Fig. 11.1
Schematic depiction of melanogenesis in melanocytes. Tyrosinase (TYR), a key enzyme in melanogenesis, tyrosinase-related protein-1 (TRP-1), dopachrome tautomerase (DCT), and PMEL17 are melanogenesis-related proteins involved in pigmentation disorders, and microphthalmia transcription factor (MITF) plays a role in the transcriptional regulation of these factors. MITF expression levels are regulated by various transcription factors, including lymphoid enhancer-binding factor 1 (LEF-1)/T-cell factor (TCF), which is a downstream regulator of the Wnt-β-catenin signaling pathway, and cAMP responsive-element-binding protein (CREB). Melanocortin-1 receptor (MC1R)/α-melanocyte-stimulating hormone (α-MSH) signaling increases cAMP production and leads to the phosphorylation of CREB. Stem cell factor (SCF) and its receptor c-KIT link to the Ras-MAP kinase signaling pathway and regulate MITF function via MITF phosphorylation
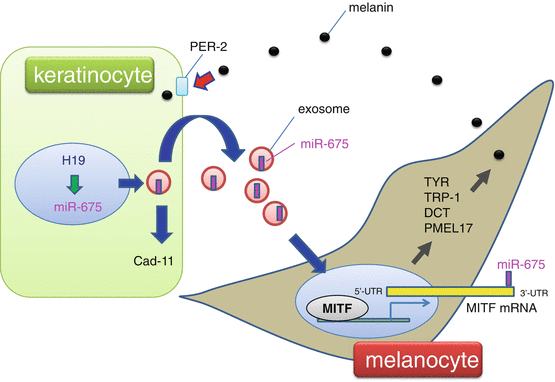
Fig. 11.2
Role of miR-675 in melasma. Downregulation of the H19 noncoding RNA was observed in the hyperpigmented skin of a melasma patient. H19 knockdown reduces miR-675 expression. miR-675 is released from keratinocytes via exosomes, a type of extracellular vesicle derived from the endosomal system, and inhibits MITF mRNA expression by targeting its 3′-UTR in melanocytes. Cadherin 11 (Cad-11) is another target of miR-675
Sunscreen for protection from sunlight and skin-lightening agents are used to treat melasma. Various well-known agents, such as hydroquinone, arbutin, kojic acid, and ascorbic acid, reduce pigmentation by interfering with several processes involved in melanogenesis 3′-UTR (e.g., the inhibition of melanocyte proliferation, TYR activity and expression, MITF expression, and 5’-UTR melanosome formation). These depigmenting agents have little effect and sometimes have severe side effects. Therefore, it is imperative to identify new agents with better efficacy and fewer side effects.
The oral antidiabetic drug metformin [11] and omeprazole [12], a gastric proton pump inhibitor, reduce melanin content by decreasing cAMP accumulation and TYR degradation, respectively.
We found that inhibitors of a disintegrin and metalloprotease (ADAM) reduce melanin content by disrupting the processing of the melanosomal protein PMEL17 [13]. PMEL17 (also called gp100 and silver) is a type I transmembrane melanosomal glycoprotein that forms a fibrillar matrix on which melanin is deposited in melanosomes. Proteolytic processing of PMEL17 is required for the formation of functional fibrils during melanogenesis. We demonstrated that ADAM inhibitors disrupt the formation of fibrils and their assembly into sheets in melanosomes via the regulation of PMEL17 processing. ADAMs are a family of proteases involved in ectodomain shedding; they play a role in various cellular processes [14, 15]. ADAM17 plays a critical role in the ectodomain shedding of many soluble proteins, including tumor necrosis factor-α (TNF-α), KIT ligand, and its receptor. In humans, ADAM17 is a candidate gene for the regulation of pigmentation in East Asians [16]. Mutations in ADAM10 were identified as a cause of reticulate acropigmentation of Kitamura, which is characterized by reticulate, slightly depressed pigmented macules mainly affecting the dorsa of the hands and feet [17]. As ADAMs are involved in multiple signaling pathways that regulate melanogenesis, ADAM proteases could be new potent therapeutic agents for pigmentation disorders.
Identifying the factors involved in the pathogenesis of melasma could facilitate the development of new treatment options for the disorder.
11.2 UV Exposure and Barrier Dysfunction
Based on a transcriptional analysis of melasma skin samples, 279 upregulated genes and 152 downregulated genes have been identified [8]. As expected, the expression levels of melanogenesis-associated factors, such as TYR, TRP-1, DCT, and MITF, are increased in melasma lesions, indicating higher melanogenic activity in lesional skin. As the number of melanocytes is not significantly increased, increased epidermal pigmentation due to increased melanogenesis in lesional melanocytes is expected to be the main physiological mechanism in melasma.
A bioinformatics analysis identified significant modifications of lipid metabolism in melasma. Lipid metabolism genes, such as peroxisome proliferator-activated receptor alpha (PPARA), PPAR gamma coactivator 1 alpha (PPARGC1A), arachidonate 15-lipoxygenase, type B (ALOX15B), and diacylglycerol O-acyltransferase 2-like 3 (DGAT2L3), were found to be downregulated. It is well known that lipid metabolism of the stratum corneum has an important role in barrier homeostasis. Interestingly, barrier function of melasma lesional skin is damaged compared with perilesional skin [8, 18]; a thinned stratum corneum and delayed barrier recovery rate have been observed for lesional skin.
UV irradiation may play a major role in melasma development based on the location of lesions and the development and/or aggravation of symptoms after sun exposure. Furthermore, chronic UV exposure is known to influence fatty acid metabolism and barrier function of the skin [19]. Free fatty acids and triglycerides in the epidermis of photoaged or acutely UV-irradiated human skin are significantly decreased [20]. The expression levels of genes related to lipid synthesis, including PPAR, are also markedly decreased [20]. Ligand activation of PPARs stimulates differentiation, induces lipid accumulation, and accelerates epidermal barrier regeneration in keratinocytes; accordingly, PPAR is an important regulator of lipid metabolism by mediating fatty acids [21]. Therefore, impaired barrier function owing to UV irradiation might be a factor in the pathogenesis of melasma.
11.3 Wnt Signaling
Although the effects of UV exposure are well established, the mechanism of UV-induced pigmentation may not be same for skin pigmentation conditions induced by different causes. In addition, UV irradiation may not be necessary in melasma development. Wnt inhibitory factor 1 (WIF-1) has been identified [9] as a factor involved in melasma pathogenesis that does not exhibit expression changes after UV irradiation. WIF-1 is a secreted antagonist of Wnt signaling; it inhibits the Wnt pathway by binding directly to Wnt ligands and preventing the ligands from binding to cell surface receptors.
The canonical and noncanonical Wnt pathways have important roles in the regulation of melanogenesis. In the canonical pathway, Wnts prevent the degradation of β-catenin, which promotes translocation to the nucleus to stimulate the transcription factor MITF. WIF-1 is reduced in melasma lesions and is expressed in both cultured normal human keratinocytes and fibroblasts, but not in melanocytes. WIF-1 knockdown stimulates TYR expression and melanosome transfer in keratinocyte/melanocyte cocultures. By treatment with recombinant WIF-1 in cocultures, TYR expression is significantly reduced with MITF expression. However, another report has shown that WIF-1 and other Wnt pathway modulators, such as Wnt5a, are upregulated in melasma lesional skin based on a microarray analysis [8]. Currently, decreased expression of WIF-1 is implicated in melasma development via the stimulation of melanogenesis and melanosome transfer through the activation of Wnt signaling.
11.4 Female Sex Hormones
As pregnancy and changes in uterine and ovarian hormones are risk factors for melasma, the female sex hormones estrogen and progesterone have been implicated in the development of hyperpigmentation in melasma [2]. Estrogens stimulate melanogenesis in human melanocytes by inducing the synthesis of melanogenic enzymes, and melanocytes express estrogen receptors [4]. An immunohistochemical analysis has shown that estrogen receptor and progesterone receptor expression levels are increased in affected skin [22, 23].
Recently, based on a microarray analysis, the upregulation of PDZ domain protein kidney 1 (PDZK1) expression was detected in the hyperpigmented skin of melasma patients [4]. PDZK1 is a member of the sodium–hydrogen exchanger regulatory factor (NHERF) family. NHERFs have PDZ domains, which mediate protein–protein interactions and have been shown to bind ion transporters. Estrogen increases MITF and TYR expression via PDZK1 in melanocytes. Knockdown of PDZK1 reduces estrogen-induced TYR expression. PDZK1 overexpression increases estrogen-stimulated TYR expression with ER-α and ER-β expression. PDZK1 also upregulates the expression of several ion transporters such as sodium–hydrogen exchanger (NHE), cystic fibrosis transmembrane conductance regulator (CFTR), and SLC26A3. These transporters are upregulated by estrogen, and specific inhibitors of transporter proteins inhibit the estrogen-induced expression of TYR. Interestingly, PDZK1 upregulation stimulates melanosome transfer to keratinocytes, irrespective of protease-activated receptor-2 (PER-2) expression, which is involved in melanosome transfer. PDZK1 has emerged as an important factor in the development of hyperpigmentation in melasma and is particularly associated with estrogen.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree