12
Lichen Planus and Lichenoid Disorders
Valeria Mateeva1 and Snejina Vassileva2
1Department of Dermatology and Venereology, Military Medical Academy, Sofia, Bulgaria
2Department of Dermatology and Venereology, Medical University, Sofia, Bulgaria
Lichen planus (LP) is the prototype of a group of inflammatory dermatoses, referred to as lichenoid disorders, characterized clinically by small, often pruritic papules, and histologically by a dense, band‐like lymphocytic infiltrate in the papillary dermis, that often obscures the dermoepidermal junction (DEJ). The resulting damage of the DEJ is referred to as “lichenoid reaction pattern” or “interface dermatitis,” and is characterized by epidermal basal cell damage manifested by cell death and/or basal vacuolar change, apoptosis, and formation of shrunken eosinophilic cells with pyknotic nuclear remnants, scattered along the basal layer of the epidermis (Civatte bodies).
A number of etiologically and clinically distinct disease entities fall into the group of lichenoid disorders merely because of histopathologic features of lichenoid dermatitis. Of them, lichenoid drug eruptions (LDEs), also known as “drug‐induced lichen planus,” show true clinical and histological similarity to “idiopathic” LP and constitute its main differential diagnosis in clinical practice.
Although the exact etiology of LP is unknown, the disorder is currently regarded as a T cell‐mediated autoimmune disease in which inflammatory cells attack basal keratinocytes because of change in the antigenicity of their cell surface. The cause of this immune‐mediated basal cell damage is unknown, but LP has been associated with a number of intrinsic and extrinsic factors that can trigger the onset and relapse of the disease. Consequently, conventional therapies of LP are based on the beneficial role of various topically applied and/or systemically administered immunosuppressive and immunomodulatory agents. However, these approaches are not always effective to suppress disease activity and relapses occur, especially in forms with oral involvement, which can be often refractory to conventional treatment. Recent studies have shown interest in implementing a holistic approach to the management of LP and LDE, aimed at reducing the influence of the various factors preceding, triggering, or exacerbating those conditions.
Introduction
Lichen planus (LP) is a chronic inflammatory immune‐mediated muco‐cutaneous disorder that most commonly affects middle‐aged adults of both sexes. The estimated prevalence of cutaneous lichen planus (CLP) in the general population is <1%, while between 1% and 2% are reported to suffer from oral lichen planus (OLP) [1, 2]. There is a slight female predominance in adults with a male/female ratio of 2:3 [3]. Childhood LP is more common in African Americans [4], and boys seem to be relatively more often affected than girls [5].
CLP and OLP may have a serious impact on the quality of life and well‐being of the affected individuals because of their recalcitrant nature and disturbing subjective symptoms.
Given the strong existing interconnection between CLP/OLP and psychoemotional factors such as stress, depression, and anxiety, these conditions are clear examples of where a holistic approach for treatment is rational. The integrative management (IM) of CLP and OLP combines the best of conventional dermatology with some of the more holistic options and includes conventional and emerging therapies, but also complementary and alternative methods, such as diet, vitamin supplementation, herbs, lifestyle modifications, mind–body approaches, traditional Chinese medicine (TCM), and Ayurveda. Nowadays, in the Western world, there is a trend toward a healthier lifestyle that is closer to nature and a preference for limiting the use of pharmaceuticals where possible, so IM has gained popularity.
Pathogenesis
The etiology and pathogenesis of LP remain incompletely elucidated. Various hypotheses have been proposed, while the most advanced one focuses on a genetic predisposition to Th1 polymorphisms that might be responsible for promotion of T cell‐mediated immune reaction to an induced antigenic change [6].
The hypothetical antigen is processed by the dendritic cells (Langerhans cells) and is presented to the T‐lymphocytes, leading to their activation. The activated CD4+ and CD8+ T cells attack and cause damage to the keratinocytes of the basal epidermal layer [7].
Various external factors such as medications, stress, chemical substances, and infections may participate in the development of LP. A number of infectious agents, such as HCV (hepatitis C virus), HIV (human immunodeficiency virus), HSV2 (herpes simplex virus type 2), Candida, and Treponema pallidum, may be involved in the onset of LP through promotion of the lymphocytic activation [7]. There is strong evidence that HCV is associated with LP and may play a role in the pathogenesis of the disease [8–10].
The association of OLP with amalgam restorations has been extensively investigated. Hypothetically, a hypersensitivity to the mercury that is contained in the amalgam alloy may lead to development of the clinical lesions of OLP [11]. According to some authors, these lesions are more commonly contact oral lichenoid lesions, rather than genuine OLP [12].
Clinical Manifestations
CLP typically involves the flexural skin surfaces of the extremities. The classic lesion of CLP is a small, itchy erythematous to violaceous, flat‐topped, polygonal papule (Figure 12.1).
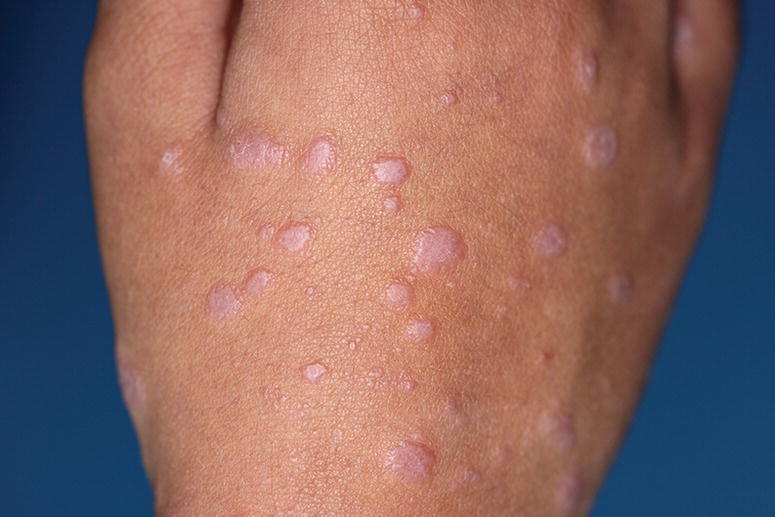
Figure 12.1 Classical erythematous to violaceous, flat‐topped, polygonal papules of CLP.
Whitish reticulated networks often develop on the surface of papules (Wickham striae). Usually the lesions are distributed in a bilateral and symmetrical manner.
LP can affect the oral and genital mucous membranes. OLP usually manifests clinically with bilateral white striations, papules, or plaques that may become erythematous or erosive. It affects the buccal mucosa, gingiva, or tongue. According to its clinical appearance OLP is classified into: reticular, papular, plaque‐like, erosive, atrophic, and bullous types [13, 14]. Reticular OLP is usually asymptomatic [15], but may sometimes cause discomfort and pain. Erosive OLP is by definition associated with burning and pain, and causes a significant impairment of the quality of life due to the chronic nature of the condition [8, 16].
Erosive and atrophic types of OLP may present an increased risk for malignancy and regular follow‐up is recommended to exclude the presence of dysplasia [17].
Vulvovaginal LP affects peri‐menopausal women and is characterized by a similar pattern as OLP. Major subtypes include papulosquamous, erosive, and hypertrophic LP of the vulva [18]. Chronic erosive vulvovaginal LP can lead to scarring with a resorption of the labia minora, clitoral burying, and stenosis of the vaginal introitus. These changes may complicate the sexual life of the affected females and thus cause significant distress and impairment of their quality of life [19].
Nails are affected in 1–10% of the adult cases of LP [20]. Nail LP is more frequent in children with a reported prevalence of around 20% [21]. The affected nails show clinical changes such as longitudinal ridging and splitting (onychoschizia), longitudinal striation (onychorrhexis), nail absence (anonychia), subungual hyperkeratosis, and thinning of the nail plate [22].
CLP can spread to the scalp causing cicatricial alopecia. Classic follicular LP (lichen plano‐pilaris, LPP), frontal fibrosing alopecia (FFA), and Graham‐Little‐Piccardi‐Lassueur syndrome are clinical forms of LP of the scalp [23]. Clinically, the typical lesion of LPP is a follicular hyperkeratotic papule.
In general, LP is described as self‐limiting. Nevertheless, lesions of LP may sometimes persist for months to years, and may significantly decrease the quality of life of the affected subjects through clinical appearance, associated subjective symptoms, residual scarring, and post‐inflammatory hyperpigmentation.
Emotional and Mental Aspects
Psychosomatic factors such as emotional stress play a substantial role in triggering LP and OLP. The Griesemer index, which is used to rate dermatologic disorders on a percentage scale from 100% to 0% based on frequency of emotional triggering of a condition, attributes to LP a score of 98% [24]. Stressful events precede the onset of LP in up to 68% of the cases [25, 26]. Stress is defined as a biological reaction to internal or external stimuli that disturbs the organism’s homeostasis [27]. Half of patients with LP associate the exacerbation of the disease with stressful events in their lives [28, 29]. Data from the past three decades suggest that chronic and long‐lasting stress may contribute to the onset and exacerbation of a variety of diseases [30–32].
Although the majority of patients suffering from OLP do not report a triggering factor, stressful events such as illness or death of a loved one could precede or aggravate the development of the clinical lesions [33, 34]. This interconnection between emotional stress and skin conditions may be mediated by a chronic activation of the sympathetic system [35, 36].
The stress‐induced theory for the development of LP is supported by elevated cortisol levels in saliva reported in patients with OLP [37].
Various endocrine and immune parameters in LP may be associated with psychological stress. The T cell‐mediated immune response that leads to the appearance of the clinical lesions in LP is activated in response to stressogenic stimuli. A possible pathway for inducing LP may be a dysregulation of immune functions with alteration of the proportion of T‐helper (Th) 1/Th2 cytokines (i.e. interleukin‐18 [IL‐18], IL‐4, IL‐6, TNF‐α, IL‐10) and increased Th2 response [38]. Additionally, stress may alter the expression of surface antigens on T cells such as CD25, CD16, CD8, and CD45RA on T‐lymphocytes [28].
The elevated stress level in LP may be treated with cognitive‐behavioral methods, relaxation training, or self‐hypnosis [39]. Furthermore, individuals who suffer from OLP often demonstrate high levels of depression and anxiety related to their condition and impaired life quality [40]. The disease significantly affects the quality of life in 90–100% of the patients with LP [41]. Areas of feelings are severely impaired. Women tend to express higher levels of stress in the workplace and a higher rate of body dissatisfaction compared to males. Furthermore, females are more prone to avoiding social activities because of low self‐esteem and feeling of unattractiveness [42]. More than 50% of the patients with LP are reported to have psychiatric morbidities such as depression, social phobia, anxiety, and panic attacks [43, 44]. The prevalence of depression among patients with LP ranges from 21 to 63% in clinical studies [27, 45, 46]. In almost 90% of the cases the depressive state is graded as severe [42].
Psychological problems in patients with LP need to be addressed early and managed in parallel with the dermatological condition.
Diagnosis
Diagnosis of LP is made on the basis of clinical presentation and histological findings on biopsy specimens obtained from lesional skin. Direct immunofluorescence (DIF) study may be needed in cases of erosive OLP and bullous CLP in order to differentiate these conditions from autoimmune bullous disorders [47].
Treatment
The treatment of CLP and OLP is often challenging. Conventional therapies may fail to provide longstanding improvement [48]. An integrated approach, which acknowledges the beneficial effect of complementary and alternative treatments in addition to the conventional therapeutic modalities, may provide additional benefit for patients suffering from these conditions.
Conventional and Emerging Therapies
The therapy for CLP and OLP aims at minimizing the flares of the disease, alleviating the associated pain in OLP, and improving the overall quality of life. Topical preparations are the mainstays of therapy, but systemic therapy is indicated in widespread and severe cases.
Currently, a number of treatment modalities have shown effect in CLP and OLP (Table 12.1).
Table 12.1 Conventional and emerging therapies for treatment of CLP and OLP.
| Conventional and Emerging Therapies for CLP and OLP | ||
| Topical | Systemic | Surgical |
|
|
|
Topical Therapies
High‐Potency Topical Corticosteroids (TCSs)
Topical corticosteroids (TCSs) are proven to reduce the size of the lesions in LP and improve the clinical status [49], and are currently the mainstay treatment of OLP [50]. They act through suppression of the activated T cells and thus reduce the immune reaction. They are applied directly to the lesions and cause alleviation of the inflammation and pain [51]. Clobetasol, betamethasone, dexamethasone, and triamcinolone are used for the treatment of OLP [52]. There is no evidence for superiority of a particular TCS over the others in reducing the subjective symptoms associated with OLP, particularly the pain in erosive forms of the disease [48].
Formulations for intraoral use designed for the treatment of OLP are developed to improve the efficacy of traditional topical therapies, such as topical steroids and calcineurin inhibitors (CNIs). Adhesive tablets of clobetasol in combination with a mucoadhesive polymer (polysodium methacrylate, methylmethacrylate) demonstrate promising results [53]. A microsphere formulation of clobetasol 0.025% shows better results in relieving the pain associated with OLP than classical formulation (lipophilic ointment in a hydrophilic phase 0.025%) [49].
The prolonged use of TCS is associated with well‐known side effects, which may limit their application in chronic conditions such as OLP.
Antifungals may be used as adjunctive treatment in OLP, managed with TCSs. The addition of an antifungal agent does not affect the therapeutic outcome [54], but significantly reduces the risk of secondary candidiasis [48].
In cases with an increased susceptibility to oral candidiasis (diabetes, immunodeficiencies), CNIs such as tacrolimus may be preferred over clobetasol [52].
Non‐steroid Topical Medications
Although topical steroids are the mainstay in the treatment of LP and OLP, non‐steroid topical medications also have a place in the therapeutic armament. They are considered in cases of steroid intolerance, contraindication for their use, or in severe cases, where local corticosteroid medications have failed to demonstrate sufficient efficacy.
- Topical CNIs
Tacrolimus is effective in treating erosive, refractory, and recurrent forms of OLP and reduces the size and duration of the lesions [55, 56]. Usually, a long‐term intermittent treatment is required to obtain optimal results. Some authors suggest greater efficacy of tacrolimus in comparison with clobetasol for treatment of OLP [52], especially the erosive form [57]. However, the use of tacrolimus in OLP is limited by the increased carcinogenic risk that may be associated with the treatment [58]. Tacrolimus should be strictly applied only to the affected area and continuous treatment should be avoided.
Another topical CNI, pimecrolimus 1% cream, is reported to show similar efficacy in OLP as moderate potent TCS triamcinolone acetonide [59, 60].
- Topical retinoids
Isotretinoin is the most frequently used retinoid in the treatment of OLP. The most frequently reported side effect is a transient and moderate burning sensation. Keratotic OLP may respond better to topical retinoids than the erosive forms, but more comparative and controlled clinical trials are needed to clarify this matter [61].
Topical tretinoin 0.1% demonstrates statistically significant amelioration of OLP lesions in patients with OLP, with minimal side effects restricted to the treated area [62]. Authors suggest that tazarotene 0.1% is more efficacious than placebo in reducing signs and symptoms in reticular OLP. Mild side effects such as slight burning sensation and taste abnormalities are reported with the use of the drug [63].
- Other
Novel topical therapies intended for treatment of erosive OLP include thalidomide [64], imiquimod 5% cream [65], sirolimus (rapamycin) [66], intralesional Bacillus Calmette‐Guerin polysaccharide nucleic acid (BCG‐PSN) [67], cyclosporine, and topical sulfasalazine [52]. Topical sulfasalazine should be considered in OLP unresponsive to TCSs, particularly in patients with high concentrations of IL‐1β and IL‐8 in the saliva [68]. A novel topical niosomal gel formulation of hydroxychloroquine applied once daily may be promising in controlling the clinical symptoms and pain in CLP and OLP [69].
Phototherapy
Narrow‐band ultraviolet B phototherapy (NB‐UVB), oral psoralen plus UVA (PUVA), and PUVA bath have been studied for treatment of disseminated and generalized CLP. NB‐UVB acts through photoinduction of apoptosis of T‐lymphocytes and immunosuppression, and leads to reduction of the inflammation and clinical improvement in CLP [70–72]. An average of 30–40 photo sessions are recommended to achieve significant clinical improvement of CLP in up to 70–80% of the patients [73, 74].
Two‐thirds of patients with generalized CLP are reported to respond favorably to NB‐UVB treatment. No remarkable adverse events are described. The promising response to the treatment, in association with its excellent safety profile proven in small case‐series, prompts further clarification of the place of NB‐UVB in the treatment of generalized LP [75]. Studies show that the long‐term therapeutic efficacy of oral PUVA is similar to UVB‐311 nm in the treatment of patients with disseminated CLP, while PUVA produces a better initial clinical response rate [76]. PUVA bath, or external PUVA, when the photosensitizing agent is applied topically, has comparable efficacy to oral PUVA and NB‐UVB [77, 78].
Systemic Therapies
Systemic Corticosteroids
Systemic therapy with oral glucocorticoids (GCs) is indicated in cases of severe mucocutaneous LP. Usually they are recommended as first‐line systemic treatments and are administered in short courses [79]. Doses range from 0.5–1 mg kg d−1 and the treatment typically lasts 6–8 weeks, with gradual tapering of the daily dose. Relapses after interruption of the treatment are not uncommon [80]. Comparative studies show a significantly higher complete response rate in patients treated with oral prednisone than in those managed with low‐dose enoxaparin (low‐molecular weight heparin) after 8 weeks of treatment [81]. On the other hand, NB‐UVB phototherapy seems to be more effective than oral GCs in CLP [82]. Authors suggest no statistically significant difference in the complete response rate between oral betamethasone 5 mg twice weekly and cytostatic agent methotrexate 10 mg/weekly [83].
Oral GCs such as betamethasone are indicated for widespread and recalcitrant cases of OLP [84]. Nevertheless, they have limited use in this condition because of their chronic course and the need to balance the benefits versus the potential well‐known side effects of the prolonged use of systemic GCs [11].
Immunosuppressants
Given the high rate of recurrences after cessation of systemic corticosteroid therapy in severe LP, immunosuppressants are often used as a therapeutic alternative. Methotrexate, cyclosporine, azathioprine, and mycophenolate mofetil present an interest in the treatment of a wide number chronic inflammatory dermatoses, including LP, as they lack the adverse effects related to the use of systemic corticosteroids [85].
Methotrexate, a folate analogue, has a wide range of anti‐inflammatory and immunosuppressive effects such as reduction of cell proliferation rate, increase in T cell apoptosis, changes in the expression of cellular adhesion molecules, influence on cytokine production, etc. [86], and is the gold standard for management of psoriasis. Particular strengths of this drug are its affordable price and good safety profile when regular monitoring is applied. Several studies demonstrate the efficacy of methotrexate in the treatment of generalized CLP [87], including in pediatric cases [21]. Doses in adults range from 10 to 15 mg/weekly.
Trials suggest comparable efficacy of oral methotrexate 10 mg week−1 and oral betamethasone after a three‐month treatment [83]. Authors report an average improvement of more than 50% with methotrexate 15 mg/weekly after a 12‐week therapy [88], and around 80% clearance after a 14‐week drug application [21]. Methotrexate [89, 90] and mycophenolate mofetil are reported to be effective in some cases of OLP, including recalcitrant erosive forms of the disease [11, 91].
Mycophenolate mofetil has shown efficacy in the treatment of LP. Furthermore, its better safety and tolerable adverse‐effect profile in comparison with cyclosporine or azathioprine [92] points out the advantage of mycophenolate mofetil as the immunosuppressant of choice for this condition [93].
Oral Retinoids
Oral acitretin is administered for treatment of LP at a dose of 30 mg kg d−1 for a period of eight weeks [94]. The drug is more efficient than placebo in reducing the clinical signs of CLP and alleviating the associated pruritus [78].
Oral alitretinoin (9‐cis retinoic acid) at a dose of 30 mg daily is reported to reduce disease severity of severe refractory to standard treatment OLP in a substantial proportion of patients. The treatment is well tolerated with minor side effects and may thus represent one new therapeutic option for this special group of patients [95].
Enoxaparin
Enoxaparin is a low‐molecular‐weight heparin (enoxaparin). A low‐dose enoxaparin regimen is shown to inhibit the expression of the enzyme heparinase [96].
The latter is produced by the CD4 + lymphocytes and is thought to play an important role in the lichenoid interface reaction. The heparinase is thought to facilitate the penetration of lymphocytes into the subendothelial basal lamina [97].
A dose of 5 mg/weekly of subcutaneous enoxaparin is proven to have a therapeutic effect in LP. Although enoxaparin is less effective than oral prednisone 0.5 mg kg d−1 in controlling the clinical symptoms and the itch, there is no difference in the overall response rate between both treatments. Furthermore, the number of patients with adverse events seems lower in the enoxaparin group [81].
Griseofulvin
A systematic review and meta‐analysis of treatments for CLP shows higher overall response rate of griseofulvin in comparison with placebo. The treatment regimen consists of 500 mg d−1 griseofulvin for 4 or 8 weeks [78, 98, 99].
Oral Sulfasalazine
Sulfasalazine 2.5 mg d−1 for 6 weeks for LP demonstrated higher overall response rates than placebo in one trial. Additionally, the drug was significantly better than placebo in controlling the disease‐associated pruritus [100].
Oral Thalidomide
Thalidomide (a‐N‐phthalimidoglutarimide), a potent anti‐inflammatory drug, modulates TNF‐a‐mediated cellular responses. Thalidomide has shown variable results in the management of mucocutaneous LP [101]. Its pharmacological action probably consists of preventing lymphocytic proliferation in response to mitogenic and antigenic stimuli [102]. Small case series show promising results of oral thalidomide 100 mg d−1, applied until all lesions and symptoms completely regress. Side effects include neuropathy, which usually manifests at the beginning of the treatment and may cause a significant withdrawal from the therapy [103]. Larger, randomized, double‐blind, placebo‐controlled trials are necessary to evaluate the real efficacy of this treatment.
Metronidazole
Oral metronidazole has been reported to be effective in some patients with idiopathic LP. The therapeutic effect of the drug in LP is probably mediated by its immunomodulatory properties. The response rate of metronidazole treatment of generalized CLP is around 80% [104]. However, oral metronidazole 200 mg three times daily is reported to be less effective than betamethasone dipropionate 0.05% cream, and oral 8‐methoxypsoralen plus sunlight exposure (PUVASOL) [105].
Hydroxychloroquine
Regulatory T cells (Tregs), important mediators in a number of inflammatory and autoimmune diseases, may play a role in the immunopathogenesis of OLP. Tregs appear to be downregulated after hydroxychloroquine treatment in OLP [106]. The overall response rate in LP after a treatment with hydrochloroquine 400 mg d−1 for 6 months is around 70%. A comparative study suggests that hydroxychloroquine is better than griseofulvin 500 mg d−1 in treating LP [107].
Dapsone
A nonrandomized case‐control study suggests a similar efficacy of oral dapsone 150 mg d−1 and topical betamethasone 0.1% [108]. These results are confirmed by another randomized study, that shows similar efficacy of oral dapsone 100 mg twice daily plus iron and folic acid tablets, and three other treatment modalities: triamcinolone acetonide 0.1% applied twice daily, topical tacrolimus 0.1% twice daily, and topical retinoid twice daily [109].
Biologicals
TNF‐α definitely plays a role in the pathogenesis of LP [110]. TNF‐α inhibitors such as adalimumab [111], and etanercept [112] have been studied for treatment of OLP and have demonstrated efficacy in single cases.
Rituximab, an anti‐CD20 monoclonal antibody, has also been reported effective for treating a case of erosive LP [113].
It is worth noting that both TNF‐α and rituximab are also known to cause LDEs [114, 115].
Apremilast
Apremilast, a phosphodiesterase‐4 inhibitor, is a drug used for treatment of psoriasis and oral ulceration in Behcet’s disease [116, 117]. Apremilast inhibits the production of TNF‐alpha, IFN‐gamma, IL‐2, IL‐5, IL‐8, and IL‐12 [118]. These cytokines are involved in the pathogenesis of OLP and contribute to the activation of the cytotoxic T‐cells, which cause the basal keratinocytic apoptosis in OLP [119]. Recent reports describe the efficacy of apremilast in recalcitrant cases of erosive OLP after failure of topical and other systemic therapies, such as immunosuppressants and hydroxychloroquine [120, 121]. The suggested dose is 30 mg twice daily. Authors report great patient satisfaction with the treatment and complete disappearance of the subjective symptoms.
To summarize, oral apremilast may be a safe and effective treatment for erosive OLP.
Surgical Therapy
Low‐energy lasers and free gingival grafts have been investigated for treatment of OLP [122, 123]. Further studies are needed to evaluate the efficacy of these techniques.
Stem Cell Transplantation
Mesenchymal stem cells (MSCs) transplantation is hypothetically proposed for the treatment of OLP [124]. MSCs are multipotent non‐hematopoietic progenitor cells that are used in the regenerative medicine because of their capability to differentiate into various cell types. Moreover, MSCs exercise an immunosuppressive action on a number of immune cells. Their immunosuppressive properties could be beneficial for the treatment of T‐cell‐mediated autoimmune diseases such as OLP.
Complementary and Alternative Treatments
CLP and OLP are conditions that are difficult to treat and the therapeutic results after conventional therapy may be disappointing. Furthermore, the use of systemic medications in more severe cases may be complicated by their potential side effects.
A holistic and integrative approach to dermatological diseases has gained popularity in recent years. Its basic principle is that the health of the skin is a reflection of the whole body and incorporates therapeutic practices from conventional and alternative/traditional medicine [125].
The IM of LP involves standard dermatological therapies in combination with dietary and lifestyle modifications, herbal medications, vitamin supplementation, mind–body approaches, Ayurveda, etc. (Figure 12.2).
Figure 12.2 Alternative/complementary treatment modalities used in the integrative management of lichen planus. OLP, oral lichen planus.
Hygiene
A good dental and oral hygiene is recommended in patients with OLP. Suboptimal hygiene is related to an increased risk for development of oral cancer [126].
Dietary Modifications
A diet rich in fruit and vegetables is advisable in patients with OLP as it may reduce the risk of development of squamous cell carcinoma. Certain foods may aggravate the pain associated with erosive OLP. A soft diet is implemented in these cases [127].
Topical and Oral Herbal Medications
- Curcuminoids
Curcuminoids (CUs) are components of the root turmeric (Curcuma longa) and are known for their anti‐inflammatory properties. High doses of CUs (6000 mg/daily) are reported to be effective in reducing the clinical signs and subjective symptoms in patients with OLP [128]

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree