and Emir Q. Haxhija2
(1)
Department of Plastic Surgery and Burns, Institute for Mother and Child Health Care of Republic Serbia, University of Belgrade, New Belgrade, Serbia
(2)
Department of Pediatric and Adolescent Surgery, Medical University Graz, Graz, Austria
Keywords
Pigmented lesionChildrenTreatment10.1 Introduction
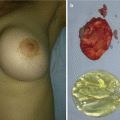
The skin is comprised of two basic layers: the epidermis that contains four major cell types, keratinocytes, melanocytes, Langerhans cells, and Merkel cells, and dermis that contains nerves, blood vessels, lymphatics, muscle fibers, pilosebaceous, and apocrine and eccrine units [1]. Melanocytic nevi are moles with localized proliferations of pigment cells (melanocytes) in the skin [1, 2]. The exact mechanism how nevi develop in the skin is not known [2]. In children, pigmented lesions can pose significant diagnostic and therapeutic challenges [2–4]. By the end of 10 years, nevus count reaches more than 30 in white (5–10 in African, Asian, and Native American population) [3]. Melanocytic nevi presented at birth are named congenital melanocytic nevi (CMN), and all other nevi developed after birth are named acquired melanocytic nevi (AMN) (Fig. 10.1) [2–7]. Melanoma is extremely rare in childhood, but there is tendency of rising incidence [2–4]. Ultraviolet (UV) light represents the primary environmental factor, increasing the number of total body nevi which positively correlates with melanoma risk [3, 4].

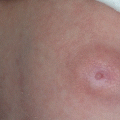
Fig. 10.1
Acquired melanocytic naevus
10.2 Congenital Melanocytic Nevi
Congenital melanocytic nevi (CMN) are classically defined as being present at birth, and they represent a disruption of the normal migration, proliferation, and differentiation of melanoblasts [2–6]. In the developing fetus, congenital melanocytic nevi CMN are thought to arise between the 5th and 24th week of gestation [2, 5].
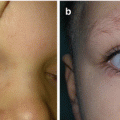
The classification of CMN has been standardized and updated in 2012, based on size, location, number of satellite nevi, and additional morphologic characteristics [3, 7, 8]. The following characteristics of nevi are included: a) Size of CMN projected adult size: Small < 1.5 cm; Medium—M1: 1.5–10 cm, M2 > 10–20 cm; Large—(L1: >20–30 cm, L2: >30–40 cm); Giant—G1 > 40–60 cm, G2 > 60 cm; Multiple medium CMN—3 or more medium CMN without single, predominant CMN; b) Location of CMN: head, trunk, and extremities with subgroups; c) Number of satellite nevi: S0, no satellites S1 < 20 satellites, S2, 20–50 satellites, S3 > 50 satellites; and d) Additional morphologic characteristics: color, rugosity, nodules, and hairiness (Fig. 10.2a–d) [4, 8]. Approximately 2–3% of the neonates have CMN [3]. The incidence of large CMN is estimated at 1 in 20,000–50,000 live births [2, 3, 6, 9, 10]. The very large (“bathing trunk”) CMN occur in approximately 1 in 500,000 live births [1, 11, 12].
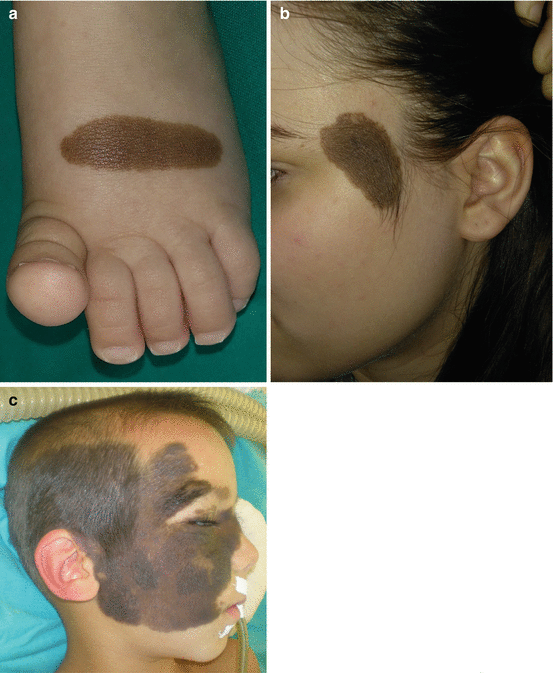
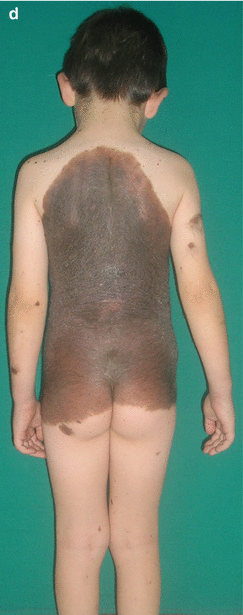


Fig. 10.2
Congenital melanocytic neavi: (a) small; (b) medium; (c) large; (d) giant congenital melanocytic nevus
CMN (small to medium sized) are usually present as round to oval pigmented lesions, with color that ranges from tan to black, with clear borders, and hypertrichosis [2, 3, 5]. Large CMN usually have irregular borders, multicolored pigment pattern, and rugose texture [1–5]; CMN undergo morphologic changes over time and become less or more pigmented, rugose, and verrucous and develop hypertrichosis [1, 3–6]. The evolution of CMN is especially intensive during first months of life [3, 6]. On dermatoscopy, CMN demonstrate reticular, globular, or reticuloglobular pattern [5]. Histologic findings of CMN include involvement by nevus cells of deep dermal appendages, neurovascular structures, deep dermis and subcutaneous fat, infiltration of nevus cells between the collagen bundles, and nevus cell-poor epidermal zone [5, 6]. The differential diagnosis for CMN includes AMN, epidermal nevus, nevus sebaceous, café au lait spot, and Mongolian spot [1, 5].
The exact risk for development of melanoma in CMN is not clear [5, 6]. The lifetime risk for melanoma arising in small CMN is between 0 and 5% (similar to AMN), and the risk for melanoma in large CMN is estimated to be between 5 and 10% [1, 3–6, 11, 13]. If there is CMN of 40–60 cm diameter in adult size, with a truncal location, numerous satellites nevi, and involvement of the leptomeninges, melanoma risk is high [2, 6]. The other features that indicate biopsy or excision include ulceration, uneven pigmentation, a change in shape, and nodularity [1]. Magnetic resonance imaging (MRI) screening of the central nervous system (CNS) early in life is recommended for the patients with high risk for malignant transformation [3, 5, 12].
Neurocutaneous melanosis (NCM) is characterized by an excess deposition of melanocytes along the leptomeninges [1–7]. Congenital nevus-like nevus (CNLN) or “tardive” becomes evident during infancy or early childhood with features indistinguishable from those of true CMN [3, 13]. These nevi measuring 1.5 cm or larger are found in 1–4% of older children and adults [3, 6].
10.2.1 Treatment of CMN
There are many different strategies for treatment of patients with CMN with an ultimate goal that the clear, deep margin of resection has to be achieved [1–3, 5, 6, 8–16]. Approximately half of the malignancies that have occurred in large CMN developed in the first 3–5 years of life, and therefore, decisions regarding prophylactic removal should be made early in life [3, 6, 11].
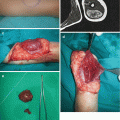
Treatment decisions should be tailored to individual patients, taking into account lesion size, location, appearance, and leptomeningeal involvement [2, 3]. One-stage excision with primary closure is the mainstay of surgical management of small CMN (single easily excisable and facial CMN are offered surgery for cosmetic reasons) (Fig. 10.3a–c) [3, 6, 11]. Routine prophylactic excision of small and medium CMN is not recommended [2, 3, 6].
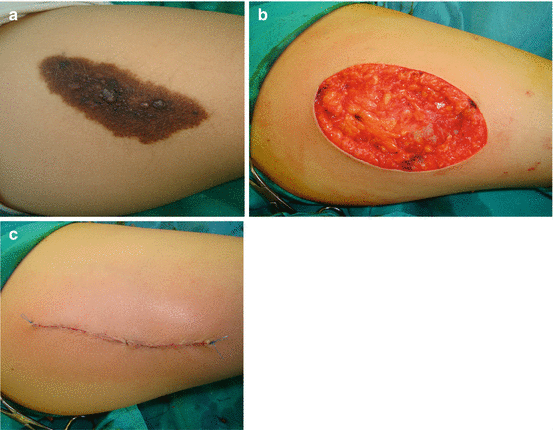

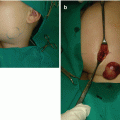
Fig. 10.3
Excision of congenital melanocytic nevus of the right femoral region: (a) preoperative view; (b) complete excision; (c) the result
Many strategies have been advocated for the removal and reconstruction of large and giant CMN such as serial excision, tissue expansion, and excision with skin grafting and skin substitutes, sole or in combination [1–6, 8–16]. The guiding principles are elimination (reduction) of the risk for malignant transformation, preservation of function, and improving cosmetics [1, 3, 6, 13].
The juvenile skin does not have the laxity of an adult, making local flaps difficult to use in children [5]. Staged excision down to fascia, with a flap reconstruction (advancement, transposition, or rotational), after tissue expansion of uninvolved skin, represents the primary surgical approach for removal of large CMN (Fig. 10.4a, b) [3, 5, 6]. The donor site must match with color, texture, and contour of recipient site, and it has to be free of infection or scars [5]. Tissue expansion works better in infant and young child; however, some locations are not suitable for this technique (it is associated with more morbidity and a higher failure rate in the extremities) [15].
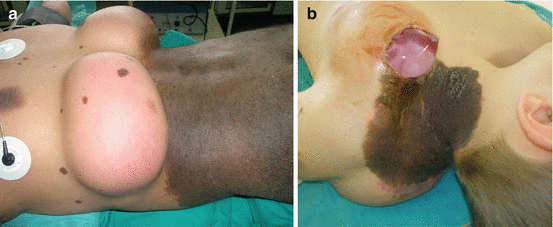

Fig. 10.4
Treatment of congenital melanocytic neavus with tissue expanders: (a) two expanders in lumbar region; (b) expander protrusion through wound dehiscence
CMN can be managed effectively by serial excision (carefully in sensitive areas like periorbital or perioral because of possible distortion of these structures) with only one scar as a final result, avoiding morbidity from skin grafting and complications following tissue expansion [5, 16].
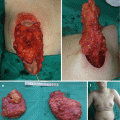
Excision and split-thickness skin graft (STSG) have usually poor aesthetic and functional outcome (back of the trunk is the only exception) (Fig. 10.5a–d) [3, 5, 16]. In the periorbital region and ear, full-thickness skin graft provide good match in both color and thickness for the recipient area [14]. Expanded full-thickness skin graft (FTSG) is an excellent choice for coverage of the dorsum of the hand and foot [5]. Other options for covering the wound after excision of giant CMN are autologous cultured skin substitutes (CSS) and artificial skin [11, 12].
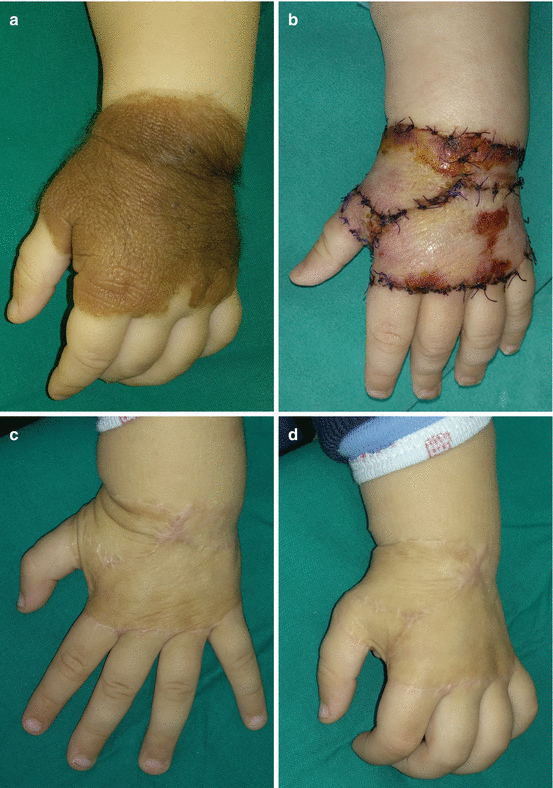

Fig. 10.5
Treatment of congenital melanocytic neavus with complete excision and placement of full-thickness skin grafts: (a) preoperative view; (b) skin grafts placement; (c, d) 6 months postoperatively
Techniques such as curettage, dermabrasion, and laser therapy are used when excision is not feasible, but in that case there is a problem with recurrence because only the epidermis and upper pole of dermis are removed and nevomelanocytes remain in the dermis [3, 6]. Laser treatment is mostly used for treatment of facial CMN that are not amenable for surgical excision [6].
The optimal choice of treatment of CMN varies by body region [5, 10, 14]. Tissue expansion as a single or serial procedure is treatment of choice for scalp region [10, 11]. At the face, subunit principle has to be followed [5, 10]. For periorbital region, full-thickness graft and expanded full-thickness graft are used, leaving the residual brow nevus [5, 14].
Tissue expansion is very effective for anterior trunk, but it should not be used in the region of breast in females until the growth is finished, and for giant CMN of the back, flanks, and abdomen, excision and split-thickness non-meshed graft give satisfactory result [5, 11].
For large and giant nevi on extremities, expanded local transposition flaps are used with best results [15]. Partial excision and skin grafting are proposed by some authors for the larger lesions distal to the knee or elbow, but after satisfactory early results pigmented cells “bleed” through the graft [5, 8, 15]. Artificial skin can also be used for covering of large full-thickness skin defects after excision of CMN [12].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree