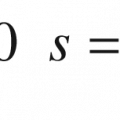div class=”ChapterContextInformation”>
6. Effects of Soap-Water Wash on Human Epidermal Penetration
Keywords
Skin decontaminationPercutaneous penetrationWash-in effectPesticideStratum corneumIntroduction
The superficial layer of skin, stratum corneum (SC), represents the first barrier against external toxicants [6]. Chemicals contaminants on skin surface can be absorbed into SC within minutes and remain there to form a “chemical reservoir ”, which plays a critical role in percutaneous delivery and skin decontamination. Earlier studies [5, 7, 8] demonstrated the positive correlation between chemical content in the SC reservoir determined by tape stripping at 30 min postdermal exposure and systemic absorption for up to 4 days in human and/or animals in vivo. This suggests the importance of removing hazardous chemical from SC reservoir. Wash-in effect , referring to enhancement of chemical percutaneous absorption caused by water-based skin decontamination strategies, was summarized by Moody and Maibach [12]. Overhydration of SC, effects of surfactant on skin barrier integrity, and friction occurring in the washing process were believed to be factors responsible for the accelerated release of chemical from SC reservoir to deeper layer of skin [4].
Moody et al. reported significant wash-in effects in several in vitro percutaneous penetration studies of pesticides , including 2,4-dichlorophenoxyacetic acid [14], 2,4-dichlorophenoxyacetic acid dimethylamine [13], dichlorodiphenyltrichloroethane [15] and N,N-diethyl-meta-toluamide [16] using rat, guinea pig, and human skin models. However, penetration enhancement was not observed for caffeine, benzoic acid, and malathion after a soap-water wash (5% soap solution) at 6 h postchemical exposure time [19]. Soap-water wash can reduce the chemical residue in SC reservoir, decrease skin penetration [19], and subsequently provide modest protection in model animals against toxic chemicals [2, 3, 11].
Here, to evaluate decontamination efficacy of soap-water wash , better understand the mechanisms of wash-in effect, and provide insights into improvements for skin decontamination, in vitro human epidermal penetration studies of four model chemicals, hydroquinone, benzoic acid, paraoxon, and clonidine, were conducted with flow-through diffusion cells. We selected these compounds as they have similar Log P values and molecular weights as common chemical warfare agents and pesticides, which are the key interests of this decontamination study. Also hydrophobic compounds with relatively low molecular weight have high penetration ability and low decontamination efficiency [20]; therefore, better understanding of wash-in effect of these compounds may help improve skin decontamination. Epidermis, instead of the commonly used dermatomed skin, was chosen to monitor the real-time penetration rate of chemicals through skin barrier. SC absorption rates of these chemicals at various SC hydration levels (0–295% of the dry sample weights) were determined and compared with the results of epidermal penetration studies to verify the correlation between SC hydration and wash-in effect.
Materials and Methods
Chemicals
[14C(U)]-hydroquinone (50 mCi mmol−1), [7-14C]-benzoic acid (55 mCi mmol−1), and [14C(U)]-paraoxon (77 mCi mmol−1) were synthesized by American Radiolabeled Chemicals, Inc. (St. Louis, MO), and [imidazoline ring 14C]-clonidine (55.77 mCi mmol−1) was from Perkin-Elmer Life Sciences, Inc. (Boston, MA). Reagent-grade ethanol was from Fisher Scientific (Pittsburg, PA). Soluene-350® tissue solubilizer and ULTIMA GOLDTM scintillation cocktails were manufactured by PerkinElmer Life and Analytical Sciences (Boston, MA). Nonradiolabeled hydroquinone, benzoic acid, paraoxon and clonidine as well as trypsin 1:250 for epidermis digestion were obtained from Sigma-Aldrich (St Louis, MO).
Dosing solutions of hydroquinone, benzoic acid, paraoxon, and clonidine were prepared in ethanol with 0.05 mCi ml−1 specific radioactivity and 20 mg ml−1 corresponding nonradiolabeled chemicals. Receptor fluid was 0.01 mole L−1 PBS aqueous solution containing 5% (v/v) polyethylene glycol and was prepared with phosphate-buffered saline (PBS) tablets obtained from Diagnostic BioSystems (Pleasanton, CA) and polyethylene glycol form Sigma-Aldrich (St Louis, MO).
Epidermal Absorption/Penetration Experiment
Adult human cadaver skin was obtained from the Pathology Department of the University of California, San Francisco. Skin was taken from the abdominal region of eight donors and dermatomed using a Padgett Electro-dermatome (Padgett Instruments, Inc., Kansas City, MO) with a target thickness of 400 μm, immediately sealed in aluminum foil, and stored at 4 °C until use. Prior to experiments, epidermis was separated from dermis by heating for 30 seconds in a 60 °C water bath and cut into circular sections to fit diffusion cells. Physical condition of skin samples was assayed visually to exclude any surface damage.
Circular epidermis samples were clamped onto a glass diffusion cell (Laboratory Glass Apparatus, Inc. Berkeley, CA) in a continuous flow-through diffusion cell system, with 1 cm2 surface area open to air. Circular metal nets were placed underneath skin samples to prevent epidermis from collapsing into diffusion cells. Receiving chambers (below the skin, 3 ml volume) were filled to capacity with receptor fluid stirred magnetically at ~600 rpm. Temperature of diffusion cells was maintained using a Lauda E100 Heating Circulator (Lauda Dr. R. Wobser Gmbh & Co. Kg, Germany) to achieve a skin surface temperature of 32 °C. Receptor fluid was pumped to the diffusion cell at a rate of 30 mL/h using a Pump Pro® MPL (Watson-Marlow, Inc., Wilmington, MA), and collected every 10 minutes using a Retriever Fraction Collector (Teledyne ISCO, Inc., Lincoln, NE) for 90 minutes. Surface of epidermis was dried with cotton pad and allowed to air dry for another 30 min before topical dosing.
A volume of 5 μl dosing solution was applied on skin surface. After 30 minutes, the dosed set was washed once with a small cotton ball soaked with 2% hand soap solution (Softsoap, New York, NY), twice with cotton balls soaked with distilled deionized water, and then dried with two dry cotton balls. Each wet cotton ball was placed on the dosed set for 3 minutes. No wiping was applied during the washing process. Sixty minutes after dosing, epidermis was removed from the diffusion cell and digested for 2 hours with 2 mL Soluene® 350. A volume of 15 mL scintillation cocktail was added to each collected sample and stabilized overnight before assayed.
Stratum Corneum Absorption Experiment
Human cadaver skin was prepared into SC membranes with modified method of Kuriharabergstrom, T et al. [9]. After immersing skin in 60 °C water for 1 min, epidermis was carefully peeled off from the dermis and placed on a cotton pad soaked with 0.5% (wt/wt) trypsin solution with SC side up for at least 15 hours at 36 °C. The pH of trypsin solution was adjusted between 8.0 and 8.6 with 5% (wt/wt) sodium bicarbonate. After digestion, SC was thoroughly rinsed with water three times and then dried under vacuum overnight at room temperature.
To control SC hydration levels, prepared SC membranes were equilibrated in humidity chambers containing saturated magnesium chloride and potassium nitrate solutions at room temperature for at least a week before use. The percentage relative humidity of humidity chambers was measured to be 28% and 90%, respectively. Dried SC sample were conditioned with Drierite desiccant (anhydrous calcium sulfate, mesh size: 8, Xenia, OH) in a vacuum oven at 50 °C under 18 mmHg of vacuum for a week. Over-hydrated samples were prepared by soaking in deionized water for an hour before use.
Water content of samples was measured by determining the difference between before and after drying weights; prepared SC membranes were accurately weighed and then dried with Drierite desiccant at 50 °C under 18 mmHg vacuum for a week. Weights of vacuum-dried samples were immediately measured after removal from the vacuum oven.
SC absorption was measured as per the modified procedure described previously [21]. Briefly, SC membranes were cut into 17-mm long squares for each dosing. After spreading 5 μL of dosing solution on the SC, scintillation glass vials containing SC samples were immediately capped to avoid change in the SC water content. After exposure (15 s, 5, 15, 30 and 60 min), SC membranes were washed by swirling for 10 s in 1 mL water thrice in different vials (additional wash was applied to certain samples to make certain the radioactivity of last wash was less than 1/10 of the radioactivity in SC sample). A 15 mL of scintillation cocktail was added to each collected fraction and left overnight before being measured for radioactivity in the liquid scintillation counter.
Radioactivity Measurement
Radioactivity measurements were performed in a Model 1500 Liquid Scintillation Counter (Packard Instrument Company, Downer Grove, IL, USA). 14C counting accuracy was audited by quenched and unquenched standards. Counting efficiency was ≥95%. Background control and test samples were each counted for 3 min for radioactivity.
Results
Epidermal Absorption/Penetration
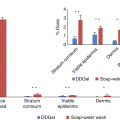
Epidermal absorption/penetration levels of model chemicals in ethanol as percentage applied dose
MWa (g mol−1) | Log Pb | Nonwash | Soap-water wash | P-valuee | |||
|---|---|---|---|---|---|---|---|
Hydroquinone | 110.11 | 0.59 | n = 4c | n = 8 | |||
─ | ─ | Decontamination | 83.0 ± 4.8 | ||||
Epidermis | 92.6 ± 0.8 | Epidermisd | 11.9 ± 0.8 | ||||
Receptor fluid | 1.4 ± 0.5 | Receptor fluid | 0.5 ± 0.2 | 0.006 | |||
Mass balance | 93.6 ± 0.9 | Mass balance | 95.5 ± 1.9 | ||||
Benzoic acid | 122.12 | 1.63 | n = 4 | n = 4 | |||
─ | ─ | Decontamination | 82.3 ± 7.1 | ||||
Epidermis | 92.9 ± 1.3 | Epidermis | 8.0 ± 5.1 | ||||
Receptor fluid | 3.9 ± 0.3 | Receptor fluid | 4.2 ± 2.0 | >0.05 | |||
Mass balance | 96.8 ± 1.2 | Mass balance | 94.5 ± 1.9 | ||||
Paraoxon | 275.2 | 1.98 | n = 5 | n = 5 | |||
─ | ─ | Decontamination | 82.4 ± 10.8 | ||||
Epidermis | 96.5 ± 2.0 | Epidermis | 9.8 ± 8.7 | ||||
Receptor fluid | 1.2 ± 0.2 | Receptor fluid | 3.1 ± 1.2 | 0.001 | |||
Mass balance | 97.5 ± 2.1 | Mass balance | 95.8 ± 1.0 | ||||
Clonidine | 230.09 | 2.49 | n = 8 | n = 8 | |||
─ | ─ | Decontamination | 89.0 ± 3.1 | ||||
Epidermis | 91.9 ± 3.8 | Epidermis | 9.2 ± 2.5 | ||||
Receptor fluid | 0.2 ± 0.1 | Receptor fluid | 0.2 ± 0.2 | >0.05 | |||
Mass balance | 92.3 ± 3.7 | Mass balance | 95.8 ± 1.3 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree