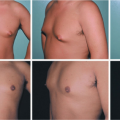
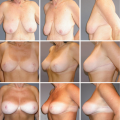
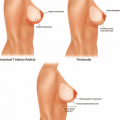
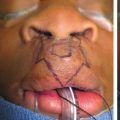
One-Stage Mastopexy with Breast Augmentation: A Review of 321 Patients
W. Grant Stevens, M.D.
Mark E. Freeman, M.D.
David A. Stoker, M.D.
Suzanne M. Quardt, M.D.
Robert Cohen, M.D.
Elliot M. Hirsch, M.D.
Marina del Rey and Los Angeles, Calif.
From Marina Plastic Surgery Associates and the Keck School of Medicine, University of Southern California.
Received for publication November 18, 2006; accepted February 27, 2007.
Copyright © 2007 by the American Society of Plastic Surgeons
DOI: 10.1097/01.prs.0000282726.29350.ba
Background: One-stage mastopexy with breast augmentation is an increasingly popular procedure among patients. In the past 9 years, there has been a 506 percent increase in mastopexy procedures alone. Although some recommend a staged mastopexy and breast augmentation, there are currently no large studies evaluating the safety and efficacy of a one-stage procedure.
Methods: A retrospective chart review was conducted of 321 consecutive patients who underwent one-stage mastopexy and breast augmentation. Data collected included the following: patient characteristics, implant information, operative technique, and postoperative results. Complication and revision rates were calculated to evaluate the safety and efficacy of the one-stage procedure.
Results: No severe complications were recorded over an average of 40 months’ follow-up. The most common complication was deflation of a saline implant (3.7 percent), followed by poor scarring (2.5 percent), recurrent ptosis (2.2 percent), and areola asymmetry (2.2 percent). Forty-seven patients (14.6 percent) underwent some form of revision surgery following the one-stage procedure. Thirty-five (10.9 percent) of these were for an implant-related issue, whereas 12 patients (3.7 percent) underwent a tissue-related revision. This 10.9 percent implant-related revision rate is less than a previously documented 13.2 percent 3-year reoperation rate for breast augmentation alone. The authors’ 3.7 percent tissue-related revision rate also compares favorably to an 8.6 percent revision surgery rate in patients who underwent mastopexy alone.
Conclusions: Although it has been stated that the risks of a one-stage procedure are more than additive, the results of our review suggest otherwise. Although a revision rate of 14.6 percent is significant, it is far from the 100 percent reoperation rate required for a staged procedure. (Plast. Reconstr. Surg. 120: 1674, 2007.)
Although one-stage mastopexy with breast augmentation has received an increasing amount of attention at plastic surgery meetings, there is a dearth of literature documenting outcomes of the procedure. The operation has been performed by surgeons for decades and was first described1–6 by Gonzalez-Ulloa and Regnault in the 1960s. With the increasing amount of breast surgery performed by plastic surgeons each year,1 the need for data regarding complication and revision rates has become more pressing. Several recent studies advocate the judicious use of the combined procedure,7–12 and others report that they commonly combine the procedures without adding additional risks.13 The goal of this study was to review our experience with a large number of combined, one-stage mastopexy and breast augmentation cases. The safety and efficacy of the procedure were determined by evaluating our long-term complication and revision rates.
Patients and Methods
A retrospective chart review of 321 consecutive one-stage breast augmentation with mastopexy procedures was performed. All cases were completed at a single outpatient facility by one of two surgeons (W.G.S. or D.A.S.) over a 14-year period (1992 to 2006). The average follow-up period was 40 months, with a range of 6 months to 13 years.
All patients had preoperative and postoperative photographs, general anesthesia, lower extremity sequential compression devices before induction
of anesthesia, and perioperative antibiotics. During the procedure, extensive undermining of mastopexy flaps was avoided when possible and no drains were used. Patients were intermittently ambulated the day of surgery and maintained on oral pain medication.
of anesthesia, and perioperative antibiotics. During the procedure, extensive undermining of mastopexy flaps was avoided when possible and no drains were used. Patients were intermittently ambulated the day of surgery and maintained on oral pain medication.
All study patients were candidates for both breast augmentation and mastopexy, as defined by having significant breast ptosis and hypoplasia. Degree of breast ptosis was determined using the Regnault classification,14 and any preoperative asymmetry was recorded. Each patient’s age, body mass index, smoking status, and type of mastopexy (inverted-T, vertical, circumareolar, or crescent) was recorded. Implant-related data such as fill (saline versus silicone), shape, texture, volume, and position (submuscular versus subglandular) were also collected. Procedure-related data such as operating surgeon, length of surgery, American Society of Anesthesiologists level, and concomitant procedures were noted.
Follow-up data including incidence of complications, treatment of complications, number of revision procedures, reason for revision, and patient or surgeon dissatisfaction were also recorded. Safety and efficacy were determined by measuring complication and revision rates, which were calculated retrospectively. To better understand their cause, complications were divided into two categories: implant-related and tissue-related complications. Tissue-related complications included breast and areola asymmetry, poor scarring, recurrent and persistent ptosis, loss of nipple sensation, pseudoptosis, infection, hematoma, and depigmentation of the areola. Implant-related complications included saline implant deflation, capsular contracture, implant malposition, and palpability. Statistical significance of the data was determined using chi-square analysis and Fisher’s exact test.
Results
A total of 321 patients underwent one-stage breast augmentation with mastopexy over the 14-year period. There were 25 unilateral and 296 bilateral cases, leading to 617 individual breast procedures. Of these patients, 118 patients had undergone some form of previous breast surgery and 203 were primary cases. The most common previous procedure was breast augmentation alone, which accounted for 79 of the 118 patients (67 percent). The majority of operations were for cosmetic concerns, whereas nine patients (3 percent) were undergoing reconstructive procedures.
The average age of women in our study was 39 years, the average body mass index was 22.7 m/kg,2 184 patients (57 percent) had delivered children, and of those, 161 (87 percent) had breast fed. Preoperative asymmetry was documented in 181 patients (56 percent), and 17 (5.3 percent) were diagnosed with tuberous breast deformity. Twenty-eight patients (8.7 percent) smoked cigarettes before surgery but agreed to stop smoking at least 2 weeks before the procedure. The average operation time was 128 minutes, which included 52 percent of patients having concomitant procedures.
Saline implants were used in 191 breasts (31 percent) and silicone implants were used in 426 (69 percent). Of the saline group, 13 patients received a Poly Implant prosthesis (Poly Implant, La Seyne-sur-Mer, France) or prefilled implants bilaterally. The average volume of implant was 317 cc for all types. The majority of implants were textured and round, with only five patients (2 percent) receiving anatomically shaped implants and 37 patients (12 percent) receiving smooth implants. Two hundred eighty patients (87 percent) had the implant placed in a submuscular pocket and 41 patients (13 percent) underwent subglandular placement. The distribution of the techniques for mastopexy was as follows: inverted-T, 60 percent; circumareolar, 21 percent; vertical, 15 percent; and crescent, 4 percent.
There were no incidences of death, myocardial infarction, pulmonary embolus, deep vein thrombosis, or major flap or nipple loss in any patients. The most significant complications were seen in four patients with submuscular implants who developed a postoperative infection, three of which required implant removal (0.9 percent) and subsequent revision. Saline implant deflation was the most common complication, occurring in 12 patients (3.7 percent) over 40 months, even though saline implants were used in only 191 breasts (31 percent). Complications were divided into implant-related versus tissue-related categories, and their distribution is listed in Tables 1 and 2.
Table 1. Implant-Related Complications | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Table 2. Tissue-Related Complications
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|
|---|