Key Points
- ▪
Lymphovenous bypass (LVB) usually require anastomosing lymphatic vessels less than 0.8 mm in diameter to small subdermal venules.
- ▪
A high resolution microscope, super-fine instruments and sutures, and the training of supermicrosurgery are the basic requirements for the success of this technique.
- ▪
Early-stage lymphedema, especially involving upper limb lymphedema, with functioning lymphatic vessels are the preferred candidates for lymphovenous bypass.
- ▪
Preoperative detection of the available lymphatic vessels with indocyanine green fluorescence lymphography is helpful for the LVB.
Introduction
Lymphedema is a chronic, debilitating condition that causes physical and psychological morbidity, affecting up to 250 million people worldwide. In the United States and other developed countries, cancer and its treatments are the most common causes of lymphedema. Lymphedema can evolve into progressive swelling, fibrosis, functional deficits, and chronic infections, thus adversely affecting quality of life and healthcare costs.
Unfortunately, no definitive treatment for lymphedema currently exists. The current mainstay of lymphedema management is complex decongestive therapy (CDT) and compressive garments. Although effective in many patients, CDT is time consuming, difficult to perform on a regular basis, expensive, and can lead to high rates of non-compliance and patient disatisfaction.
Surgical techniques can be described as either reductive or physiologic in their approach. Reductive methods, which include direct excision (Charles procedure) or circumferential liposuction, aim to debulk lymphedematous tissue. Physiologic methods, such as vascularized lymph node transfer or lymphovenous bypass (LVB), attempt to restore lymphatic drainage by reconstructing shunts for unimpeded flow of lymphatic fluid to the venous system. The advent of microsurgery, and more recently supermicrosurgery, has had a major impact on the evolution of these physiologic procedures, which have gained popularity to help reduce the severity of lymphedema.
The first reported experimental lymphaticovenous anastomosis was performed by Jacobson in 1962 in a canine model. Yamada first performed reported clinical application of lymphovenous bypass for obstructive lymphedema in 1967. Degni described end-to-side anastomoses between a saphenous vein and surrounding lymphatic vessels in the inguinal region. O’Brien and colleagues reported using LVB to treat lymphedema in the 1970s; since then, many others have refined the technique. However, LVB failed to gain significant popularity, partly because of criticisms that the lymphatic vessels are often unable to be found, and that once the venous pressure exceeds that of lymphatic pressure, this leads to backflow and the thrombosis of the bypass, resulting in only temporary improvement.
Koshima introduced ‘supermicrosurgical’ approach to LVB in 1996, where subdermal lymphatics located distally in the extremity are bypassed with adjacent subdermal venules less than 0.8 mm. This procedure is termed lymphaticovenular anastomosis (LVA). The rationale for his approach to LVA was that the distal lymphatics are less affected by lymphedema and thus more readily available for bypass, and also the venous pressure is lower in subdermal venules compared to larger veins and thus there is less venous backflow, leading to more permanent improvement.
The introduction of supermicrosurgical technique was a key development in reconstructive microsurgery since the 1980s. This technique allowed a successful fingertip replantation and contributed to development of perforator flaps. Koshima applied supermicrosurgical technique for lymphaticovenular anastomosis to manage lymphedema patients in the 1990s. This technique made possible the precise anastomosis of small vessels less than 0.8 mm. Developments of supermicrosurgical instruments and suture materials contributed to advances in microsurgical approach for the treatment of lymphedema.
Lymphedema is classified into primary lymphedema and secondary lymphedema. Most secondary lymphedema is related to malignant tumors and their treatment. Secondary lymphedema, following surgical and radiation treatment of women with breast cancer, accounts for 90% of upper extremity lymphedema patients. The risk factors significantly associated with breast cancer-related upper extremity lymphedema are radiotherapy and axillary node dissection. With the development of sentinel lymph node (SLN) biopsy for axillary staging, there has been reduced morbidity compared to axillary lymph node dissection. Upper limb lymphedema has been reported to develop in 3.0% of those who are undergoing SLN biopsy, as compared with 17.1% of those who undergoing traditional axillary node dissection. Other risk factors that appear to be related to upper limb lymphedema occurrence are location and size of the tumor, number of removed lymph nodes, radiation and obesity.
After lymph nodes are excised for the treatment of malignancy, related lymphatic channels in the distal side begin to dilate if the drainage is obstructed. Degeneration of the smooth muscle cells in the lymphatic channels occurs from the proximal site to the distal site. Consequently, the impairment of the contractile power and dysfunction of the valve in the lymphatic channels cause backflow of the lymph fluid. Excessive interstitial accumulation of lymph fluid may also lead to functional insufficiency of lymphatic channels. In such an environment, collagen fibers proliferate in the subcutaneous adipose tissue and dermis due to the activation of fibroblasts. Furthermore, recurrent cellulitis promotes further fibrosis and exacerbates the condition of lymphedema.
Concepts
Therapy of lymphedema is divided into conservative and surgical treatment. Although conservative treatment including lymphatic massage and compressive elastic garments have been advocated for first-line treatment of lymphedema, the long-term results of such decongestive therapy have not been uniformly encouraging. Discontinuation of conservative treatment usually results in a return to the former state.
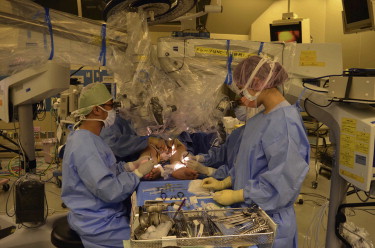
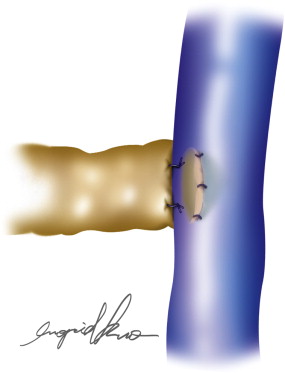
Lymphovenous bypass is a microsurgical approach that is designed to augment the rate of return of lymph to the blood circulation. The recent development of the supermicrosurgical technique involving the lymphatic duct less than 0.8 mm in diameter anastomosed to the subdermal venules has made LVB a potential surgical treatment for lymphedema. A high-resolution magnification microscope as well as super-fine microsurgical instruments and sutures are required for the successful performance of this technique. The practice of an animal LVB model in the lab is also important for surgeons to perform this technique.
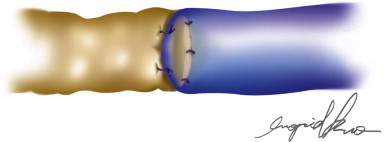
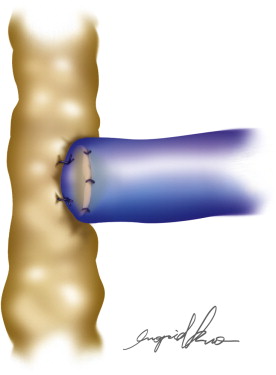
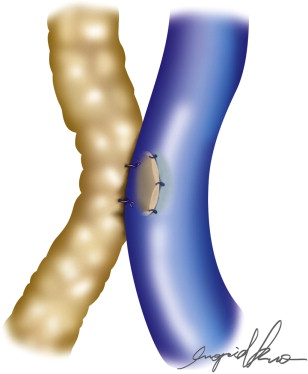
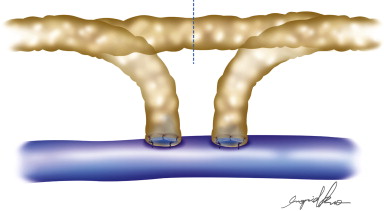
Further refinements in techniques have led to the development of end-to-end, side-to-side, and side-to-end anastomotic configurations ( Figures 20.1–20.5 ). One must consider various factors when choosing one technique as compared to another. Concomitant venous disease is sometimes present in these patients and assurance of valvular competence and proximal venous flow is an important determinant to the configuration. Similarly, considerations for the state of the lymphatic system, valvular competence, and bidirectional lymphatic flow will guide surgical decision-making. A side-to-end anastomosis may be considered if bidirectional lymphatic flow is present in the lymphatic duct and no venous backflow is present ( Figure 20.2 ). In this example, lymphatic system decompression may occur from either the proximal or distal side.
Patient Selection
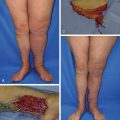
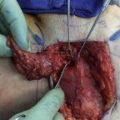
LVB has been reported to be used to treat lymphedema involving the upper and lower extremities as well as the trunk and scrotum. However, LVB has been proven to be most effective in patients with an earlier stage of lymphedema involving the upper extremity. It is possible to perform multiple LVBs with multiple surgical microscopes to increase the efficiency of anastomosis ( Figure 20. 6 ). However, combined surgical treatments with multiple LVBs and a functional lymph node transfer is recommended in severe cases which may not respond to bypass alone.