polymer dressings | Highly absorbent, binds matrix metalloproteinases | Not for use on dry wounds. |
Gauze has been used in the past as a wound dressing due to its inexpensive nature. However, it is no longer in favor because gauze may inappropriately dry the wound, cause injury with dressing changes, and slow down the healing process [51]. Films are often used in minimally exudative wounds because they are occlusive and retain moisture at the wound site [51]. Hydrocolloids are composed of cellulose gelatin and pectins that absorb exudate and form an occlusive dressing [52]. However, they can cause skin maceration around wounds, and are not appropriate for highly exudative wounds [52]. Hydrogels are useful dressings in patients with dry wounds, as the gel can rehydrate the site [51]. However, hydrogels may cause overhydration and subsequent maceration [51]. Alginates (seaweed‐derived dressings), hydrofiber dressings, super absorbent polymer dressings, and to a lesser degree foams, are useful in highly exudative wounds. All have absorbent properties, and alginates also have hemostatic properties in the case of chronic wounds with excess bleeding [51–53]. Hydrofiber dressings such as Aquacel® form a gel after absorbing exudate, allowing for pain‐free dressing changes [52, 54]. Superabsorbent polymer dressings can absorb several times their dry weight in water, and recent studies have demonstrated a potential capacity to inhibit collagenase activity and bind MMPs to improve wound healing [55].
Debridement is a key component to wound healing. Devitalized tissue, contamination, and necrotic materials activate abnormal MMPs at the wound site that inhibit healing. Debridement can remove the tissues and biofilms that have formed over the wound site to allow for an optimal wound healing environment. Most commonly, surgical sharp debridement, either with a curette or a scalpel, is performed [56]. Another option is biological debridement using enzymatic debridement with collagenase [57] or the use of maggots [58], but these are not commonly utilized.
Controlling infections is important to wound healing. While bacteria are a normal part of the skin flora and wounds, it is thought that high levels may impede wound healing [51]. Wound infections often present with pain, erythema, swelling, heat, and/or purulent exudate. There are many approaches to prevent and treat infection. Many clinicians use various dressings impregnated with silver, iodine, honey, or other antimicrobials [51]. In the case of severe infection, or if osteomyelitis develops at the site of a deep ulcer, systemic IV antibiotics should be given to prevent severe complications.
Adjuvant Therapies
Negative Pressure Wound Therapy (NPWT)
Negative pressure wound therapy (NPWT) consists of a dressing, an evacuation tube, and a vacuum device. The dressing is placed over the wound, and the NPWT device maintains the negative pressure environment. NPWT improves wound healing by maintaining moisture balance, improving blood flow, removing exudate, and applying pressure to the wound [51]. It is shown to improve keratinocyte migration and stimulate granulation tissue and angiogenesis [59, 60]. Several studies have shown improved rates of growth and reduced rates of infection in patients receiving NPWT [51, 59].
Skin Grafts and Skin Substitutes
Skin grafts have been used for millennia in the treatment of chronic wounds [61]. In skin grafts, healthy skin is taken from an area of the body that is thought to have better healing, and placed on the wound site [61]. Autologous grafts work to improve wound healing both by acting as a replacement and providing growth factors and other stimuli for healing [61]. These skin grafts are classified by their depth, with epidermal grafts, partial thickness grafts, and full thickness grafts being possible therapeutic options [61]. The main problem with autologous skin grafts is donor site morbidity, where this site either has poor healing or poor cosmesis [61].
In recent years, there has also been a major increase in artificial skin substitutes in patients where an autologous skin graft would be inappropriate. Artificial skin substitutes consist of biologically derived substances combined with a vehicle which allows it to be more easily placed on a wound [51]. A variety of substances have been used in these skin substitutes, from porcine xenografts to grafts that incorporate neonatal fibroblasts [51]. The exact mechanism by which skin substitutes improve skin healing is not fully understood, but it has been shown that skin substitutes appear to induce a shift from the chronic non‐healing wound microenvironment to an acute wound environment [62]. Additionally, in the case of substitutes that include fibroblasts, they may provide growth factors and cytokines that help to support a wound healing environment [51]. However, the high cost associated with artificial skin substitutes may limit their practical application [51].
Hyperbaric Oxygen
Patients who receive hyperbaric oxygen enter a chamber with 100% oxygen, and the atmospheric pressure is increased. There are many different ways to administer hyperbaric oxygen, and patients usually need a significant number of treatments [63]. While the exact mechanism to improve wound healing is not known, it is thought that it increases the amount of oxygen in the patient’s blood, allowing better oxygenated blood reach the wound site [63]. The main risks of hyperbaric oxygen treatment are ear discomfort from ear pressure, sinus trauma, and a small risk of oxygen poisoning [63]. This therapy is primarily used in ischemic wounds.
Specific Disease Treatment
Venous Leg Ulcers
The mainstay of treatment for VLUs is local wound care and compression therapy. Compression therapy is essential as it helps to force the fluid from the interstitial space into the vascular and lymphatic compartments, relieving the main progenitor of the disease [64]. While there is high‐quality evidence to show that compression improves the healing of VLUs, there is little evidence to suggest that one type of compression is better than the others. It is thought that higher level compression (34–45 mmHg compression) is more effective than low (15–25 mmHg compression), and that multicomponent compression may achieve better outcomes [64]. In addition to compression, limb elevation is a simple way to improve patient edema and microcirculation, although its direct effects on healing VLUs is unknown [54, 65]. Pentoxifylline, a competitive inhibitor of adenylate cyclase, is used to treat VLUs and has been shown to have anti‐inflammatory properties [54]. In a recent Cochrane review, pentoxifylline plus compression was found to be more effective than compression alone [66]. In addition to medical management, there are now several new endovascular surgical techniques to limit venous reflux, which are thought to also improve healing [64].
Arterial Ulcers
In addition to local wound care, the best treatment for arterial ulcerations is improved blood flow by revascularization [29]. Where revascularization is not possible or ineffective, amputation may become necessary [29].
Diabetic Foot Ulcers and Pressure Ulcers
The key management of these wound types is the same: offloading of pressure. Both wounds are caused by excessive local pressure, and offloading of this pressure by such measures as contact casts, frequent bed turns, or diabetic shoes should improve wound healing. DFUs are also at high risk of infection, and clinicians should have a low threshold of suspicion for infection. Diabetics should be encouraged to improve blood sugar control to reduce the risk of recurrence [17, 67]. In the case of pressure ulcers, nutritional optimization is an effective adjuvant technique (Table 18.2).
Table 18.2 Specific wound therapy in addition to local wound care [17].
| Wound type | Key therapya |
| Venous leg ulcers | Compression therapy, leg elevation, pentoxifylline, endovascular ablative procedures |
| Arterial ulcers | Revascularization surgeries, amputations |
| Diabetic foot ulcers | Offloading, careful monitoring for infection, glucose control |
| Pressure ulcers | Offloading, nutrition optimization |
a These are the key elements of therapy with the highest level of evidence. However, many adjunct therapies are available.
Complementary and Alternative Treatments of Chronic Wounds
Due to the worldwide prevalence of chronic wounds and their notorious difficulty in treatment, there are many complementary and alternative medicine (CAM) regimens that have been clinically evaluated for their efficacy. Standard treatment for chronic wounds can be expensive, painful, and frustrating, which has led to a growing number of patients looking for “natural remedies” to help heal wounds. CAM is commonly practiced throughout the world, with estimates that up to 80% of populations in Asia and Africa utilize CAM as their primary form of medicine, and in China CAM practices represent up to 40% of all healthcare [68]. Despite its prevalent utilization around the world, mechanism‐oriented clinical research is lacking to support adoption in the Western world. Yet, some alternative treatments demonstrate efficacy in the absence of proof of mechanism of action. Among other widely practiced alternative wound healing methods, the topical application of honey, vitamins, and herbs as well as dietary modifications are some examples of CAM therapies, with some evidence supporting their use in clinical studies (Table 18.3).
Table 18.3 Summary of complementary and alternative treatments of chronic wounds.
| Treatment | Role in wound healing | Condition | Study design/level of evidence |
| Honey | Antimicrobial, anti‐inflammatory, and antioxidant properties. | Post‐operative wound infections [69] | RCT/1b |
| Honey may be reasonably used to treat burn wounds, but insufficient evidence to recommend its use in other types of wounds. | Burns [70] | RCT/1b | |
| Honey‐soaked wound dressings have been shown to be more effective than conventional bandages in healing wounds in people with diabetes. | DFUs [71] | RCT/1b | |
| Propolis | Enhances keratinocyte proliferations, stimulates collagen synthesis, and improves wound debridement in burns. | Burns [72, 73] | Animal Study/5 |
| Propolis was as effective as SSD at improving wound healing and reducing inflammation in second‐degree burns. | Burns [74] | RCT/1b | |
| Propolis ointment with elastic bandages was superior to unna boots in rate of wound healing in chronic VLUs. | VLUs [75] | RCT/1b | |
| Vitamin C | Topical ascorbic acid significantly reduced symptoms of burn injury compared to control samples in burn wound skin samples | Burns [76] | Expert Opinion/5 |
| Vitamin E | Topical alpha‐tocopherol applied daily for 20 days enhanced epithelialization, improved angiogenesis, and decreased bacterial load in superficial burns, posttraumatic ulcers, and skin graft wounds. | Burns, posttraumatic ulcers, and skin graft wounds [77] | Nonrandomized clinical trial/2b |
| Vitamin E and Aloe vera impregnated films enhanced penetration of vitamin E in skin of 5 healthy volunteers, offering promising potential for use in wound healing. | Heathy Volunteers [78] | Nonrandomized clinical trial/2b | |
| Topical vitamin E applied to designated surgical sites in children for 15 days prior, immediately after, and for 30 days following surgery had a 0% rate of keloid development after six months, while 6.8% of the control group (petrolatum only) developed keloids. |