16
Melanoma
Karls Raimonds
Department of Infectology and Dermatology, Derma Clinic Riga, Dr. R. Karla klīnika, Riga Stradins University, Riga, Latvia
Introduction
Malignant melanoma is an aggressive, and in many cases therapy‐resistant, malignancy of melanocytes, creating a costly and significant clinical problem for any healthcare system. The function of melanocytes is best studied and defined in the skin. Within the basal layer of the epidermis, each melanocyte develops an intimate relationship with many surrounding keratinocytes, sending out slender dendritic processes that transfer packets of melanin pigment (melanosomes) to keratinocytes. The melanin granules orient in an umbrella‐like fashion over the keratinocyte nucleus parallel to the skin surface. This melanin is able to absorb ultraviolet radiation (UVR) and possesses potent antioxidant properties capable of neutralizing UVR‐generated free radicals. Paradoxically, melanocytes are injured and transformed by the same agent that melanocytes are programmed to defend against, UVR. Moreover, the cancers that melanocytes help prevent (squamous cell carcinoma [SCC] and basal cell carcinoma [BCC]) are less lethal than the cancer that melanocytes become (melanoma).
The type of melanin pigment (eumelanin vs pheomelanin), as well as the number, size, and density of melanosomes, determines skin pigmentation.
Melanoma is not an exception in tumor biology and, therefore, its origin predominantly results from the accumulation of mutations in genes crucial for regulation of cell proliferation, differentiation, and apoptosis, or other death pathways.
Melanoma is a phenotypically and molecularly heterogeneous disease: cutaneous, uveal, acral, and mucosal melanomas have different clinical courses, are associated with different mutational profiles, and possess distinct risk factors. The discovery of the molecular abnormalities underlying melanomas has led to the promising improvement of therapy, and further progress is expected in the near future.
Exposure to solar UV radiation, fair skin, dysplastic nevi syndrome, and a family history of melanoma are major well‐known risk factors for melanoma development. The interactions between genetic and environmental risk factors that promote melanomagenesis are currently the subject of ongoing worldwide research. Avoidance of UV radiation and surveillance of high‐risk patients have the potential to reduce the population burden of melanoma. Biopsies of the primary tumor and sampling of draining lymph nodes are required for optimal diagnosis and staging. Several clinically relevant pathologic subtypes have been identified and need to be recognized. Therapy for early disease is predominantly surgical, with a minor benefit noted with the use of adjuvant therapy. Management of systemic melanoma is a challenge because of a paucity of active treatment modalities.
Although more than 95% of tumors are found in the skin, melanoma is not exclusively a skin cancer. Sites of primary extracutaneous melanoma include ocular, mucosal, gastrointestinal, genitourinary, leptomeninges, and lymph nodes (if melanoma of unknown primary cancer). With the exception of neuroectodermally derived melanocytes that give rise to the retinal pigment epithelium, melanocytes originate from neural crest cells. During the early weeks of gestation, melanocyte precursors differentiate and migrate from the neural crest to numerous tissues (including the skin).
The current spectrum of malignant melanoma includes two clinical extremes. At one end of the spectrum, thin primary cutaneous melanoma is characterized by a relatively uniform treatment and a high cure rate. At the opposite end, metastatic melanoma is characterized by no proven effective therapy and poor outcomes.
The causal relationship of UV exposure to melanoma is complex, and acute, intense, intermittent sun exposure in youth equates to higher risk of melanoma. The risk of melanoma also increases with proximity to the equator. The intensity of UVR increases considerably at midday, around which time (10 a.m.–4 p.m.) sun avoidance is strongly encouraged. Fueled in part by public education campaigns, outdoor workers, stories of melanoma diagnoses among celebrities, and marketing efforts by the manufacturers of sunscreen and photoprotective clothing, melanoma awareness and sun avoidance behaviors have substantially increased. However, sunbed use, low sun protection factor sunscreens, and sun‐seeking behaviors remain popular among young people.
Epidemiology
While most cancers have shown both decreased incidence and mortality over the past several decades, the incidence of melanoma has continued to grow, and while mortality has recently stabilized in some countries, it is still growing in many countries. In recent decades, a heightened awareness of melanoma has led to an increased rate of diagnosis of early‐stage disease. The incidence of melanoma has been steadily increasing worldwide, resulting in an increasing public health problem.
The highest recorded incidence of melanoma worldwide is in Queensland, Australia, with an incidence equal to 55.8/105/annum for males and 41.1/105/annum for females. Reported incidence rates vary for Europe and are the highest in Switzerland and Scandinavian countries. In Europe, there is a north–south gradient in incidence rates with the highest rates in northern countries and the lowest ones in the southern countries. This is probably due to both the increased protection against UV exposure of the highly pigmented skin of southern Europeans and the different pattern of sun exposure (chronic in southern Europeans, intermittent in northern Europeans) [1]. The incidence of melanoma is increasing at a faster rate than any other preventable cancer in the United States [2]. Between 1992 and 2004, incidence annually increased by about 3.1%. Certain populations, such as men 60 years of age and lower socioeconomic status groups, face a greater burden from disease. While this increase may be attributed to higher rates of melanoma screening, the sharpest increase in incidence was evident among low socioeconomic status areas, where individuals are least likely to undergo screening, suggesting that increasing incidence rates are not simply an artifact of screening [3].
For any given stage and across all ages, men have shown worse melanoma survival rates than women, and low socioeconomic status groups have increased levels of mortality. Novel risk factors can help identify populations at greatest risk for melanoma and can aid in targeted early detection.
Trends in Melanoma Mortality
In fact, in many countries, melanoma was and still is one of only three cancers in men where mortality rates have increased [4]. Men have had poorer melanoma survival rates than women, with both genetic and behavioral factors proposed to explain these findings [5, 6]. More than 50% of all melanoma deaths are in white men 50 years of age and older [7]. The fact that lower education level and socioeconomic status are also associated with decreased survival are mentioned in several sources [8, 9].
Clinical Manifestation
The primary clinical and histologic subtypes of melanoma include in situ, superficial spreading, nodular melanoma, lentigo maligna, lentigo maligna melanoma (LMM), acral lentiginous, desmoplastic, and amelanotic (see Table 16.1). Melanoma staging is completed via clinical and histologic assessment using the American Joint Committee on Cancer (AJCC) TNM system.
Table 16.1 Melanoma clinical subtypes.
| Type of melanoma | % | Relation to sun exposure | Localization | Clinical aspects |
| In situ melanoma | Rare | Intermittent | Anywhere | Flat, in most cases regularly colored lesion |
| Superficial spreading melanoma | 70 | Intermittent | Back – man, legs – woman | Flat or slightly elevated irregularly lesion |
| Nodular melanoma | 5 | Intermittent | Trunk, limb. Anywhere | Nodule. Ulcerated polyp. Elevated plaque |
| Lentigo maligna | 4–15 | Long term | Cheeks and nose – woman, cheeks, forehead, ears, bald scalp – man | Flat, regularly or irregularly colored |
| Lentigo maligna melanoma | 4–15 | Long term | Cheeks and nose – woman, cheeks, forehead, ears, bald scalp – man | Flat and elevated regularly or irregularly colored |
| Acral lentiginous melanoma (glabrous skin) | 5 | No correlation with sun exposure | Glabrous skin of palms and soles | Flat, nodule, plaque |
| Acral lentiginous melanoma (subungual) | 5 | No correlation with sun exposure | Subungual | Irregular pigmented subungual line, no nail affection at early stage, destroyed nail in late stages |
| Amelanotic melanoma | 5 | Intermittent | Anywhere | Pink or red nodule. Ulcerated polyp. Elevated plaque |
| Desmoplastic melanoma | 2 | Long term | Head and neck | Mostly elevated papula or nodule |
| Melanoma in giant congenital nevus | Rare | No correlation with sun exposure | Trunk | Growing nodule |
| Melanoma arising from blue nevus | Rare | No correlation with sun exposure | Head | Recent history of enlargement or change in pre‐existing nevus |
| Melanoma of childhood | 0.4 | No correlation with sun exposure | Trunk | Mostly spitzoid appearance |
Tumor thickness in millimeters (Breslow depth) is the most important prognostic factor in the primary stage of melanoma. The tumor thickness classes were newly defined in the AJCC classification of (≤1.0 mm, 1.01–2.0 mm, 2.01–4.0 mm, >4 mm). In contrast to the previous classifications in the current classification besides tumor thickness and ulceration, the mitosis rate is included in primary melanomas of ≤1 mm (Figure 16.1).
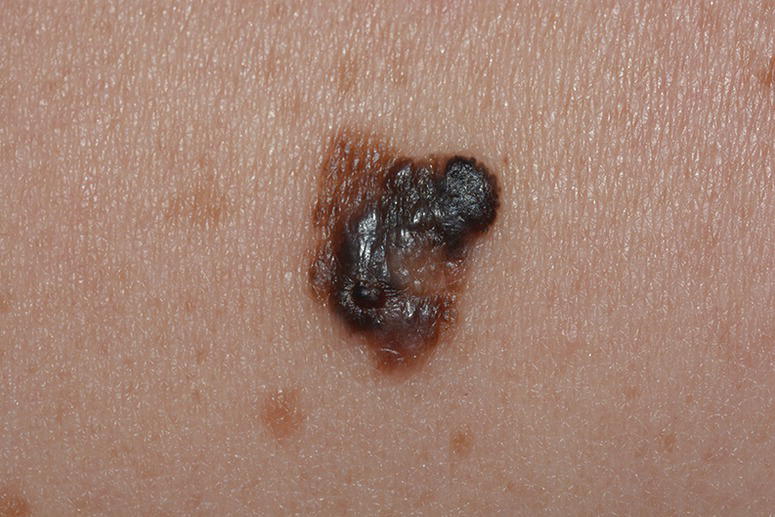
Figure 16.1 Superficial spreading melanoma with nodular part. History of the lesion up to two years. Slow radial growing part and nodular part with invasive growing. Superficial growing type as a rule shows positive ABCD melanoma signs. Histology: superficial spreading melanoma with invasive part, Breslow 1.4 mm, Clark III. Patient 56 y.o. Localization: right shoulder.
Fast‐Growing and Slow‐Growing Melanomas
The rapidity with which melanoma can invade the dermis probably depends on multiple factors, including the proliferative rate of the tumor cells, the degree of neoangiogenesis, and the ability of the surrounding tumor microenvironment to permit or impede the migration of malignant cells [10].
Liu et al. [11] and Lipsker [12] published data on melanoma’s different growing rates. Both authors point out that different types of melanomas exists in relation to their biological ability to grow and metastasize. Liu calculated that the thickness of one‐third of invasive melanomas are growing 0.5 mm or more per month. These rapidly growing melanomas are more likely thick tumors associated with high mitotic rate and more frequently found in older men with fewer melanocytic nevi and freckles. Furthermore, they usually lack the clinical airborne contact dermatitis (ABCD) features of melanoma, being frequently symmetric and amelanotic nodules.
Lipsker suggests a simple rule: each growing skin tumor that cannot be clearly diagnosed must be rapidly excised. This is the simplest rule to apply in daily practice so as not to miss those melanomas that are responsible for most of the deaths attributable to melanoma.
Another point of discussion is the existence of different forms of melanomas as outlined by Lipsker:
- Thin melanomas, slow‐growing, with a strong increase in incidence across time and associated with intermittent sun exposure, typical in persons with large number of nevi, and positive BRAF mutations.
- Thick melanomas, fast‐growing, with stable incidence and presumably not associated with sun exposure, a large number of nevi, BRAF mutations.
- Classic LMM, with a more slowly increasing incidence and associated with continuous sun exposure but not with large number of nevi and BRAF mutations.
This categorization seems very plausible from an epidemiologic and biologic point of view.
Three Roots of Melanoma
Development of new technologies such as in vivo reflectance‐mode confocal microscopy (RCM) allow doctors to compare findings with histopathological findings in case of nodular melanoma and superficial spreading melanoma. Also, stem cell research demonstrates clinical and epidemiological differences between melanoma subtypes. As was mentioned above, melanoma is a tumor of melanocytic origin, i.e. cutaneous melanomas arise from transformed epidermal melanocytes.
Researchers have identified stem cells of melanocytic lineage in the human hair follicle, the epidermis, and recently also in the dermis [13–15]. On the basis of the stem cell‐derived cancer model and the notable clinical, morphologic, and epidemiologic differences among various subtypes of melanoma, it was hypothesized that different melanomas arise from distinct types of cutaneous stem cells [16].
- Malignant cells initially proliferate along the basal layer of follicules (lentigo maligna). This proliferation could be with unpredictable continuation (half a year or even decades), and these malignant melanocytes may acquire the capacity not only for proliferation but also for vertical invasion into the dermis. Starting from this point melanoma in situ turns into invasive melanoma LMM.
- Malignant melanocytes could directly proceed to rapid vertical growth in the case of deep nodular melanoma with spontaneous mutations in these deeply seating polypotent melanocytes. There are a lot of lymphatic and vascular structures, and this type of melanoma always accompanied with very high risk for metastatic progression (Figure 16.2).
- Malignant melanocytes start to develop in the epidermis in the third type of melanoma. The length of time of the horizontal growing stage depends on different factors, but at the moment there is breakage on the basal membrane the early invasive stage of melanoma starts to develop.
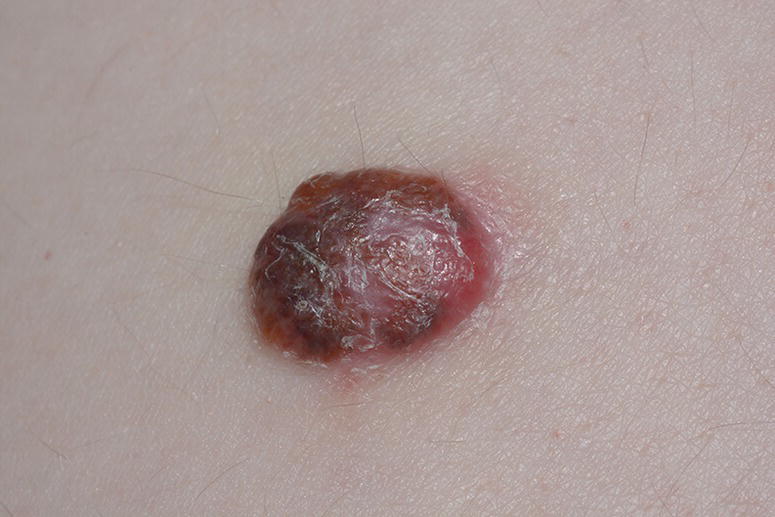
Figure 16.2 Nodular amelanotic melanoma, fast‐growing type. History of the lesion for four months. Breslow 2.2 mm, Clark III. Patient 67 y.o. Localization: dorsum, lumbal area.
To this end, stem cell research has opened up new and intriguing horizons that may enable better understanding of the development of cancer in general and melanoma in particular.
Anatomical Localization
The most common body sites for melanoma historically have included the trunk, head, and neck in men, and extremities in women [17]. Caini et al. [18] found that nevus count was associated with melanoma on typically nonexposed sites, such as the legs or trunk. In addition, skin and hair color correlate with body sites for melanoma, which may be attributed to genetic variability. Recent studies have found a left‐sided predominance for skin cancers, including melanoma, possibly related to poor ultraviolet‐filtering side windows in automobiles [19] (Figure 16.3).
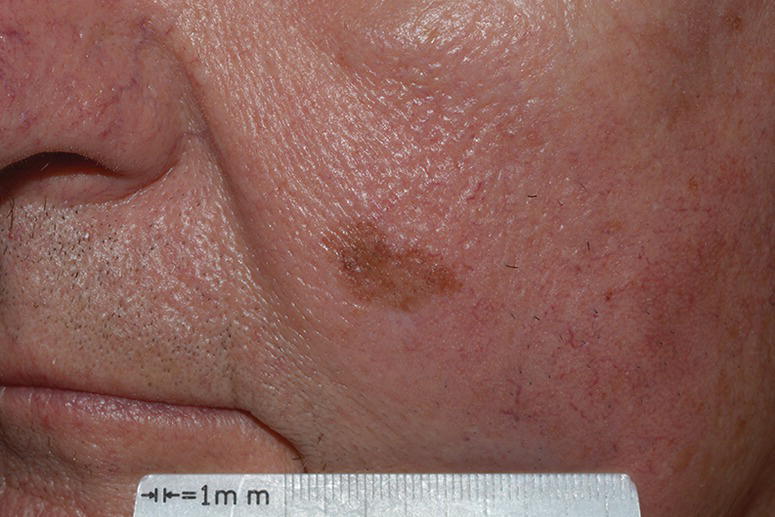
Figure 16.3 Lentigo maligna. Slow‐growing melanoma on sun‐exposed area. History of the lesion more than 12 years. Morphology: lentigo maligna (melanoma in situ). Male, 76 y.o. Case confirming left‐side predominance for skin cancers related to left side windows in automobiles.
Melanoma in Different Skin Colors
Since the 1960s, malignant melanoma incidence has increased in Caucasian populations, and consequently this neoplasm has become one of the most common cancers in fair‐skinned populations. In the United States, the incidence of melanoma among blacks and Hispanics is dramatically lower than that among whites. Melanoma is rare among deeply pigmented ethnic groups relative to individuals of Northern European decent.
Blacks
Black patients have a lower incidence of melanoma than whites, but the five‐year relative survival in many studies shows that blacks with melanoma have a much lower survival rate compared with 92.9% for whites. Moreover, the majority of melanomas in black patients are on nonexposed skin areas, such as the lower limb, hip, and trunk. These atypical locations may contribute to the lower survival for black patients [20].
The gap in survival is due to some difficulty in melanoma detection, as well as lack of attention from doctors and awareness by patients. Education level is the strongest predictive factor of skin cancer awareness, whereas cultural behaviors and personal experience of sunburns are the main factors determining sunlight avoidance.
Hispanics
For Hispanic patients, data from the registries shows that there is an increase per year in incidence of invasive melanomas, and there was a disproportionate increase in the number of tumors thicker than 1.5 mm. Other data shows there is correlation between Hispanic patients of lower socioeconomic status having a higher risk of thick tumors than Hispanic patients of high socioeconomic status, and patients with low social‐economic status had higher rates of the nodular melanoma subtype [21, 22].
Overall, the lifetime risk of developing melanoma is about 2.4% in Caucasians, 0.1% in Blacks, and 0.5% in Hispanics [23].
The incidence rate of this disease varies widely in relation to race. White populations have an approximately tenfold greater risk of developing cutaneous melanoma than black, Asian, or Hispanic populations. However, both white and African American populations have a similar risk of developing plantar melanoma, and noncutaneous melanomas (e.g. mucosal) are more common in nonwhite populations.
Risk Factors
Whiteman and Green [24] developed hypothetical tables for absolute risks for melanoma among white patients, combining information on environmental, phenotypic, and genotypic causal factors. The 10‐year risk of cutaneous melanoma appeared to be highest for older persons residing in southern Australia and the southern United States with ≥21 nevi on their arms.
Several factors have been hypothesized to be responsible for the worldwide increase in the incidence of melanoma. Of these, the major factors that are responsible for most of this increase are increased exposure to UVR. Exposure to UVR, namely UVA (315–400 nm) and UVB (280–315 nm), is a major risk factor for melanoma development. Cumulative UV radiation exposure from sunlight or tanning beds contributes to UV‐induced DNA damage, oxidative stress, and inflammation in the skin. The increase in incidence of melanoma is predominantly due to the depletion of the ozone layer, allowing penetration of UV rays into the atmosphere, but possibly also due to behavioral change (such as increased use of sun or tanning beds), and increased surveillance [25, 26].
Before launching an effective educational or early detection program, it is necessary to identify patients with risk factors for developing melanoma. Selective education and screening could be considered for individuals with environmental and host risk factors. The following characteristics increase the risk of melanoma: middle‐aged and older men, family members of melanoma patients, personal history of melanoma, nonmelanoma skin cancer patients, transplant patients, low socioeconomic status, many moles/atypical moles, fair skin, blue/green eyes, blonde/red hair. (See Table 16.2.)
Table 16.2 Risk factors for melanoma (summary).
| White race Adulthood (>14 yr) Number of nevi (higher than average) Large congenital nevi (>15 cm in diameter) Presence of dysplastic nevi Personal history of melanoma Familial history of melanoma (first‐degree members) Personal history of other skin cancer Skin phototype I/II Blistering sunburns in childhood Intense intermittent sun exposure Regular tanning bed before the age of 30 Chronic photochemotherapy Immune suppression Genetic disorders (e.g. xeroderma pigmentosum) |
Environmental Factors
Socioeconomic Status/Poor Health Access
It is well known that persons with higher education or income are more likely to be diagnosed with melanoma, but less is known about the disproportionate burden of mortality for low socioeconomic status individuals or for minority populations [27, 28]. Thicker melanomas (>1.5 mm) were also three times more common in patients with low levels of education or unemployment [29].
Sunlight
A large body of evidence supports the role of solar UVR exposure as the most important environmental risk factor for developing malignant melanoma. Multiple studies have shown that intermittent sun exposure, assessed indirectly by history of solar sunburns, appears to be a major determinant of risk for melanoma (in contrast to non‐melanoma skin cancers, which are linked more to cumulative sun exposure) [30].
The most important environmental risk factor for cutaneous melanoma is intermittent exposure to UV radiations, but nodular melanoma and acral lentiginous melanoma appear to have little relationship to sun exposure [31].
Solar radiations in the ultraviolet B range (80–320 nm) have been proposed as the critical component. This etiological link is further supported by the finding of higher melanoma incidence in populations that live in lower latitudes, higher incidences in white populations, lower incidences among dark‐skinned populations, and data from genetic and migration studies [32–34].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree