Key Points
- ▪
Lymph node transfer is still considered as experimental surgery.
- ▪
Lymph node transfer can be easily combined with routine microvascular breast reconstruction.
- ▪
Lymph node flap contains lymph nodes, lymphatic vessels and fat surrounding the superficial circumflex iliac artery or its perforator.
- ▪
The vessels and nodes draining the abdominal wall—which should not have direct connections to the lower limb—are included in the flap.
- ▪
Reverse nodal mapping with technetium and gamma probe can be helpful to exclude sentinel nodes draining the lower limb and to minimize the risk of donor site (lower limb) lymphedema.
- ▪
Axillary scar removal is an important step of the procedure.
- ▪
In this method, lymphatic vascular anastomoses are expected to form spontaneously.
- ▪
According to experimental studies, lymphatic vascular regrowth/maturation is a slow process. Thus, clinical results are seen with a delay (six months to two years).
Introduction
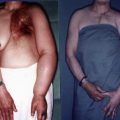
In industrialized countries, lymphedema is mainly encountered as a sequelae of the treatment of malignancy, particularly in women treated for breast cancer. The incidence of lymphedema varies from 9 to 41% in patients that have undergone radical axillary lymph node dissection. The incidence is significantly smaller after sentinel node biopsy, from 4 to 10%.
A large number of mastectomy patients also suffer from lymphedema following mastectomy, which is an independent risk factor for post breast cancer lymphedema. For these patients, combined autologous breast reconstruction and groin lymph node transfer may be an optimal choice.
Concepts
Regional Anatomy
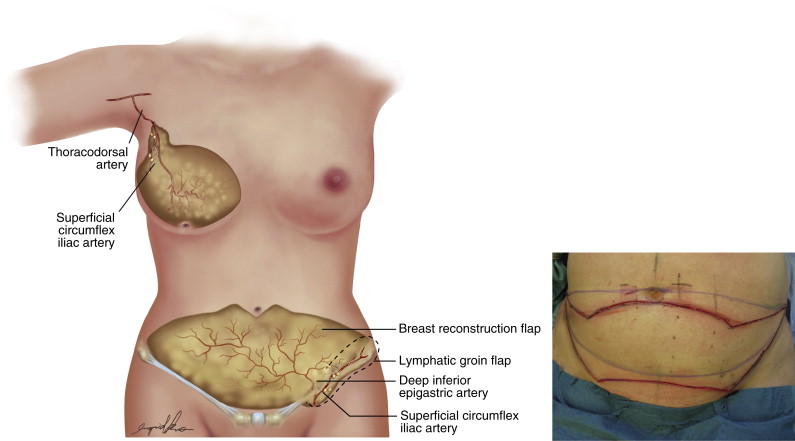
Soft tissue in the lower abdominal wall provides an optimal donor site for autologous breast reconstruction. The deep inferior epigastric perforator (DIEP), the muscle sparing transverse rectus abdomis musclocutaneous (msTRAM) and superficial inferior epigastric artery (SIEA) flaps are the most recognized free flap options for autologous breast reconstruction after breast cancer surgery. For postmastectomy lymphedema patients, the lower abdominal wall breast reconstruction flap can be easily combined with the inguinal lymph node (LN) flap. The LN flap contains lymph nodes, lymphatic vessels and fat from the groin area surrounding the superficial circumflex iliac vessels (SCIA) or its perforator (SCIP). The lymphatic vessels and nodes draining the abdominal wall—which should not have direct connections to the lower limb—are included in the flap with an abundant amount of surrounding fat tissue. Reverse nodal mapping with technetium and gamma probe can be helpful to exclude sentinel nodes draining the lower limb.
Flap Design
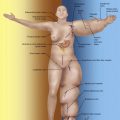
During the preoperative markings, we position our abdominal flap as caudally as possible to support the blood flow for the inguinal lymph node flap via the inferior epigastric vessel pedicle (see the preoperative picture in Figure 15.1 ). The selected abdominal flap method (DIEP, msTRAM, or SIEA) is based on the anatomy: location, size and number of the perforator vessels in the lower abdominal wall, and the surgeon’s preference. The breast reconstruction flap is usually based on the deep inferior epigastric vessel pedicle from the contralateral (to the reconstructed breast) inguinal area (see Figure 15.1 ), and can also be based on the ipsilateral side if needed. The LN flap contains lymph nodes, lymphatic vessels and fat from the groin area surrounding the SCIA or its perforator SCIP (see Figure 15.1 ). The LN-BR flap contains two blood vascular pedicles: the deep inferior epigastric artery/vein (DIEA/V) and the SCIA/vein. However, there is a large variation in the blood vascular anatomy in this area; in some patients, the SCIA and SIEA vessels merge before the level of femoral vessels.
Patient Selection
Lymph node transfer can be conveniently combined with traditional microvascular lower abdominal wall breast reconstruction. In our practice, all patients are previously examined by their oncologist and considered to be cancer free. All patients have received preoperatively information about the importance of weight control and conservative treatment methods: exercise, compression therapy and manual lymphatic drainage. We have used this method only in delayed breast reconstruction patients. We have operated on both mild and severe lymphedema cases, but we have not done prophylactic lymph node transfer surgery (meaning lymph node transfer surgery combined with the immediate breast reconstruction). Combined flap (LN-BR flap) for lymphatic reconstruction (LN flap) and free abdominal breast reconstruction (BR flap) is modified from the technique previously introduced by Dr Corinne Becker (see flap design in Figure 15.1 ).
Preoperative Considerations
In our practice, perforator vessels for both flaps (the inguinal lymph node flap and the abdominal flap) are preoperatively searched and marked using computed tomography angiography or ultrasound device. Using imaging allows us to better define any vascular anatomic variations of the surgical areas. A two-team approach is recommended enabling the simultaneous raising of the abdominal flap and preparation of the recipient vessels. Axillary thoracodorsal or circumflex scapulae, or internal mammary vessels can be used as recipient vessels, according to the surgeon’s preference. However, thoracodorsal vessels are primarily preferred, since it is important to remove all scar tissue from the axilla up to the level of the axillary vessels, and they are thus exposed. Removal of the scar is continued widely along the axillary vessels until normal fat tissue is reached. All fibrotic and avascular tissue surrounding the vessels, nerves and muscles is dissected and adhesions released. We consider wide scar removal as an important step of the procedure. Our own clinical observation is that there is often a dense fibrotic scar up to the level III lymph nodes, and its removal improves the axillary venous flow. The other benefit of using axillary thoracodorsal vessels as recipient vessels is that it is located close to the recipient area of the lymphatic flap, and thus a single blood vascular anastomosis (from the axillary vessels to inferior epigastric vessels) may be sufficient for both LN and BR flaps.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree