One of the most significant benefits of translational research in dermatology has been the development of prenatal diagnosis for couples at risk of recurrence of severe inherited skin diseases. Indeed, over the last 30 years a greater understanding of the molecular basis of epidermolysis bullosa (EB), as well as technical refinements in laboratory procedures, has facilitated the development of several different approaches for prenatal diagnosis. Initial tests were based on fetal skin biopsy sampling, but these have largely been superseded by DNA based analyses, mostly using fetal DNA derived from chorionic villus sampling taken at around 10–12 weeks’ gestation. Further advances, however, have led to the introduction of licensed preimplantation genetic screening for some forms of EB, an approach that defines a disease-associated genotype before implantation into the uterus. Pioneering research also continues to try to develop less invasive approaches with the prospects of maternal blood sampling early during the first trimester as a feasible objective. The availability of several different options for prenatal diagnostic testing therefore has led to an increased choice for families at risk of recurrence of EB.
Recent progress in clinical and molecular genetics have helped refine the diagnosis and management of epidermolysis bullosa (EB), although the new discoveries have yet to lead to substantial advances in treatment through gene, protein, cell or drug therapies. Disease prevention therefore remains the main option for couples at reproductive risk of EB and one of the major translational benefits of research has been the development of prenatal diagnostic testing.
The techniques have changed over the years, from being heavily reliant on analysis of fetal skin biopsy (FSB) samples acquired during the second trimester to the examination of DNA from first-trimester chorionic villus samples. Furthermore, efforts to design simpler, less-invasive methods of prenatal diagnosis that can be performed earlier in pregnancy, without compromising the sensitivity of the assay and the accuracy of the results, are ongoing. In the absence of a cure for EB, prenatal testing along with appropriate counseling is an integral part of the management of families at risk of some forms of EB.
History of prenatal diagnosis
Before the availability of accurate prenatal diagnosis, couples at reproductive risk were faced with limited options. These options included normal conception and acceptance of the risk of having an affected offspring, avoidance of future pregnancies, adoption, artificial insemination with screened donor sperm, or egg donation and in vitro fertilization with the spouse’s sperm.
The first prenatal diagnostic examination for a subtype of EB was reported in 1980. These innovative prenatal tests involved the ultrastructural examination of FSBs but were only relevant to a limited number of disorders, including severe forms of junctional and recessive dystrophic EB. The early biopsies were performed during the second trimester with the aid of a fetoscope to visualize the fetus. This involved the insertion of a fiberoptic endoscope into the uterus, under sedation and local anesthesia. However, with improvements in sonographic imaging, FSBs subsequently could be taken under ultrasound guidance.
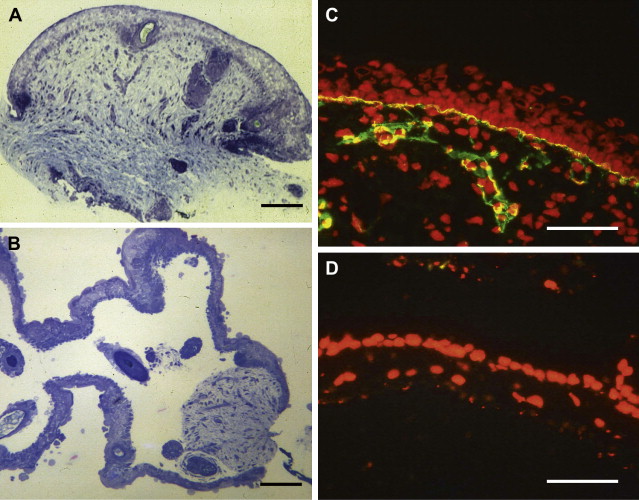
FSB samples obtained during the early 1980s could only be examined by light microscopy and transmission electron microscopy. For EB, the diagnosis was made by finding a split at the dermal-epidermal junction by light microscopy ( Fig. 1 A, B), and then the precise level of cleavage was determined by electron microscopy. The introduction of a number of monoclonal and polyclonal antibodies to various basement membrane components during the mid-1980s, however, led to the development of immunohistochemical tests to help complement ultrastructural analysis in establishing an accurate diagnosis ( Fig. 1 C, D).
Although FSB has been largely superseded by DNA-based prenatal tests in the majority of cases, FSB may still be indicated if insufficient DNA data are available. Couples can be counseled that FSB sampling is an invasive procedure with an approximately 1% rate of fetal loss more than the background incidence of spontaneous abortion. Sampling error, inadequacy of samples for analysis, and difficulty in interpreting the morphologic and immunohistochemical features can pose problems and may necessitate repeat sampling. From a practical perspective, FSBs cannot be performed before the 16th week of gestation, and the prospect of a second-trimester termination of an affected pregnancy is often associated with considerable emotional and physical distress. It is also important to note that there are few centers with the necessary experience and expertise to undertake FSB procedures and to adequately analyze the very small pieces of fetal skin.
Recently, it has also been demonstrated that immunohistochemical labeling of other fetal material, notably chorionic villi, can also be used for prenatal diagnosis. It has been shown that villous trophoblasts, sampled during the first trimester, display immunoreactivity to α6β4 integrin and plectin, thereby permitting diagnosis/exclusion of EB associated with pyloric atresia through tissue diagnosis.
Development of molecular diagnostics and progress in prenatal testing
As the molecular basis of EB has been elucidated, FSB has gradually been superseded by DNA-based diagnostic screening using fetal DNA from amniotic fluid cells or chorionic villi ( Fig. 2 ).
After implantation of the embryo, the chorion, the outermost layer of the embryonic sac derived from the trophoblast layer of the blastocyst, attaches to the uterine wall. The chorion is lined by microscopic projections referred to as chorionic villi. These projections are the fetal components of the placenta and contain the same genetic material as the fetus; therefore, they are a useful source of fetal DNA.
Similarly, the cells within the amniotic fluid, surrounding the fetus, are derived from fetal epidermis as well as the gastrointestinal and genitourinary mucosae. Chorionic villus sampling (CVS) is usually performed between 10 and 12 weeks’ gestation, whereas amniocentesis, the method for obtaining amniotic fluid and its cells, is conducted later, at approximately 16 weeks’ gestation. Therefore, CVS is the favored method for DNA-based prenatal testing in many units.
The most severe forms of EB are usually inherited in an autosomal recessive manner. Couples who have already had 1 affected child can be offered DNA-based prenatal testing in subsequent pregnancies if the causative gene is known and the pathogenic mutations have been identified. Indeed, initial reports of molecular prenatal testing for EB were published in 1995.
Before any prenatal test, however, samples from both parents and any previous affected siblings are analyzed for pathogenic mutations. This initial screening is crucial for accurate genetic counseling and to establish the reliability of the prenatal test, as it determines the pattern of inheritance, tracing the transmission of the mutated gene(s) from generation to generation. In autosomal recessive EB, ideally both parents must be shown to be heterozygous carriers of the pathogenic mutation(s). The possibility of de novo mutations, nonpaternity, and uniparental disomy (the inheritance of both copies of a chromosome pair from just 1 parent) should be excluded before considering the suitability of the prenatal test. For DNA-based prenatal tests, fetal DNA is extracted from the chorionic villi or amniotic cells and analyzed for genetic mutations. After CVS, tissue obtained needs to be cleaned under a dissecting microscope to exclude maternal cells, such as decidua or blood, which could contaminate the sample and affect the accuracy of the results. The actual analysis of fetal DNA can usually be accomplished between 48 and 72 hours after its receipt in the laboratory. Couples can be counseled that the risk of fetal loss following these procedures is approximately 0.5% to 1%, depending on the expertise of the unit conducting the procedures.
Since the initial DNA-based tests were introduced for EB, many more severe inherited skin disorders have been diagnosed or excluded prenatally in couples at reproductive risk, including harlequin ichthyosis, tuberous sclerosis, congenital erythropoietic porphyria, Netherton syndrome, ectodermal dysplasia/skin fragility syndrome, and Ehlers-Danlos syndrome. With advances in molecular diagnostics, DNA-based prenatal testing is now feasible for any severe inherited skin disorder for which the causative gene is known and the pathogenic mutations and/or informative markers are defined.
Despite the advances in prenatal diagnostic techniques, reproductive choice and preventative options for couples at risk of EB are still limited. With all the prenatal tests mentioned so far, the diagnosis can only be made once pregnancy is established, with the subsequent option of terminating an affected pregnancy. This raises fundamental moral issues for many couples at risk. Moreover, some couples will not consider termination of pregnancy because of religious reasons or personal beliefs.
Development of molecular diagnostics and progress in prenatal testing
As the molecular basis of EB has been elucidated, FSB has gradually been superseded by DNA-based diagnostic screening using fetal DNA from amniotic fluid cells or chorionic villi ( Fig. 2 ).
After implantation of the embryo, the chorion, the outermost layer of the embryonic sac derived from the trophoblast layer of the blastocyst, attaches to the uterine wall. The chorion is lined by microscopic projections referred to as chorionic villi. These projections are the fetal components of the placenta and contain the same genetic material as the fetus; therefore, they are a useful source of fetal DNA.
Similarly, the cells within the amniotic fluid, surrounding the fetus, are derived from fetal epidermis as well as the gastrointestinal and genitourinary mucosae. Chorionic villus sampling (CVS) is usually performed between 10 and 12 weeks’ gestation, whereas amniocentesis, the method for obtaining amniotic fluid and its cells, is conducted later, at approximately 16 weeks’ gestation. Therefore, CVS is the favored method for DNA-based prenatal testing in many units.
The most severe forms of EB are usually inherited in an autosomal recessive manner. Couples who have already had 1 affected child can be offered DNA-based prenatal testing in subsequent pregnancies if the causative gene is known and the pathogenic mutations have been identified. Indeed, initial reports of molecular prenatal testing for EB were published in 1995.
Before any prenatal test, however, samples from both parents and any previous affected siblings are analyzed for pathogenic mutations. This initial screening is crucial for accurate genetic counseling and to establish the reliability of the prenatal test, as it determines the pattern of inheritance, tracing the transmission of the mutated gene(s) from generation to generation. In autosomal recessive EB, ideally both parents must be shown to be heterozygous carriers of the pathogenic mutation(s). The possibility of de novo mutations, nonpaternity, and uniparental disomy (the inheritance of both copies of a chromosome pair from just 1 parent) should be excluded before considering the suitability of the prenatal test. For DNA-based prenatal tests, fetal DNA is extracted from the chorionic villi or amniotic cells and analyzed for genetic mutations. After CVS, tissue obtained needs to be cleaned under a dissecting microscope to exclude maternal cells, such as decidua or blood, which could contaminate the sample and affect the accuracy of the results. The actual analysis of fetal DNA can usually be accomplished between 48 and 72 hours after its receipt in the laboratory. Couples can be counseled that the risk of fetal loss following these procedures is approximately 0.5% to 1%, depending on the expertise of the unit conducting the procedures.
Since the initial DNA-based tests were introduced for EB, many more severe inherited skin disorders have been diagnosed or excluded prenatally in couples at reproductive risk, including harlequin ichthyosis, tuberous sclerosis, congenital erythropoietic porphyria, Netherton syndrome, ectodermal dysplasia/skin fragility syndrome, and Ehlers-Danlos syndrome. With advances in molecular diagnostics, DNA-based prenatal testing is now feasible for any severe inherited skin disorder for which the causative gene is known and the pathogenic mutations and/or informative markers are defined.
Despite the advances in prenatal diagnostic techniques, reproductive choice and preventative options for couples at risk of EB are still limited. With all the prenatal tests mentioned so far, the diagnosis can only be made once pregnancy is established, with the subsequent option of terminating an affected pregnancy. This raises fundamental moral issues for many couples at risk. Moreover, some couples will not consider termination of pregnancy because of religious reasons or personal beliefs.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree