Key Words
extremity, limb, trauma, salvage, reconstruction, leg, foot, knee, amputation
Synopsis
Management of lower-extremity trauma involves a multidisciplinary approach that usually requires full trauma evaluation. The decisions for amputation vs. salvage as well as reconstruction require close collaboration by orthopedic and plastic surgeons, and must be individualized to patient, injury, and situation. Timely and adequate debridement, bony fixation, and wound closure are essential to limit the zone of injury and to reduce the risks of infectious and other complications. When immediate closure is impractical, negative pressure wound therapy should be considered. Reconstruction of soft tissue defects should respect the reconstructive ladder. Most wounds are amenable to primary or secondary closure or local tissue rearrangement. Pedicled flaps or free tissue transfer may be required for more complex or larger defects. Special considerations should be given to the unique role of the lower extremity in weight-bearing, as well as to the need to fit footwear or prosthetic devices to the reconstructed limb.
Clinical Problem
Lower-extremity trauma is an exceedingly common problem that, if not properly managed, can result in death or severe disability. Fortunately, most lower-extremity injuries heal with simple techniques of wound repair. However, for wounds that involve exposure of intact or fractured bone, orthopedic hardware, and vital structures such as tendons, arteries and nerves, more advanced reconstructive techniques are necessary to preserve a functional lower extremity.
The initial management of the lower-extremity patient should consist of a complete trauma evaluation in the context of polytrauma. Even isolated lower-extremity trauma may be life-threatening. Potential complications include vascular injury with massive blood loss, compartment syndrome, necrotizing fasciitis, and sepsis. Once life-threatening injuries have been triaged and resolved, detailed evaluation of lower-extremity injuries can proceed.
The key principles involve detailed assessment of the wound to identify issues that may threaten limb salvage and to determine which patients might be better served by amputation. Some important factors are (1) the presence of a fracture or open joint, (2) sensation and motor function of the extremity and foot, and (3) vascularity of the lower extremity and foot.
For successful limb salvage to occur, the patient must have the potential for bony union, joint and motor function sufficient for ambulation, and a sensate plantar surface to prevent ulceration and chronic denervation changes to the foot. Methods of bony fixation include external fixation, open reduction and internal fixation with extramedullary hardware, and internal medullary rodding. A multidisciplinary approach with orthopedic integration is necessary to assure bony union and success of reconstruction.
Appropriate debridement and adequate timing of coverage are essential. Typically, initial debridement should be performed until the wound is clean and clear of any obvious infection or necrotic material. Definitive orthopedic hardware placement should proceed with immediate soft tissue reconstruction to prevent bacterial colonization of the hardware and to reduce the risk of chronic osteomyelitis. When prompt definitive fixation is impractical, external fixation should be performed, followed by soft tissue reconstruction and closure of the wound. Delayed removal of the external fixator and placement of definitive orthopedic hardware may then be achieved with reduced risk of contamination.
Joint stability and adequate motor strength are necessary for ambulation. Fusion of the knee or ankle in a position favorable to ambulation is acceptable. However, joint instability or complete loss of motor control is not. A flail limb is not a candidate for reconstruction. Neurological status of the sole of the foot is also critical. Without plantar sensation, the foot will undergo inevitable ulceration and then Charcot deformity. In patients with permanent loss of plantar sensation, a stable amputation at a more proximal sensate location, followed by adequate prosthetic rehabilitation, will result in superior ambulation.
Relative contraindications for limb salvage include advanced diabetic or other neuropathy, severe peripheral vascular disease, limb shortening bony injury, and inadequate social support for extended hospitalization and rehabilitation. Patients that are expected to do well with a prosthesis and wish for early return to work are likely to do better with amputation and a short rehabilitation, versus the long hospitalization and rehabilitation that are often required for successful limb salvage. On the other hand, factors favoring attempted limb salvage include young patient age, limited access to prosthetics, and female gender. Ultimately, every injury as well as each patient requires careful, individualized consideration of all of the important factors to determine whether early amputation or an attempt at limb salvage is best for that patient.
Pre-Operative Management
Once the decision has been made to pursue limb salvage, the next most important pre-operative consideration is the timing of surgery. Initial irrigation and debridement of open fractures and joints should occur as soon as possible. Some authors suggest debridement within 6 hours of injury. However, there are no data to support this assertion. Rather, most authors would agree that debridement within the first 24 hours is adequate. Initial debridement is aimed at removing devitalized tissue and foreign material from the fracture and wound site. Once the wound is adequately debrided, immediate definitive orthopedic reconstruction or placement of an external fixation device can proceed. It is essential to communicate plans for definitive soft tissue reconstruction with orthopedic colleagues before bony fixation so as to avoid tissue damage that could limit options for local, regional, or free flap reconstruction. In cases where definitive orthopedic treatment is being deferred, careful planning will allow for safe re-elevation and re-inset of the flap at the time of definitive fixation. Fasciocutaneous flaps are generally easier to re-elevate because they tolerate division of their vascular pedicle after a few weeks of delay. In contrast, muscle flaps will frequently necrose when their pedicle is divided, even after considerable time has passed. For such flaps, the pedicle should be placed away from areas that are likely to require re-elevation.
Definitive closure of the wound should be performed immediately after the placement of orthopedic hardware and as soon as possible after the initial injury. Several studies have shown improved outcomes with respect to flap survival and avoidance of chronic infections when wounds are closed in the early period, variably defined as within 72 hours up to 6 days after the injury. Late closure is associated with a larger zone of injury, which would require more extensive debridement before closure. The expanded zone of injury potentially limits options for local, regional, or free flap reconstruction. The “zone of injury” is the area of local tissue that, although not directly traumatized, has been influenced by the inflammatory milieu caused by the wound. The increase in edema, inflammatory cytokines, and altered vascularity of the tissues in the zone of injury increases the risk of thrombosis of the vessels after microsurgical anastomosis as well as the risk of congestion of locoregional flaps due to reduced venous outflow. Additionally, tissue edema makes surrounding tissue less pliable, and therefore adjacent tissue transfer is more challenging. The effect of the zone of injury is accentuated with increasing time after the injury, with inflammatory conditions such as burns or systemic illness, and with infection in the area. Although the zone of injury is often overlooked, recognizing and minimizing it may be critical to successful lower-extremity reconstruction.
For wounds that cannot be closed early due to a variety of factors, such as comorbidities, accompanying injuries, limited orthopedic availability, or other practical considerations, aggressive management must be performed to prevent bacterial overgrowth and chronic osteomyelitis. Studies have shown that wound VAC application while awaiting delayed closure can reduce the risks of complications associated with delay. Lower-cost occlusive negative pressure dressings should provide similar benefits.
Pre-operative planning for wound coverage involves analysis of the size and location to determine the most suitable flap, evaluation of the vascular status of the limb to determine the blood supply for the flap, and continued perfusion distal to the defect.
The most detailed analysis of the vascular supply is by fluoroscopic, CT, or MR angiography. Where angiography is impractical or unavailable in developing countries, capillary refill, pinprick, tissue color, and turgor of the distal extremity should be evaluated. Anterior tibial/dorsalis pedis and posterior tibial pulses should be readily palpable, and the distal peroneal artery should be detectable posterior to the lateral malleolus using an 8-MHz vascular Doppler probe. In the absence of angiography, careful observation of distal perfusion after occlusion of secondary vessels is sufficient to ensure antegrade flow from the proximal vessels tested (akin to the Allen’s test in the hand). It is essential to confirm antegrade flow in any potential pedicle or recipient vessel. At least two-vessel flow to the foot must be confirmed if the operative plan will require ligation of one of the major arteries to the foot.
The supplies required for lower-extremity reconstruction depend widely on the type of injury and flap selection. Lack of certain supplies may limit the reconstructive options. Lower-extremity reconstruction often requires skin grafting. Therefore dermatome and skin mesher are essential. With adequate understanding of the anatomy, elevation of muscle flaps and fasciocutaneous flaps such as the cross-leg or reverse sural flap may be performed with basic plastic surgery instruments. Although use of a vascular Doppler is very helpful, it is not crucial. Smaller flaps such as propeller flaps are based on single perforators and therefore require an 8-MHz handheld vascular Doppler with a sterile probe. Finally, free tissue transfer requires the use of an intraoperative Doppler, operating microscope, specialized microsurgical instruments, 8-0 or 9-0 microsurgical suture, and experience with the specialized techniques for microsurgical anastomosis. In resource-constrained environments, free tissue transfer may be performed under loupe magnification. Nevertheless, without the proper instruments and fluency with microsurgical technique, free tissue transfer is almost certain to fail.
Surgery
Wounds covered by healthy granulation tissue or consisting of exposed muscle may be directly skin grafted. It is important to remember that large portions of composite wounds may be skin grafted, leaving only the portions that cannot be skin grafted (hardware, bone, nerve, tendon, or vessel) for flap coverage. With that kept in mind, the size and location of the wound will guide flap options.
Small wounds of the proximal two-thirds of the leg may be covered with adjacent tissue transfer such as a propeller flap or keystone flap. Larger defects of the proximal one-third of the leg, including exposure of the knee joint and patella, may be covered with a medial and/or lateral gastrocnemius flap. Defects of the tibia in the mid-third of the leg can usually be covered with the pedicled soleus muscle flap. Defects of the distal third of the leg and ankle are much more difficult to cover due to the lack of adjacent tissue and the fact that any local donor site defect will likely expose either the Achilles tendon or the bones of the ankle. Frequently, the best option for coverage in this area is free tissue transfer. Free flaps are selected based on accessibility, defect size, pedicle length, and donor site morbidity. Consideration must be given to prevent excessive bulkiness of flaps that may prevent shoe fitting. Pedicle length must be sufficient to allow for anastomosis to vessels that are outside of the zone of injury. Vein grafts may be used to extend the vessel length when necessary. Commonly utilized free flaps include muscle flaps such as the latissimus dorsi, rectus abdominis, and gracilis muscles as well as fasciocutaneous flaps such as the anterolateral thigh flap, thoracodorsal artery perforator flap, and deep inferior epigastric perforator flap. The most common lower-extremity recipient vessels are the anterior and posterior tibial vessels. End-to-side arterial anastomosis must be performed especially if Allen’s test is unreliable or the foot is supplied by a single vessel.
In situations when microsurgical reconstruction is not available, a distally based “reverse” sural artery flap is an option. Another option for selected patients is the cross-leg flap, which is a pedicled flap from the contralateral leg.
In the following sections, we describe the more common regional flaps and their use for lower-extremity reconstruction. Free flap techniques and flap descriptions are covered in Chapters 2.7 and 2.8 . Additional regional flaps throughout the body are covered in Chapter 2.6 .
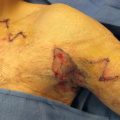
Gastrocnemius Flap—Most Useful Flap for Proximal 1/3 Lower Leg Defects
The gastrocnemius muscle (gastroc) has a medial head and a lateral head, supplied, respectively, by the medial and lateral sural artery and by branches of the popliteal artery ( Fig. 6.2.1 ). Additionally, anastomotic vessels cross between bellies, within the muscle substance. Both heads of the gastroc originate independently from the medial and lateral femoral condyles and converge in the Achilles tendon to insert onto the calcaneus. As such, the medial and/or lateral gastroc may be harvested and transposed independent of each other and used as muscle or musculocutaneous flaps. Sensation to the overlying skin is supplied by branches of the saphenous nerve medially and by branches of the sural nerve laterally.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree