15
Nonmelanoma Skin Cancer
Anagha Bangalore Kumar1, Yasser Al‐Qubaisy2, and Katlein França3,4
1 Department of Medical Oncology, Mayo Clinic, Rochester, MN, USA
2 Department of Dermatology and Dermatological Surgery, Prince Sultan Military Medical City, Riyadh, KSA
3 Department of Dermatology & Cutaneous Surgery; Department of Psychiatry & Behavioral Sciences, Institute for Bioethics & Health Policy, University of Miami Miller School of Medicine, Miami, FL, USA
4 Centro Studi per la Ricerca Multidisciplinare e Rigenerativa, Università degli Studi Guglielmo Marconi, Rome, Italy
Introduction
Nonmelanoma skin cancer (NMSC) is the most common type of cancer in Caucasians [1]. It is estimated that 75% of NMSC is attributed to Basal cell carcinoma (BCC). Cutaneous squamous cell carcinoma (SCC), from epidermal keratinocytes, is the second most common NMSC [2, 3]. A small fraction of NMSC is also formed by Angiosarcoma, cutaneous lymphomas, dermatofibrosarcoma protuberans, Merkel cell carcinoma, and tumors of adnexa [4]. It is estimated that at present, 5.4 million new cases of NMSC are being diagnosed every year worldwide, and the highest burden of NMSC is in Australia [5, 6]. In this chapter we will be limiting our discussion to BCC and SCC.
Etiology
There are various environmental and genetic factors implicated as causative agents for NMSC. UV (ultraviolet) radiation is the most significant risk factor for NMSC. The incidence of BCC is higher in people with white skin, on sun‐exposed areas, and increased in sunnier zones. It is interesting to note that SCC is more often associated with chronic UV exposure whereas BCC is more often seen in those with childhood and intermittent sun exposure [6, 7]. UV light is thought to induce DNA damage and also form reactive oxygen species (ROS) which potentially encourage carcinogenesis [8]. Tanning beds are known to increase the incidence of both SCC and BCC [9, 10]. Phototherapy, especially psoralen ultraviolet A (PUVA) therapy, has also been studied as a risk factor for NMSC [11].
Genetic mutation on chromosome 9q22 is associated with basal‐cell nevus syndrome [12]. Mutations in various G‐protein receptors like PTCH and SMO (members of hedgehog pathway) as well as other genes coding for Cytochrome 450, glutathione S‐transferase, and p53 have been implicated in the causation of BCC [13–18]. SCC is mostly seen to arise from pre‐existing lesions like actinic keratosis (AK) and Bowen’s disease, perhaps portraying cumulative damage over time [19]. It is estimated that 70%–90% of SCC has p53 mutations. Mutations of WNT, Ras, p16INK4, NF‐k Beta (nuclear factor kappa‐light‐chain‐enhancer of activated B cells), and c‐Myc have all been implicated as possible causative agents for SCC [20]. Human papilloma virus (HPV) has been shown to prolong the keratinocyte cell cycle and increase destruction of p53, and has been implicated as a causative agent of SCC [21, 22]. Organ transplant recipients are at an increased risk of developing NMSC when compared to the general population [23]. Increased age, arsenic exposure, leukemia, lymphoma, chronic inflammation, and chronic wounds are all risk factors for NMSC [24, 25].
Basal Cell Carcinoma (BCC)
BCC arises from the pluripotent cells in the basal layer of the epidermis, and rarely from the outer root sheath of hair follicles, sebaceous glands or other cutaneous appendages [26]. These are notorious for local tissue destruction. However, they rarely metastasize.
BCC is the most common cancer in humans and has been increasingly detected in younger populations [27]. The incidence in men is higher than that in women. It is estimated that the incidence in the US can be as high as 226–343 per 100 000 populations [28]. It is rarer in people with darker skin tones [29].
Etiology
Ultraviolet radiation (UVR), mostly comprising the UVB spectrum, is known to induce genetic mutation in the suppressor genes [30]. Multiple inherited BCCs can be seen in families with PTCH mutations [31]. Albinos and people with Skin types I and II are prone to develop BCC with prolonged actinic damage. Tanning beds also predispose to the development of BCC [32]. Particularly, a history of heavy sun exposure and sunburns at younger age predispose to BCC [33]. Arsenic ingestion has also been described as a predisposing factor for BCC. It can take about 30–40 years for BCC to develop after the ingestion of arsenic [34, 35].
Clinical Features
These lesions are slow growing and typically asymptomatic. In the later stages, there might be erosions and bleeding with trauma. They occur as single lesions and most of them occur on the face, especially the medial and lateral canthi, nasolabial folds, and behind the ears. BCCs occur on skin that has hair follicles [26].
Nodular BCC present as papules or nodules which are translucent or pearly in appearance. They can develop telangiectasias on their surface and are usually well defined and firm. Flakes of melanin or superficial erosions might also been seen on their surface [26].
Ulcerating BCC are typically called the “rodent ulcer crusted base and rolled out borders.” They are translucent, firm, and may have superficial telangiectasias [26].
Sclerosing BCC lesions have a small morphea‐like patch or scar which can be ill‐defined, and have associated white or peppery pigmentation. This is an infiltrating form of BCC. There is abundant fibrosing stroma in this form of BCC and in histopathology; one can appreciate the finger‐like strands of tumor tissue extending into the adjacent tissue. Sclerosing BCC can progress to nodular or ulcerating BCC [26].
Superficial multicentric BCC is the only form of BCC where superficial scaling can be present. These present as pink‐red plaques with a thread‐like border and telangiectasias. These can progress to nodular or ulcerating forms. These can bleed with excoriation and this finding is helpful in differentiating from solar keratosis [26].
Pigmented BCC appear as brown to blue or black, smooth, shiny, firm masses. These are a form of nodular BCC with increased pigmentation. Cystic lesions with umbilications and stippled pigmentation might occur as well [26].
Fibroepithelioma of pinkus classically present as pink papules on the lower back [26, 36].
Histopathology of BCC
Atypical basal cells are seen with large, deep blue staining on Hematoxylin and eosin preparations as cells with less cytoplasm and large nuclei. Peritumoral lacunae are slit‐like retractions from surrounding tissue, and their presence might point to the diagnosis of BCC [37]. There is little anaplasia and mitosis with palisading arrangement. Nodular BCC has nodules of large basaloid cells with stromal retraction. Pigmented BCC appears similar to nodular BCC with the addition of abundant melanin pigmentations in between tumor cells. Morpheaform BCC, also called infiltrative/sclerosing BCC, has dense fibrous stroma with embedded tumor cells [26].
Diagnosis is mainly clinical and biopsy is usually confirmatory. The clinical differential diagnosis includes nevi, trichoepithelioma, dermatofibroma, and other tumors. Ulcerative BCC should be differentiated from other ulcerative lesions [26].
Basal‐cell Nevus syndrome (also called Gorlin syndrome or Nevoid BCC syndrome) is caused by mutations in chromosome 9q. Usually seen in the Caucasian population, this BCC occurs early in life. Over hundreds of BCC lesions can be present at once. They grow throughout life needing multiple surgeries. Continuous lifelong surveillance and scarring pose as challenges in managing these cases. Mostly these lesions occur on sun exposed parts, but they might also occur on covered areas. The characteristic appearance is frontal bossing and a broad nasal root with hypertelorism. Undescended testes, hydrocephalus, odontogenic cysts, defective dentition, splayed ribs, and spinal abnormalities are described associations. Ocular abnormalities like strabismus, hypertelorism, cataracts, glaucoma, and colobomas can also occur [26].
Squamous Cell Carcinoma (SCC)
SCC is a malignant tumor of keratinocytes arising in the epidermis. It is often seen developing from pre‐existing precancerous lesions [38]. UVR is a known culprit of SCC. It is more prevalent in sunny areas, especially in Caucasian populations [39]. A recent study estimated that the incidence in men is twice of that in women [40]. Excessive PUVA radiation also predisposes to SCC [41]. HPV types 16 and 18 are most often implicated in the causation of SCC [42]. The incidence is higher in people with immunosuppression, such as individuals with AIDS and organ transplant recipients [43]. It is also seen as a consequence of chronic inflammation and chronic injury. Exposure to carcinogens like arsenic, tar, and crude paraffin can also lead to development of SCC [26, 43].
Clinical Features
Most lesions are slowly evolving and asymptomatic. One has to be cautious and attentive to lesions in suspected patients. The lesions of SCC are common on sun‐exposed sites. They are commonly seen arising from pre‐existing lesions except in immunosuppressed patients where they are eruptive and multiple [44].
Actinic keratosis is one of the precancerous lesions of SCC which usually occurs as multiple lesions with dry adherent scales and usually ill‐defined margins. In contrast, Bowen’s disease is generally solitary, well‐defined scaly papules or plaques. Bowen’s disease may appear verrucous in anogenital areas and eyelids, or as a dark oozy patch in intertriginous areas [38].
SCC usually presents as a skin‐colored or flesh‐colored keratotic papule, ulcer, verrucous plaques, or cutaneous horn. The margins might be ill‐defined. Some lesions might extend on to surrounding tissues or neighboring nerves [38].
Oral SCC appears from precancerous lesions like erythroplakia. It is associated with tobacco chewing, cigarettes, and alcohol consumption. Lower lip SCC arises from previously developed actinic keratosis and is usually secondary to chronic sun exposure. Genital SCC is frequently associated with HPV infection. It usually appears as a warty nodule or erythematous plaque. Erythroplasia of Queyrat is a precancerous lesion of SCC on the penis. Genital SCC is also associated with poor hygiene, phimosis, and lichen sclerosis et atrophicus. SCC also occurs from the margins of chronic wounds/ulcers (Marjolin’s ulcer) and scar tissue [38].
Keratoacanthoma is a variant of SCC that presents as a firm, large, smooth nodule with a central keratotic crater. It is usually seen in sun‐exposed areas [45].
Verrucous carcinoma develop at sites of chronic irritation and develop as verrucous lesions [46].
Metastatic SCC can present with involved lymph nodes or as newer lesions on the skin. Newer multiple keratoacanthomas should be worked up for internal malignancies [38].
Histopathology of SCC
Extension of atypical keratinocytes from the epidermal layer beyond the basement membrane and into the dermis is a telltale sign of SCC. The surrounding tissue might show solar elastosis when the tumor is induced by sun damage, or have abundant scar tissue when the tumor develops secondary to scarring. The grading of the tumor depends on the degree of cellular differentiation, loss of intercellular bridges, and amount of keratin production. Well‐differentiated squamous tumors reveal foci of keratinization with concentric squamous cells termed as horn pearls. Also, histological subtypes of SCC like tubular, verrucous, signet‐ring shaped, and clear cell SCC have been described [38, 47].
Role of Sunscreen Agents
The protective role of sunscreens in preventing melanoma is well documented. However, there seems to be some controversy about the protective role of sunscreens in NMSC [48]. Thompson et al. reported that there were fewer lesions of actinic keratosis after applying high sun protection factor (SPF) sunscreens daily over one summer [49]. Similarly, Naylor et al. in their two‐year study found that people who used high SPF sunscreens would have a lower rate of new precancerous lesions [50]. Fewer SCC were noted after daily sunscreen application in a four‐year follow up study by Green et al. In the same study, the authors report no reduction in the incidence of BCC with regular sunscreen agents [48]. However, the carcinogenic potential of UVR should not be underestimated and appropriate sun protection has to be sought.
It is advisable to use sunscreens as an adjuvant protective element rather than the sole factor for protection against UVR. It is important to avoid UVR, especially between 10 a.m. and 4 p.m. Wide brimmed hats and clothing should complement sunscreens. A high SPF sunscreen should be applied liberally and frequently on all exposed parts for adequate protection. These measures are very important in children as childhood sun exposure has been linked to a higher incidence of melanoma and BCC later in life [51].
Management
The type of tumor, tumor characteristics, patient preference, and availability of services influence the treatment of NMSC. Topical therapy may be useful in low‐risk cases, and for high‐risk cases the gold standard treatment for SCC and BCC is Moh’s micrographic surgery (MMS). If MMS is unavailable, excision with a wide margin with or without radiotherapy is an option [25].
Excision with Predetermined Margins
Studies have shown that with standard excision with predetermined margins, we can expect a five‐year cure rate of over 98% for BCC and 92% for SCC [52]. Wolf and Zitelli in their perspective study concluded that surgical excision with 3 mm margin would achieve a cure rate of 85%, and 4 mm wide margins would have 98% cure histologically [53]. Similarly for SCC, Brodland and Zitelli suggested that tumors that are 2 cm in diameter or less, when excised with a 4 mm margin, would achieve a cure rate of over 95%, and for tumors with diameter more than 2 cm an excision with a margin of 6 mm would achieve a cure rate of 95% [54].
Mohs Micrographic Surgery (MMS)
MMS was first described by Sir Frederic Edward Mohs in the early 1940s. With MMS all the peripheral and deep margins are examined for residual tumor by the surgeon. After all the margins are clear of tumor tissue, the wound can be closed by sutures, with a flap, or with a graft [55, 56]. MMS has shown to have the greatest cure rates amongst all treatment types of NMSC. Numerous studies have shown that MMS achieves a five‐year cure rate of 99% for BCC and 97% for SCC. Even for recurrent tumors, MMS is the most efficacious modality of treatment (Figures 15.1 and 15.2) [57–60].
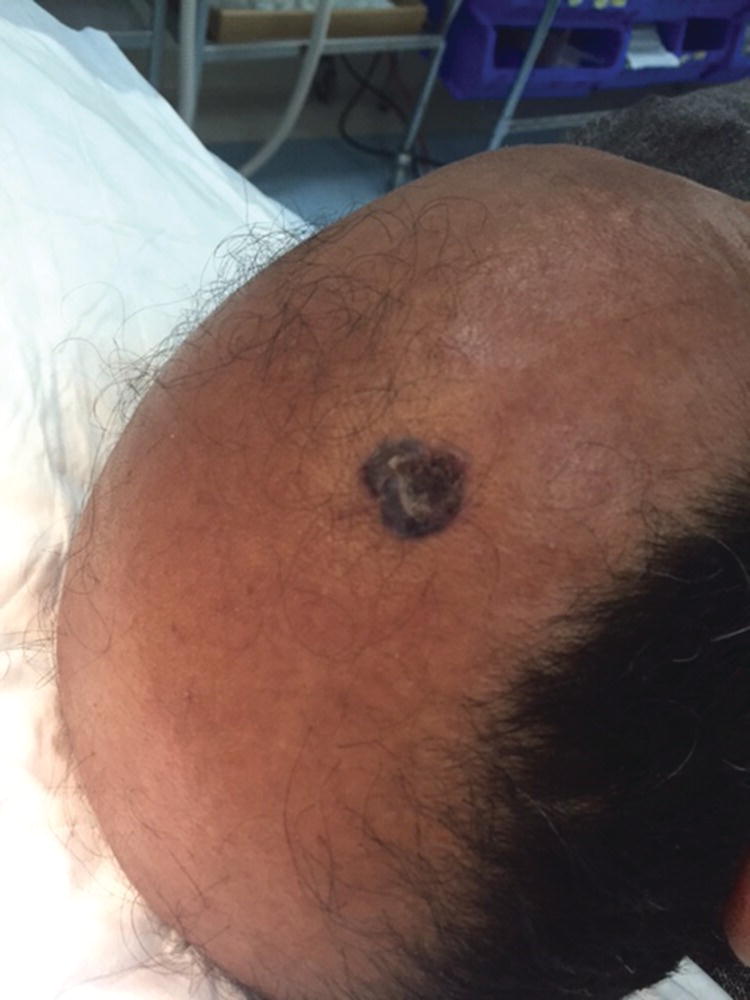
Figure 15.1 Basal cell carcinoma on the scalp before Mohs Micrographic Surgery.
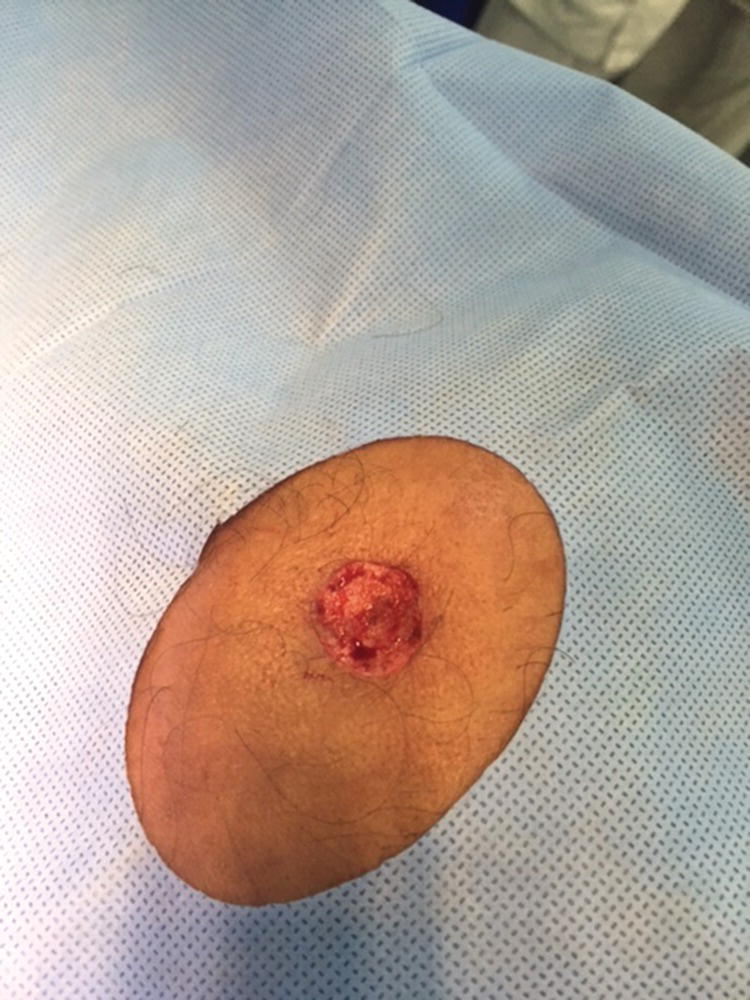
Figure 15.2 After Mohs Micrographic Surgery – lesion removed.
Radiotherapy
Radiotherapy is an effective treatment modality for patients with NMSC who are not eligible candidates for surgery. It can have high five‐year cure rates up to 90% for both BCC and SCC [52, 57]. Adjuvant radiotherapy is advised for tumors with perineural invasion. Radiotherapy is also recommended when sufficient tumor margin is not able to be resected [61]. However, it is not recommended in younger patients and in those with genodermatosis [62].
Curettage and Cautery (C&C)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree