and Emir Q. Haxhija2
(1)
Department of Plastic Surgery and Burns, Institute for Mother and Child Health Care of Republic Serbia, University of Belgrade, New Belgrade, Serbia
(2)
Department of Pediatric and Adolescent Surgery, Medical University Graz, Graz, Austria
Keywords
Lymphatic malformationChildrenTreatment14.1 Introduction
Vascular anomalies are divided based on endothelial characteristics in vascular tumors (mostly hemangiomas) and vascular malformation [1–3]. On the base of type of the vessel included in vascular malformation, they are divided mainly into simple (capillary malformation (CM), lymphatic malformation (LM), venous malformation (VM), and arterial malformation (AM)), and combined vascular malformation—two or more vascular malformations in one lesion, malformation of major named vessels, and malformation associated with other anomalies [1–3]. Vascular anomalies are also divided into slow-flow (CM, LM, VM) and fast-flow (AM) vascular malformation [2, 3].
14.2 Clinical Features
The lymphatic system develops during the sixth week of embryonic life [3, 4]. By the first theory, the lymphatic system is derived from the primordial endothelial buds sprouting from the developing venous system, the second theory is the theory of “centripetal” development, and the third one is the combination of these two theories [3–7].
The exact etiology of LM is unknown [4]. There are several theories for etiology of LM: failing of aberrant primordial endothelial buds to reestablish communication with venous system from which it arose, part of lymphatic channels becomes “pinched off” from the main lymphatic system, and abnormal budding of the lymphatic system with a loss of connection to the central lymph channels [3, 4]. No genetic basis has been determined for LMs [3]. LMs (traditionally called lymphangiomas) are benign vascular lesion presented as localized areas of abnormal development of the lymphatic system [3, 6, 7]. If LM is relatively large, it can be diagnosed by ultrasound (US) prenatally by the second trimester [3, 4]. LMs are in approximately 50% of cases presented at birth and near 90% within the first 2 years of life [3, 4, 7–10]. The reported incidence of LMs varies from 0.14 to 5% of all benign soft tissue tumors, with both genders equally affected [4, 8, 10]. In up to 75% of the cases, they are localized in the head and neck region, followed by the axilla, chest, buttock, perineum, and retroperitoneum/mediastinum [3, 4, 7, 8, 10–12]. Depending on cyst diameter (there is no clear definition of the size for the lesion), LMs are divided into macrocystic, microcystic, and combined [2–5, 8, 12, 13].
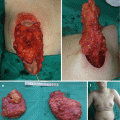
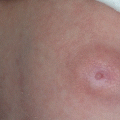
Disease location, size, and depth have a major influence on prognosis [3–5, 8–13]. LM lesions are visible, nontender, soft, and compressible swellings [3, 10, 11]. Cutaneous or mucous lesions usually present as liquid-filled vesicles (blood or purulent fluid) (Fig. 14.1) [3, 8]. Deep-seated LMs can be divided into diffuse edema (microcystic) and localized multilocular cyst (macrocystic) [8].
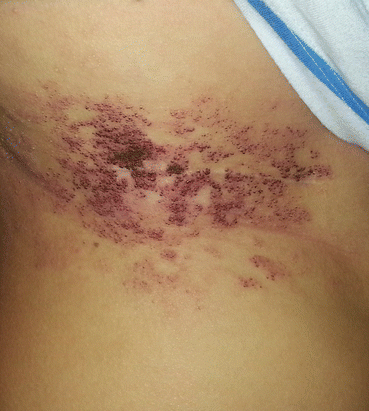

Fig. 14.1
Superficial lymphatic malformation of the abdominal wall
14.3 Complication (Symptoms) of LMs
The two most common complications associated with LM are bleeding and infection, and they result in rapid enlargement of the LM [3, 8, 10]. Intralesional bleeding occurs in up to 35% of lesions causing ecchymotic discoloration, pain, or swelling and compression of adjacent organs [3, 4]. Depending on the anatomic position of the LM, this compression can lead to acute visual disturbances, pain, headaches, respiratory distress, and dysphagia [4, 14, 15]. Infection occurs as a complication in nearly 70% of lesions [3]. Cutaneous vesicles can be associated with lymph and blood leakage [3, 4, 8].
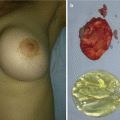
Giant cervicofacial LM is a special group of LMs that are usually placed in multiple tissue planes and involve vital structures (Fig. 14.2) [3, 4, 15]. Oral lesions may lead to macroglossia, speech problems, poor oral hygiene, caries, and malocclusion [3, 16]. Infections and intralesional hemorrhage are the most common complication of cervicofacial LMs [14, 15]. There is a five-staging system proposed by Seeres et al. based on the location and extent of the lesions in the neck (unilateral suprahyoid, unilateral infrahyoid, unilateral supra- and infrahyoid, bilateral suprahyoid, bilateral infrahyoid, and bilateral supra- and infrahyoid) [8, 16].
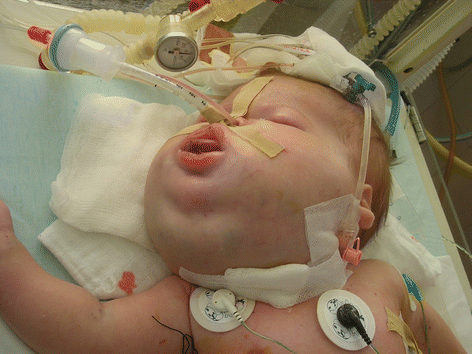

Fig. 14.2
Large lymphatic malformation of the head and neck
There are no accepted treatment protocols for cervicofacial LMs, the management is extremely difficult, and it requires multidisciplinary approach [3, 4]. The airway is the primary concern in an infant with LM [15]. Nearly 50% of patients require tracheotomy, and it should be performed without hesitation as an ex utero intrapartum treatment (EXIT) procedure or in the first days of life [3, 4, 15]. Treatment options for LMs in cervicofacial region are resection, sclerotherapy, laser coagulation, and radiofrequency ablation [3, 8, 10, 11, 15, 16].
The surgical treatment is best performed before development of facial image (3 years old), and it is usually connected with serious complications such as nerve damage, bleeding, lymphatic drainage, and infection [8, 15, 16]. Surgical excision should be avoided for the tongue and oral floor LMs, which are better to be treated with sclerotherapy (with doxycycline, bleomycin, OK-432, and ethanol) [15]. Recently, pharmacotherapy (sirolimus) is widely used as a first line of treatment for large cervicofacial LMs [17–19].
LMs in the orbit are uncommon and they account for 3% of all orbital masses [14]. Periorbital LMs present with swelling, intraorbital hemorrhage, infection, ocular proptosis, and blepharoptosis [3, 4]. Periorbital LM causes a permanent reduction in vision (40%), and 7% of patients become blind in the affected eye [3]. Treatment options are sclerosation with doxycycline, bleomycin, OK-432, and ethanol (for extraorbital lesions only), surgical treatment, and pharmacologic therapy (sirolimus) for large LMs [14–18].
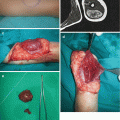
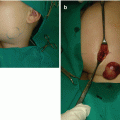
Generalized LMs present with multifocal or osteolytic bony lesions, splenic involvement, as well as pleural and/or pericardial effusions, and they are extremely difficult for treatment [18–21]. Skeletal involvement of LM is also known as Gorham-Stout syndrome, disappearing bone disease or phantom bone disease (Fig. 14.3a–c) [2, 3].
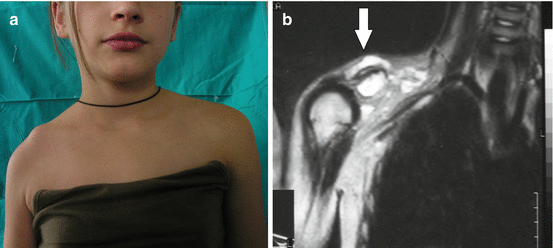

Fig. 14.3
Gorham syndrome: (a) deformation of the right shoulder area; (b) magnetic resonance imaging revealing presence of lymphatic malformation
14.4 Patient Evaluation
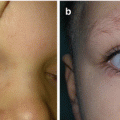
Diagnosis of LM in most cases (90%) is made on the basis of history and clinical manifestations [3, 4, 7, 8]. Small, superficial lesions do not require further evaluation [3]. Radiological techniques are used to determine anatomic extent of the disease and the rheologic nature [4, 7, 9]. Ultrasonography and color Doppler study differentiate slow-flow from fast-flow anomalies and a discrete tumoral mass from the anomalous channels of a vascular malformation [7–9]. Ultrasonography is less valuable for deep-seated lesions [4]. Magnetic resonance imaging (MRI) is the best radiological technique for characterizing LM [4, 7, 8, 11, 15]. MRI with contrast can help to distinguish LM from venous malformation (VM) or lymphaticovenous malformation (LVM) (Fig. 14.4a, b) [3, 4, 9, 15].
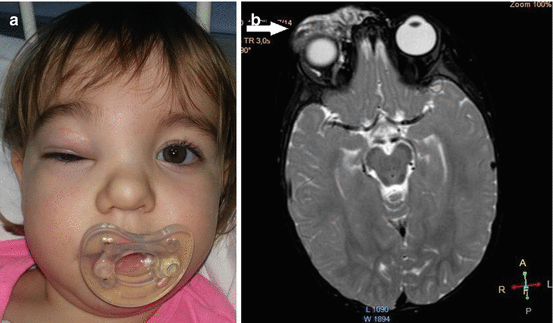

Fig. 14.4
Lymphaticovenous malformation of the right orbit: (a) clinical view; (b) magnetic resonance imaging of the head region revealing large intraorbital lymphaticovenous malformation
14.5 Treatment Option
LM is a benign lesion and small or asymptomatic lesions may be observed [3, 21]. Spontaneous regression of LMs is rarely seen [8]. Treatment of LMs is reserved in case of symptomatic lesions that cause pain and significant deformity or if the LM is a threat to a vital structures [3, 7, 8]. An infected LM often requires intravenous antimicrobial therapy [3]. Patients and families are counseled that LM can expand following any intervention, and thus additional treatments are often required in the future [3]. Treatment option for LM is usually multimodal, and treatment algorithms are not yet created [3, 6, 8, 10–12, 16–19, 22–29, 31]. Treatment of LMs includes observation, pharmacotherapy, excision, and sclerotherapy [3–31]. Disease location has a major influence on prognosis, and outcomes are not so favorable for children with orbital, parotid, laryngopharyngeal, and oral disease [10, 14, 15].
14.5.1 Sclerotherapy
Injection sclerotherapy induces endothelial inflammation in a vascular structure with the goal of causing thrombosis, occlusion, fibrosis, and contraction within a structure [4]. Sclerotherapy has emerged during the past several decades as first-line treatment option for large or problematic macrocystic/combined LM [3, 8, 12]. Sclerotherapy is less successful and less predictive for microcystic (deep-seated) LM, the response is not instantaneous, and usually multiple procedures are needed [3, 4, 6, 13]. Different sclerosing agents have been used for sclerotherapy of LMs such as ethanol, hypertonic saline, alcoholic solution of zein (ASZ) (Ethibloc), and sodium tetradecyl sulfate (STS) [6, 8, 12, 13, 22]. Ethanol has the highest complication rate (ulceration, nerve injury, systemic toxicity) [3, 20]. The most utilized sclerosing agents for LMs are doxycycline, bleomycin, and OK-432 [3, 4, 6–8, 10–12, 16, 22, 27, 29].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree