Lymphedema: Lymph Node Transfer
Adrian S. H. Ooi
David W. Chang
DEFINITION
The lymphatic system is responsible for reabsorption of excess interstitial fluid, protein, and waste products, as well as filtration and removal of foreign material from interstitial fluid and absorption of lipids from intestines. Up to 50% of the body’s circulating albumin is processed through the lymphatic system every day.
Lymphedema is an abnormal accumulation of protein-rich fluid in the interstitial compartment resulting in edema formation.
It occurs when the generalized or regional lymphatic load exceeds its transport capacity.
Long-standing lymphedema leads to chronic inflammation, adipose tissue hypertrophy and fibrosis, and eventually lymphatic channel fibrosis.
Lymphedema is a problem that affects about 200 million people worldwide.
Distribution: Lower limb 90% greater than upper limb 10% greater than genitalia 1%.
ANATOMY
Endothelial cells bud off from veins during early embryonic development and transdifferentiate to form the lymphatic vasculature.
The lymphatic primary plexus is composed of capillary-like vessels and is further remodeled to form pre-collecting, collecting, and ductal components. The major lymphatic ducts are formed by two paired lymph sacs (jugular and iliac) and two unpaired sacs (retroperitoneal and cisterna chyli).
Lymphatic channels are thin-walled vessels composed of endothelial cells connected by discontinuous junctions and surrounded by smooth muscle. The basement membranes of the larger, deeper lymphatic vessels have multiple intercellular gaps, which form valves through which fluid flows unidirectionally.
The superficial dermal lymphatics are unvalved and drain into the valved subfascial deep dermal system, which run with the superficial veins.
Lymphatic fluid is then transported through afferent lymphatic channels to lymph nodes, which are gathered in basins throughout the body. After undergoing immunological processes within the lymph nodes, the fluid flows out through efferent lymphatic channels and ultimately into the thoracic ducts and subclavian veins where they rejoin the venous circulation.
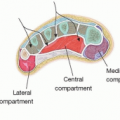
Lymph nodes are bean-shaped structures distributed along the lymphatic channels. They consist of a capsule, an outer cortex and an inner medulla. Multiple afferent lymphatics can enter via the outer cortex, and a lymph node is supplied by an artery and a vein at the hilar region, which accompany a single efferent lymphatic vessel. The lymph nodes contain lymphocytes and macrophages, which are responsible for removal of organisms, foreign substances, and tumor cells.
Lymph nodes are distributed deep in the body along the respiratory and gastrointestinal tract and superficial in the cervical, thoracic, and inguinal regions.
Lymphatic fluid transportation is under humoral influences and is propelled by a combination of:
Smooth muscle surrounding the collecting vessels that have an intrinsic autocontractile mechanism
Extrinsic compression by neighboring skeletal muscles
Negative intrathoracic pressure
PATHOGENESIS
Lymphatic homeostasis is a function of filtration of plasma across the capillary walls into the interstitial compartment and absorption by the lymphatic system. This rate of filtration is governed by the Starling equation whereby net filtration is determined by intra- and extracapillary hydrostatic and oncotic forces.
Lymphedema arises when the rate of filtration exceeds absorption by 2 to 4 L/d.
This disruption can occur at the level of the lymphatic vasculature or the lymph nodes and can arise due to a primary or secondary disease process.
True primary lymphedema can be classified as congenital (Milroy disease), pubertal/praecox (Meige disease), and tarda, which occurs in the adult age group. Lymphedema praecox is the most common form of primary lymphedema.
Lymphedema can also arise secondary to acquired damage to the lymphatic system. These include infection, trauma, malignancy, surgery, and radiation.
The most common cause worldwide is filariasis, caused by the parasite Wuchereria bancrofti. In the developed world, it is most commonly due to treatment for cancer.
For breast cancer treatment, it occurs in up to 40% of cases with lymph node dissection and radiation and 5% of cases with sentinel lymph node biopsy.
41% after gynecological cancer treatment.
12% to 55% after groin lymph node dissection.
NATURAL HISTORY
When there is disruption in the lymphatic system and as lymphatic pressure builds, flow within the lymph vessels stagnates, leading to valvular incompetence and dermal backflow.
Lymphedema is confined to the subcutaneous compartment as the deeper lymphatics are driven by the skeletal muscle pump.
Over time, macromolecular protein and hyaluronan are deposited in the interstitium leading to increased tissue colloid osmotic pressure.
Fibroblast, monocytes, adipocytes, and keratinocytes also increase within the tissue, leading to increased collagen deposition, degeneration of elastic fibers, and fibrovascular proliferation.
The basement membranes of lymphatic vessels are obliterated and the vessels become fibrotic.
This combination results in nonpitting edema, and eventually a chronic inflammatory state ensues.
The cytokines arising from the inflammation and the disruption in lymphatic flow of lipids leads to adipose deposition and hypertrophy in the interstitium.
The verrucous appearance of long-standing lymphatic extremities is caused by hyperplasia of skin at the dermalepidermal junction.
Lymphatic stasis and decreased immunological function predispose the patient to an increased risk of local skin infections, erysipelas, and later, lymphangitis. The excess tissue can lead to symptoms of pain, heaviness, and decreased mobility. Long-standing chronic lymphedema can lead to development of lymphangiosarcoma (Stewart-Treves syndrome). Lymphedema has significant adverse impact on patients’ quality of life and psychosocial health.
The International Society of Lymphangology (ISL) has classified lymphedema into three stages:
0: Latent/subclinical period
I: Pitting edema which is reversible on elevation
II: Tissue fibrosis and edema, which is nonreversible on elevation
III: Elephantiasis and trophic skin changes
PATIENT HISTORY AND PHYSICAL FINDINGS
The history is targeted at determining the diagnosis and etiology of lymphedema, duration, symptoms, and complications, and how the patient has been managing it.
Elucidating diagnosis and etiology include asking about risk factors for malignancy, travel history, prior surgical procedures, history of radiation therapy, trauma and infection, and family history. Obesity can contribute to lymphedema through the overload of lipids in the body. Other causes of extremity swelling such as congestive cardiac failure, venous insufficiency, and deep vein thrombosis have to be excluded (see differential diagnosis).
Important symptoms and complications to ask about include pain and infections in the affected extremity, a feeling of heaviness and difficulty in movement.
The examination of the patient with lymphedema should include a general physical and a targeted examination of the affected part. The general examination is aimed at excluding any active infection or malignancy and general causes of extremity edema.
Lymphedema is typically unilateral, with the swelling beginning distally and progressing over months to years.
The edema starts off soft and pitting and eventually becomes nonpitting as interstitial fibrosis sets in.
There is minimal pigment change or ulceration and peau d’orange indicating dermal fibrosis can be present.
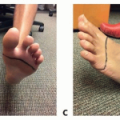
Late changes in the extremity include hyperkeratosis, papillomatosis, and the development of a positive Stemmer sign in which the skin on the dorsum of the second web space of the foot cannot be pinched as a fold.
Reproducible measurements of the limb should be taken to help with accurate follow-up. These can include circumference measurements and volumetric measurements such as water displacement tests, perometry, and bioimpedance testing. Digital photographs are also useful for comparative analysis.
Circumference measurements are typically taken at 4- to 10-cm intervals from consistent landmarks such as the patella in the lower extremity and the olecranon in the upper extremity. Figures that are used in the diagnosis of lymphedema include the following:
Greater than 2 cm difference from the nonaffected limb.
Severity can be graded as mild (greater than 10% difference), moderate (greater than 20%), and severe (greater than 30%).
Water displacement has been the standard for volume measurement and is highly accurate but cumbersome. Lymphedema is diagnosed if the volume differential of the affected limb is greater than 10% of the normal limb.
Perometry utilizes an infrared optical electronic scanner to calculate volume.
Bioimpedance testing calculates the fluid composition of an affected limb using resistance to an electrical current.
IMAGING
Lymphangiography is the visualization of lymphatic vessels using contrast medium. Vessels can be classified as anaplastic, hypoplastic, or hyperplastic. There are the direct and indirect forms of lymphangiography.
Direct lymphangiography involves transecting distal lymphatic vessels and directly injecting water-soluble iodide contrast medium into them. This has largely been abandoned because of patient discomfort and damage to the lymphatic channels.
Indirect lymphangiography injects contrast medium distally into the subepidermis and uses radiographic imaging to delineate the lymphatic system.
Isotopic lymphoscintigraphy is the current standard for lymphatic imaging. It injects Technetium-99-labeled albumin into the subepidermis of the distal affected extremity and timed post injection imaging to determine lymphatic anatomy and function. It has a sensitivity of 97% and specificity of 100%. However, when done awake, the injection can be painful, and the length of time required can be distressing to the patient.
The injection of indocyanine green (ICG) and imaging using an infrared camera have become widely used in the imaging of lymphatic vessels and are particularly useful intra-op when needing to delineate the superficial lymphatic system. A classification based on the appearance of the ICG has been developed and correlated with the Campisi classification (Table 1).1
Computed tomography (CT) and magnetic resonance imaging (MRI) scanning are useful in determining the architecture of the affected limb and elucidating any underlying
cause such as neoplasm. There is commonly a “honeycomb” appearance above the deep subcutaneous fascia. MRI with contrast has proven promising in outlining lymphatic vasculature.2
Table 1 Indocyanine Green Appearance to Classify Severity of Lymphedema | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
DIFFERENTIAL DIAGNOSIS
Venous insufficiency
Deep venous thrombosis
Vascular malformations
Lipedema
Myxedema
Fluid overload
Congestive cardiac failure
Renal failure
Liver failure
Hypoalbuminemia
Soft tissue tumors
Morbid Obesity
NONOPERATIVE MANAGEMENT
Nonoperative management is aimed at reducing the amount of swelling and treating any resulting complications. The former is accomplished by compression and massage therapy, whereas the latter involves adequate skin care and early detection of infection and treatment with the appropriate antibiotics. In a compliant patient, this helps to slow down the progress of lymphedema.
Patients should be educated about lymphedema and its complications, and given responsibility for their own care.
Conservative treatment of lymphedema has been well researched and the regimen described is complete decongestive therapy (CDT). This should be started early to have the most benefit and is useful in cases of lesser than stage III lymphedema. Analyses have shown up to 40% to 60% reduction in lymphedema volume. It consists of two phases:
Initial reductive phase (up to 8 weeks) consisting of:
Manual lymphatic drainage—Circular tissue stretching massage with varying degrees of pressure
Compression therapy during waking hours
Regular skin care
Targeted exercise
Maintenance phase consisting of
Self-conducted lymphatic drainage
Continuation of compression garments
Weight loss may help with lymphedema by reducing the flow of fatty acids through lymphatic channels and decreasing fatty deposition in the subcutaneous tissue.
Exercises such as swimming can be useful in reducing the severity of lymphedema.
Unsubstantiated nonoperative measures include coumarin (5,6 benzo-a-pyrone), diuretic use, microwave heating, and intra-arterial injection of lymphocytes.
SURGICAL MANAGEMENT
Although significant advances have been made in the surgical treatment of lymphedema, no surgical technique offers a cure, and an aggressive trial of nonsurgical therapy should first be done.
Indications for surgical treatment include failed conservative therapy, grossly impaired limb function, debilitating extremity size and weight, recurrent lymphangitis (greater than 3 episodes of infection per year), and uncontrolled pain.
Surgical techniques for the treatment of lymphedema are classified as excisional and physiological.
Excisional techniques aim to reduce the amount of lymphatic tissue and tighten the skin envelope. They are described elsewhere in this book and include but are not limited to excision and skin grafting (eg, Charles procedure), staged excision, liposuction, and, more recently, radical resection with preservation of perforators (RRPP).3 Unfortunately, as the primary disease process is not addressed, there is a high risk of recurrence with these techniques, and the appearance can be undesirable.
Physiological techniques attempt to replicate the normal lymphatic flow in the affected extremity. Procedures that have been developed and described elsewhere include buried dermal flaps, lymphovenolymphatic bypass, lymphatic bypass, and lymphovenous anastomosis (LVA).4
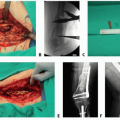
The vascularized lymph node transfer (VLNT) is a physiological technique that transfers intact autologous lymph nodes from a nonlymphedematous region of the body to the affected extremity. In a sense, this is the most physiological method of addressing the problem in a patient whose lymphedema is due to damage to or paucity of lymph nodes in a particular basin.
The lymph nodes are microsurgically anastomosed to the chosen recipient site vessels and allowed to develop collateral blood supply. Over time, reported mechanisms of lymphedema reduction include reduction of scarring at the recipient site, hydrostatic “pumping” of intraflap lymph via arterial inflow and venous outflow, and lymphangiogenesis aided by the production of VEGF-C by the transplanted lymph nodes.
Though not curative, physiological techniques have proven to alleviate symptoms and reduce lymphedema volume.
Preoperative Planning
Preoperative workup should include elucidating etiology of the lymphedema, comorbidities, the patient’s treatment to date, limb measurements, weight fluctuations, and ability to comply with therapy.
Severity of lymphedema should be clinically staged according to the ISL grading system, and treatment planned accordingly:
Patients with mild to moderate stage 1 or 2 lymphedema are best candidates for physiologic procedures such as VLNT and/or LVA.
Patients with stage 3 fibrofatty lymphedema or elephantiasis may benefit from excisional procedures prior to physiologic procedures in order to offset the lymphatic load and optimize the result of physiologic procedures.
Specific history to note with regard to vascularized lymph node (VLN) harvest includes previous surgery in the region of planned harvest or pre-existing lymphedema if harvest is planned from the groin. Scars and surface anatomical landmarks should be noted. Active infection in the affected extremity should be ruled out.
Definitive diagnosis of lymphedema is made with lymphoscintigraphy, which evaluates lymphatic function.
ICG imaging of the affected extremity prior to surgery aids in lymphatic mapping for the simultaneous performance of LVA.
If groin VLNT is planned, reverse lymphatic mapping should be performed to determine the crucial deep groin lymph node basin of the lower extremity, which should be avoided in groin VLNT harvest.
This involves the preoperative injection of technitium-99-labeled albumin subcutaneously into the inner thigh and a gamma probe used intraoperatively to detect the sentinel lymph node of the deep groin lymph node basin.5
Harvest of this lymph node should be avoided at all costs to prevent postoperative lower extremity lymphedema.
Positioning
For most VLN harvest, the patient is placed in a supine position.
For TAP flap or split LD flap with LNT, the patient is placed in lateral decubitus position.
If the lymph nodes are intended to be placed in the axilla, the arm is extended at right angles to the torso to access and prepare the recipient site.
For specific points for each source of VLN, refer to the subheadings below.
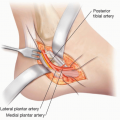
Approach
For harvest of VLN, the common sources are the submental, supraclavicular, thoracic, inguinal, and omental lymph nodes. The advantages, disadvantages, and potential complications of each are listed in Table 2.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree