Key Points
- ▪
The axillary donor site has the highest reported incidence of lymphedema.
- ▪
Preoperative imaging facilitates surgical planning and establishes a baseline for postoperative comparison.
- ▪
Reverse lymphatic mapping identifies lymphatic drainage patterns, identifying lymph nodes for transfer while minimizing the risk of donor site lymphedema.
- ▪
Use of turbocharge, supercharge and reverse-flow anastomoses can ensure adequate blood supply to the lymph nodes and facilitate flap insetting in vessel-depleted recipient sites.
- ▪
Placement of lymph nodes into a prior surgical site requires removal of all scar tissue to allow lymphatic connections to occur between the flap and surrounding lymphatics.
- ▪
In patients with a history of recurrent lymphangitis, consideration should be given to an extended course of prophylactic postoperative antibiotics to avoid early infection and potential compromise the lymph node transfer.
Introduction
The unique aspect of the axillary donor site in vascularized lymph node transfer (VLNT) is the abundant soft tissue that can be harvested with the nodes, making it well suited for large recipient site defects. In addition, the relatively large and long thoracodorsal vessels facilitate flap insetting in vessel-depleted recipient sites. Challenges associated with this donor site include the proximity between lymph nodes draining the arm and those draining the trunk, and variable lymphatic drainage between these nodes that make donor site lymphedema a concern. Variability in the vascular supply to these nodes also makes correct pedicle selection important.
Concepts
Regional Anatomy
The primary goal during axillary lymph node harvest is to obtain an adequate number of vascularized lymph nodes for transfer while avoiding disruption of the lymphatics draining the arm that could result in lymphedema. The axilla may be conceptualized as containing five groups of lymph nodes: lateral, central, posterior, anterior and apical ( Figure 18.1 ). Beginning distally along the axillary vein, the lateral group drains the arm and continues into a central group that receives drainage from the back via a posterior group based on the thoracodorsal axis and from the chest via an anterior group based on the lateral thoracic axis. The central group then drains into the apical group of nodes located proximally along the axillary vein. These nodes drain superiorly into supraclavicular nodes. In practice, these groups are not clearly defined and there is cross-over drainage between groups that makes identification of drainage patterns based on anatomic landmarks alone unreliable. It is worth mentioning that there is a separate drainage pathway, named after Mascagni, which runs along the cephalic vein in the deltopectoral groove. Some lymphatics from this pathway may cross over the clavicle and drain directly into deep cervical lymph nodes, thus bypassing the axillary nodal groups.
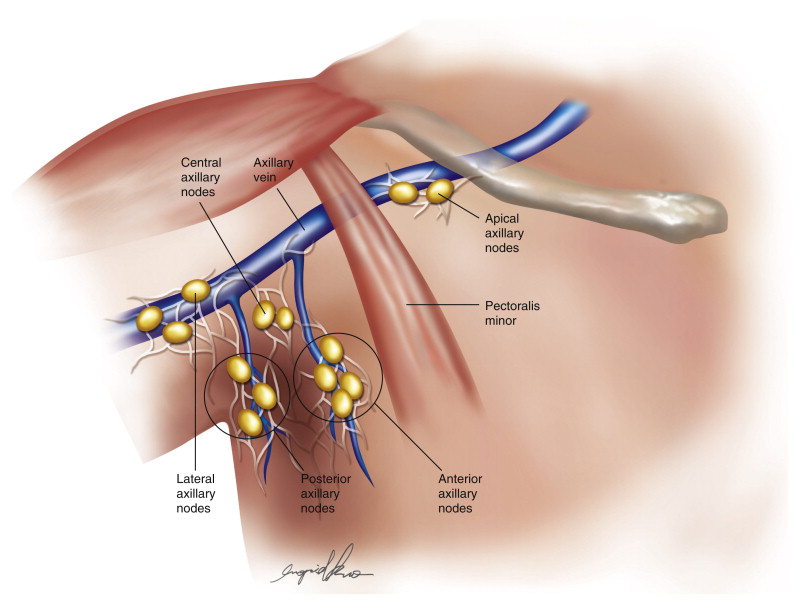
The anterior and posterior groups of lymph nodes are targeted for harvest in axillary VLNT. The blood supply to these nodes is not consistent due to variations in the vascular anatomy in the axilla. The lateral thoracic artery may arise from the thoracodorsal or subscapular artery in 29% of cases. The lateral thoracic vein typically arises as a separate branch from the axillary vein.
Flap Design
During vascularized lymph node harvest five key steps must be taken:
- 1.
identification of the nodes that are safe to harvest
- 2.
identification of the blood supply to these nodes
- 3.
preservation of nodes draining the arm
- 4.
dissection and preservation or division and repair of the thoracodorsal nerve
- 5.
inclusion of additional soft tissue as needed.
Considerations for flap design include the soft tissue requirements of the recipient site and the availability of recipient vessels. This is especially important in patients who have undergone major soft tissue resection and radiation, where removal of extensive scar and fibrotic tissue may result in a large defect with limited options for recipient vessels.
Incision planning is based on the soft tissue requirements of the recipient site. For a lymph node-only flap, preoperative imaging and the results of reverse lymphatic mapping (RLM) (discussed below) are reviewed; based on these findings the incision is made transversely over the nodes, typically 3–5 cm below the axillary crease and between the anterior and posterior axillary folds. A vertical incision may also be used for greater access, however, this leaves a more visible scar. For larger skin bearing flaps, the incision should be designed using a recipient site template configured to ensure proper pedicle location and soft tissue orientation for insetting.
Patient Selection
The axillary donor site is considered when the anticipated recipient site soft tissue deficit is larger than can be reconstructed using the groin or cervical donor sites. In addition, for patients with lower extremity edema, the groin donor site is usually avoided and the concealed axillary scar may be preferable to a cervical scar for some patients. Finally, certain patients may have limited alternative donor sites due to prior surgery or radiation. Axillary harvest is more challenging in the obese patient. It should be avoided in patients who have had prior axillary surgery.
Preoperative Considerations
Planning for lymph node transfer should include an accurate assessment of the etiology of the lymphedema, an evaluation of possible donor sites and optimization of the recipient site prior to surgery. Preoperative imaging is a helpful adjunct in surgical planning and provides a baseline for postoperative comparison.
Evaluation and Diagnosis
Patients may have primary or secondary lymphedema. ‘Secondary’ lymphedema resulting from a minor insult (e.g., a sprained ankle or arthroscopic procedure) should raise the possibility of an underlying primary lymphatic abnormality. The cause of lymphedema may affect the prognosis after surgery. Our experience has been that results are generally better in secondary lymphedema cases rather than primary cases. In primary lymphedema, there may be multiple extremities affected and grossly normal-appearing limbs may have subclinical lymphatic insufficiency. Therefore, preoperative donor site evaluation (including four-limb lymphoscintigraphy) needs to be performed in patients with primary lymphedema to minimize the risk of donor site lymphedema and to ensure that adequate lymph nodes are available for harvest.
Imaging
Magnetic resonance angiography (MRA) and lymphoscintigraphy have been the most useful studies for evaluating patients prior to surgery. Indocyanine green (ICG) lymphangiography can also demonstrate abnormal lymphatic flow.
MRA
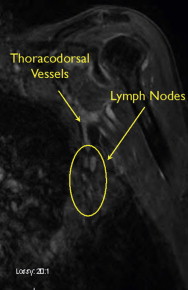
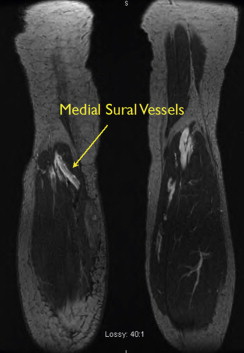
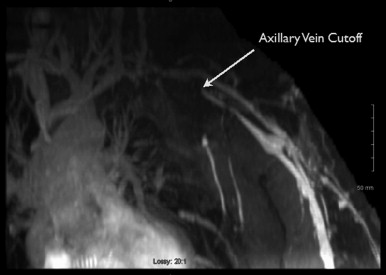
MRA is typically performed using gadofosveset trisodium (Ablavar®, Lantheus Medical Imaging, North Billerica, MA, USA). Gadofosveset trisodium stays in the blood pool longer, allowing high-resolution images of the lymph nodes and vasculature to be obtained. Both the donor site and recipient sites are imaged. Donor site imaging provides information on the vascular anatomy as well as the quantity and location of lymph nodes in the axilla ( Figure 18.2 ). MRA of the recipient site allows visualization of potential recipient vessels, evaluation of the fluid content in the limb and assessment of postsurgical changes such as venous outflow obstruction, deep vein thrombosis or venous insufficiency that may accompany postsurgical lymphedema ( Figures 18.3 and 18.4 ). MR lymphangiography is a developing technology that may provide additional information regarding lymphatic anatomy.
Lymphoscintigraphy
Lymphoscintigraphy confirms the diagnosis of lymphedema and provides a baseline assessment of the recipient site lymphatic function. Donor site lymphoscintigraphy may be indicated in patients with known or suspected primary lymphedema. Visualization of residual lymphatic transport in the recipient extremity may alter the location chosen for lymph node transfer in order to avoid injury to remaining functioning lymphatics.
ICG Lymphangiography
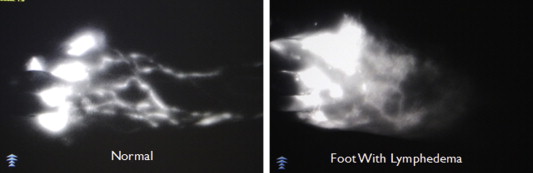
Similar to lymphoscintigraphy, ICG lymphangiography can also demonstrate abnormal lymphatic flow. It has the advantage of being able to be performed in the office and of allowing patients to see their lymphatic function in real time on the monitor. ( Figure 18.5 ). However it does not allow accurate assessment of the deep lymphatics or lymph nodes due to the limited depth visualized with fluorescence imaging.
Preoperative Optimization
Patients are encouraged to perform manual lymphatic drainage and compression in conjunction with a lymphedema therapist prior to surgery in order to optimize the recipient site for lymph node transfer. Minimizing the edema in the extremity facilitates would closure and healing.
Recipient Site Soft Tissue Assessment
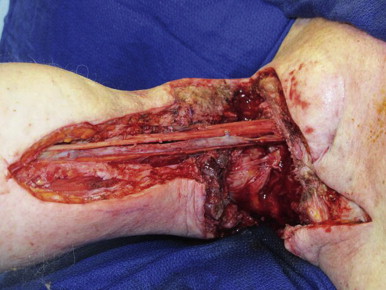
The recipient site is assessed to determine flap design. The axillary donor site is ideally suited for reconstructing postsurgical recipient sites. It offers lymph nodes, multiple pedicle configurations and abundant soft tissue that facilitate the repair of complex defects in the lymphedematous limb. Proper flap design includes assessing the recipient site for cutaneous and soft tissue deficits in addition to availability of recipient vessels and their location ( Figure 18.6 ). Strategies for pedicle selection and anastomotic configuration are discussed below under Surgical Techniques.
Surgical Techniques
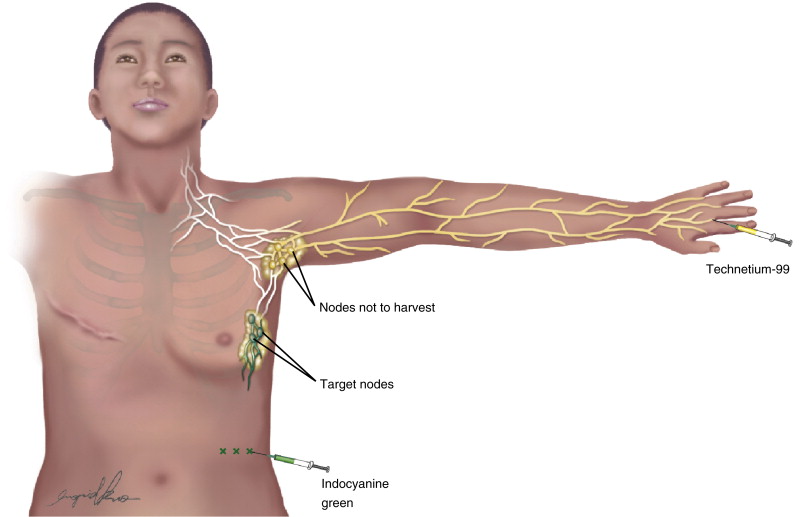
Reverse Lymphatic Mapping
![]() ( )
( )
RLM is a technique we developed to separately identify lymph nodes draining the arm and the chest. On the morning of surgery, the patient is injected with 0.2 cc of filtered Tc-99 in the first and second webspace of the hand of the donor extremity. Filtered technetium travels rapidly up the arm to the axillary lymph nodes, allowing identification of these nodes with a gamma probe during surgery. Once in the operating room, four or five ICG injections are performed along the lateral chest and back using 0.1 cc of ICG per injection site ( Figures 18.7 and 18.8 ). The injection sites are massaged and once the dye is taken up in the lymphatics their course and the lymph nodes that they drain to can thus be visualized using near-infrared fluorescence and the appropriate imaging camera. Because imaging is limited to a depth of 5–20 mm, it may be difficult to visualize the lymph nodes in patients with increased subcutaneous fat until after the incision is made and the dissection proceeds below the level of the clavipectoral fascia. Once the axilla is exposed the camera is brought in to visualize the desired nodes and then the gamma probe is used to ensure that the nodes do not have uptake of the Tc-99 that was injected in the hand. This mapping is performed repeatedly during the dissection until the nodes of interest are separated from the nodes draining the arm. After isolation of the nodes to be harvested, a relative 10-second count is taken and compared to 10-second count of the sentinel node of the arm. Typically, the count in the flap is 0–4% of the sentinel node count.