Lentigo maligna is an overgrowth of atypical melanocytes at the dermal–epidermal junction also known as melanoma in situ. Left untreated, these lesions can continue to grow, resulting in dermal invasion and progression to lentigo maligna melanoma. Many operative and nonoperative treatments have been developed with the goals of preserving function and cosmesis while at the same time addressing the diffuse nature of these lesions. Previous recommendations have led plastic surgeons to commonly perform wide local excision with 5 mm margins. More recent literature has suggested that in many cases this treatment can result in high recurrence rates. This has led to margin control procedures becoming the treatment of choice for these lesions.
Lentigo maligna (LM) is a lesion of the dermal epidermal junction, which, if untreated, harbors the risk of dermal invasion progressing to invasive LM melanoma (LMM). In fact, multiple studies have shown that approximately 16% of lesions designated as LM (melanoma in situ) on biopsy actually harbor areas of early invasive melanoma (LMM). A well-described condition, LMM has been estimated to comprise 4% to 15% of invasive melanomas. Both the in situ and invasive lesions have a propensity for developing on the head and neck and together represent the most common melanocytic malignancy of that body region. A condition more common in the elderly and fair- skinned population related to chronic sun exposure and damage, LM/LMM is a problem on the rise. Incidence rates continue to increase, and although the number of cases increases significantly with age, lesions also have been diagnosed in the second and third decades of life. As people live longer and lead more active lives, the incidence of LM/LMM will continue to rise. Coupled with this rise in incidence, the literature reflects renewed scrutiny of available treatment methods and the development of new therapeutic approaches to this disease. The histologic characteristics of LM/LMM, more specifically the diffuse nature of melanocytic overgrowth, make these lesions difficult to treat, with recurrence rates ranging from 2% to 50%.
The current nomenclature for early melanoma of the skin can be confusing. The term lentigo maligna has been used by some to describe a proliferation of atypical melanocytes at the dermal–epidermal junction, which is not great enough in number or extent to constitute a melanoma in situ. Others, including the authors, assert that LM is melanoma in situ and should be regarded and treated as early melanoma. Moreover, the term lentigo maligna melanoma has been used to describe a melanoma arising from within an LM (melanoma in situ) as opposed to using the term to properly describe an advanced invasive LM (melanoma in situ). For the purposes of this article, LM is melanoma in situ, and LMM is an advanced invasive lesion that acts as any other cutaneous melanoma and should be evaluated and treated as such.
This article provides an overview of this disease and a critical examination of the data evaluating various treatment methods. In doing so, the authors hope to further the discussion of these treatment methods and aid in clinical decision making to provide patients optimal care while keeping in mind the critical issues of preserving both function and cosmesis in the head and neck region.
Epidemiology and patient demographics
Originally described by Hutchinson in 1912 and further defined by Dubrueilh LM and LMM are diseases that typically present in the sixth and seventh decades of life. Although the average age of presentation for other subtypes of malignant melanoma has been estimated to be in the 45- to 57-year range, the average age of presentation for LM and LMM falls slightly higher, in the 66- to 72-year range. Because of varying degrees of sun exposure to the population, the incidence of LM/LMM varies from country to country and region to region. The overall annual incidence of LM/LMM in Australia at one point was estimated to be 1.3 cases per 100,000 population, compared with the United States, where the incidence was estimated at 0.8 and 0.6 cases per 100,000 population for males and females, respectively, based on SEER (Surveillance, Epidemiology and End Results) data. More recently, groups such as Swetter and colleagues further examined SEER data looking at LM/LMM for a specific region and confirmed that in an area of high sun exposure such as southern California, the incidence of LM/LMM is on the rise overall and in specific patient populations related to sex and age. As indicated from the incidence data, there are slight differences in LM/LMM in relation to the sexes. Although the head and neck region has been documented to be the dominant site of presentation, the likelihood of developing LM/LMM at specific locations on the head and neck has been shown to differ between the sexes. Although the distribution of lesions on the cheek and nose remains equal between the sexes, males have a 3.1 times increased risk of development of ear lesions, and females are at 2.5 times increased risk of a lesion developing on the forehead.
Clinical presentation/diagnosis
LM and LMM in many ways continue to present a clinical conundrum. Although the original description by Hutchison describes a tan-colored lesion, hence the term Hutchison’s melanotic freckle, savvy clinicians are aware of the vast differences in presentation that are associated with this disease. The typical presentation is that of a tan colored macule located on chronically sun-damaged skin on the head and neck of middle-aged and elderly patients ( Fig. 1 ). Lesions, however, can vary in location, size, and color, ranging in spectrum from tan to black with or without variegation. In some cases, LM/LMM may present as a pink patch with little to no pigmentation, so-called amelanotic LM/LMM. Clinicians also must keep in mind that previous treatments may alter the appearance of the lesions. Imiquimod, a treatment that will be discussed in more detail later, has been shown in some instances to remove visible pigment while having no effect on the histologic findings. Similarly, treatment with cryotherapy may result in areas of depigmentation. Many of the these factors can lead to delays in diagnosis, which may be up to four times longer that other forms of cutaneous malignant melanomas. Overall, a high clinical suspicion remains the best strategy in diagnosis.
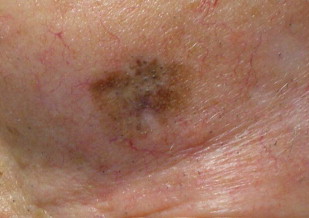
Most patients will present to clinicians with a changing lesion on the head and neck, which, in many instances will be in high-stakes areas around the eyes, nose, mouth, and ears. It is not uncommon for patients to note a change in a lesion that has been present for years. The noted change in the lesion may be related to a number of characteristics but often is related to size, as the natural course of LM is to expand in a centrifugal fashion before progressing to LMM by invading vertically into the dermis. There is no exact time frame for dermal invasion of LM. These lesions generally are considered slow-growing. However, in recent histologic evaluations of staged excision lesions, 16% were found to have unexpected foci of invasion. The risk of invasion also has been thought to be related to the size of the lesion, as in some case series, larger lesions have been the ones to frequently harbor invasive nests.
The ill-defined nature of LM/LMM on presentation may generate a wide differential diagnosis including but not limited to dysplastic nevus, pigmented basal cell carcinoma, squamous cell carcinoma, seborrheic keratosis–lenticular form, actinic keratosis, and lentigo simplex. Clinical evaluation can be aided by tools such as the Wood’s lamp, which can help highlight features such as subclinical pigmentation. Other forms of advanced imaging also can aid in diagnosis, including dermatoscopy, epiluminescence microscopy, and confocal laser microscopy. These imaging modalities continue to evolve and remain outside the scope of plastic surgery practice. For plastic surgeons, high clinical suspicion, thorough physical examination, and a low threshold for excisional biopsy remain the tenets for clinical evaluation of these lesions. Excisional biopsy should continue to be the gold standard, as it eliminates sampling error and if done properly should prevent transection of the lesions at the deep margin, if invasion is present and further staging required. These lesions, however, are often quite large and approximate critical facial structures. In such cases, an incisional biopsy of the darkest or most raised portion of the lesion is the preferred approach.
Clinical presentation/diagnosis
LM and LMM in many ways continue to present a clinical conundrum. Although the original description by Hutchison describes a tan-colored lesion, hence the term Hutchison’s melanotic freckle, savvy clinicians are aware of the vast differences in presentation that are associated with this disease. The typical presentation is that of a tan colored macule located on chronically sun-damaged skin on the head and neck of middle-aged and elderly patients ( Fig. 1 ). Lesions, however, can vary in location, size, and color, ranging in spectrum from tan to black with or without variegation. In some cases, LM/LMM may present as a pink patch with little to no pigmentation, so-called amelanotic LM/LMM. Clinicians also must keep in mind that previous treatments may alter the appearance of the lesions. Imiquimod, a treatment that will be discussed in more detail later, has been shown in some instances to remove visible pigment while having no effect on the histologic findings. Similarly, treatment with cryotherapy may result in areas of depigmentation. Many of the these factors can lead to delays in diagnosis, which may be up to four times longer that other forms of cutaneous malignant melanomas. Overall, a high clinical suspicion remains the best strategy in diagnosis.
Most patients will present to clinicians with a changing lesion on the head and neck, which, in many instances will be in high-stakes areas around the eyes, nose, mouth, and ears. It is not uncommon for patients to note a change in a lesion that has been present for years. The noted change in the lesion may be related to a number of characteristics but often is related to size, as the natural course of LM is to expand in a centrifugal fashion before progressing to LMM by invading vertically into the dermis. There is no exact time frame for dermal invasion of LM. These lesions generally are considered slow-growing. However, in recent histologic evaluations of staged excision lesions, 16% were found to have unexpected foci of invasion. The risk of invasion also has been thought to be related to the size of the lesion, as in some case series, larger lesions have been the ones to frequently harbor invasive nests.
The ill-defined nature of LM/LMM on presentation may generate a wide differential diagnosis including but not limited to dysplastic nevus, pigmented basal cell carcinoma, squamous cell carcinoma, seborrheic keratosis–lenticular form, actinic keratosis, and lentigo simplex. Clinical evaluation can be aided by tools such as the Wood’s lamp, which can help highlight features such as subclinical pigmentation. Other forms of advanced imaging also can aid in diagnosis, including dermatoscopy, epiluminescence microscopy, and confocal laser microscopy. These imaging modalities continue to evolve and remain outside the scope of plastic surgery practice. For plastic surgeons, high clinical suspicion, thorough physical examination, and a low threshold for excisional biopsy remain the tenets for clinical evaluation of these lesions. Excisional biopsy should continue to be the gold standard, as it eliminates sampling error and if done properly should prevent transection of the lesions at the deep margin, if invasion is present and further staging required. These lesions, however, are often quite large and approximate critical facial structures. In such cases, an incisional biopsy of the darkest or most raised portion of the lesion is the preferred approach.
Histology
The histologic hallmark in diagnosing LM is the presence of an increased number of atypical melanocytes at the basal layer of the epidermis in small nests or single cells, usually with extension into the periadnexal structures ( Fig. 2 ). Melanocytic atypia can be characterized in various ways, including but not limited to the presence of dendritic melanocyte processes, multinucleated melanocytes, or cytoplasmic retraction artifact. Other observed histologic markers that can aid in the diagnosis include effacement of rete ridges, atrophy of the epidermis, underlying solar elastosis, and the presence of an inflammatory dermal infiltrate. These additional criteria are intimately related to the typical presence of solar damage to the skin in and around the area of the lesions.
Despite the many histologic markers, the diagnosis of lentigo maligna does not remain without significant problems. The most significant of these problems remains the fact that LM develops in areas of significant solar damage, which in essence muddies the water surrounding these lesions. These transition areas between the lesion itself and the surrounding solar damaged skin can lead to inadequate excisions and recurrent disease depending on the method of excision and how the histology is processed and evaluated. Several immunohistochemical stains have been employed in some studies to aid in diagnosis. The most commonly used are HMB45 and MART-1.
A second problem lies in the categorization of LM as melanoma in situ. Although the criteria for diagnosing LM are widely accepted, some pathologists will make a differentiation between lentigo maligna as a melanoma precursor and a true melanoma in situ based on the number of atypical melanocytes and their pattern of arrangement within the basal epidermis. It is therefore imperative that plastic surgeons work closely with pathologists and have a knowledge of their methods of diagnosis and interpretation to understand the histologic diagnosis. The current standard nomenclature uses the term lentigo maligna to refer to malignant melanoma in situ occurring on the head/neck of elderly individuals in the setting of chronic actinic damage. LMM is an invasive melanoma that occurs within a lesion of LM. LM is a precursor to LMM.
Treatment
Treatment of LM and LMM has evolved over the years. Although surgical excision remains the gold standard, the methods by which excision is undertaken have changed significantly. Various methods are used, including direct excision with margins, Mohs micrographic surgery, and staged excision. Recurrence rates reported using these various methods range from 0.5% to 33% depending on technique. Other less-invasive treatment methods including topical and physical nonspecific destructive modalities also have been introduced but ultimately have proven less effective, with recurrence rates ranging from 20% to 100%. Many of these less-invasive therapies also lack long-term follow-up and have not stood the test of time.
The continuing dilemma that faces clinicians treating LM is balancing the morbidity of the various treatment methods with the risk of recurrence of these lesions. It has been shown that LM/LMM has higher rates of recurrence than other forms of cutaneous malignant melanoma. This dilemma is particularly difficult in a predominantly elderly population, as some patients are not suitable to undergo excision procedures. Another consideration is that the location of these lesions tends to be on the head and neck in functionally and cosmetically sensitive areas that may not lend themselves to straightforward excisions and repairs/reconstructions.
As the treatment of LM/LMM continues to evolve, it is important to remember that various specialties including plastic surgery, dermatology, ophthalmology and otolaryngology treat this disease. Although most LM/LMM cases can be treated successfully by a single clinician performing both excision and defect repair, a team approach may be necessary to achieve optimal outcomes depending on lesion size and location. The remainder of this article will examine the various surgical and nonsurgical treatments of LM and LMM, focusing on the techniques and efficacy of the treatments and their practical applications in relation to plastic surgery practice.
Nonsurgical therapy
Radiotherapy
Radiotherapy for LM first was described in the European literature by Miescher in the mid 1950s. Widely accepted and used for primary treatment of LM and adjuvant treatment of LMM in Europe, this treatment modality has been speculated to be at least as effective as surgical excision. Published recurrence rates have been found to be between 0% and 13%, with multiple studies having follow-up of 2 years or greater.
Despite the use of radiotherapy as a primary treatment abroad, it for the most part has remained a secondary option to surgical excision within the United States. Typically, radiotherapy has been reserved for patients who have significant medical comorbidities, are unable to undergo surgical treatment, or have lesions in functionally or cosmetically sensitive areas that are not favorable for surgical excision. Treatments often are given once or twice a week and can provide adequate local control without the inconvenience of needing daily therapy. Within the United States, regimens historically have used high voltage schedules of between 100 kV and 280 kV, delivering at depths of 5 to 6 mm depending on lesion size. This method has proven successful, as it provides high tissue penetration and good field coverage but also exposes the patient to greater adverse effect risk secondary to exposure of underlying structures. In an effort to decrease morbidity, European centers have employed lower voltage regimens using 10 kV to 50 kV, applying at depths of around 1 mm. Although this strategy intends to target these epidermal lesions more specifically, these soft x-ray regimens may run the risk of being too specific in the face of the diffuse nature of LM/LMM. In addition, the extension down follicular structures characteristic of LM/LMM suggests that 1 mm treatment depth might be insufficient. Close follow-up of patients treated with radiotherapy monitoring for both recurrence and treatment adverse effects is essential.
Despite the variable outcomes, radiotherapy remains another effective tool in the treatment of LM/LMM when surgical intervention is not possible or preferable. In the appropriately selected patient population, it can serve as a primary or adjuvant therapy. Many factors must be weighed in the decision to proceed with radiotherapy over surgical therapy, including patient age, comorbidities, goals of treatment, lesion characteristics/location, and patient preference. A team approach including input from colleagues in radiation oncology is imperative to ensure proper patient selection and appropriate follow-up care.
Cryotherapy
Cryotherapy employs the use of liquid nitrogen to freeze lesions, causing direct thermal injury and ultimately destruction. Cryotherapy is a nonspecific destructive modality. Melanocytes are slightly more sensitive to cold injury than keratinocytes. Thus, it is possible to use cryotherapy in a way that allows regrowth of damaged keratinocytes but largely destroys melanocytes. For this reason, cryotherapy often leads to hypo- or depigmentation of treated areas. Used for various dermatologic conditions, its application in the treatment of LM remains controversial. Several protocols have been investigated with varying characteristics. In general, lesions are treated with two to three freeze–thaw cycles of between 15 and 60 seconds of liquid nitrogen application covering an area 0.5 to 1 cm beyond the visible clinical margins. Reported recurrence rates have been highly variable. Early studies reported recurrence rates of 6.6% at 3 years, while more recent studies have noted a recurrence rate of 34.3% at 5 years and of 0% with an average follow-up of just over 6 years. Varying recurrence rates may be attributed to differences in protocols and patient selection. High recurrence rates likely are related to the lack of histologic margin control and poor depth of penetration, leaving behind tumor nests near deeper epidermal appendages.
Because of the lack of consistency and definitive controlled studies within the literature regarding the efficacy of this modality, cryotherapy is not recommended as a preferred treatment for LM. Further investigation is necessary to develop proven protocols with long-term follow-up to ensure optimal patient care. Although this modality eventually may offer plastic surgeons an efficient, office-based treatment that can be provided under local anesthesia, several concerns remain. Among these concerns are excessive postoperative pain (like a burn), the need for postoperative wound care in a mostly elderly patient population, and poor aesthetic results, including development of keloids, hypertrophic scars, and loss of pigment.
Laser Therapy
The use of laser therapy within the practice of plastic surgery has expanded over the years. Many cosmetic and dermatologic problems are treated successfully with the use of lasers. This growing success, however, has not translated into the development of an effective laser-based treatment of LM. Throughout the literature, case reports and anecdotal evidence are present regarding the treatment of LM with laser therapy. Early reports used argon laser treatments, and as technology progressed, various other laser therapies have been attempted including carbon dioxide, Q-switched Nd:YAG, and Q-switched ruby laser therapy. More recently, attempts have been made using combination therapy such as Q-switched ruby and alexandrite lasers. Despite the many attempts, no ideal treatment has been identified, as recurrences have been seen with each of the various laser therapies used. Most published reports have placed recurrence rates around the 40% to 50% mark.
In light of the poor response rates to treatment and a body of evidence that lacks prospective controlled trials with long-term follow-up, laser therapy for LM cannot be recommended at this time. Perhaps as laser technology continues to move forward and knowledge of the histopathological basis of LM progresses, an effective laser based treatment will be identified.
Immunomodulation
Immune response modulators are a relatively new treatment for LM that has had promising early results. Already used to treat other conditions including basal cell carcinoma, squamous cell carcinoma, genital warts, actinic keratoses, and other forms of malignant melanoma, these agents are delivered either topically or via intralesional injection. The two commonly used immunomodulators are topical 5% imiquimod and injectable interferon alpha. Several small series and case reports have demonstrated early efficacy of intralesional interferon alpha ; however, more recent attention has been given to topical imiquimod.
Topical 5% imiquimod showed particularly promising early results in the literature as a solitary treatment agent and as preadjuvant therapy before surgical excision. As a primary treatment modality, several studies have shown impressive clinical and histologic response rates. In a study in 2003, Naylor and colleagues showed a 93% clinical and histologic clearance rate after 4 weeks of treatment, with 80% of patients having no recurrence at 1 year follow-up. Several other small series have shown similar results, with combined response rates in the literature estimated to be 81% for clinical clearance and 85% for histologic clearance. These early reports suggesting efficacy of imiquimod suffer from inadequate follow-up and reliance on clinical clearance as an indicator of histologic clearance. More recent studies have cast doubt on the reliability of the earlier reports. Among the other concerns remains the issue that some patients are complete nonresponders to therapy or respond clinically but not histologically to treatment. In a series reported by Fleming and colleagues in 2004, one patient had complete clinical response with resolution of the lesion’s pigmentation; however, histologically, there was no response to treatment. This scenario could prove extremely dangerous, as clinicians could mistake loss of pigmentation with resolution while the lesion itself remains intact. In another report by Cotter and colleagues in 2007, patients underwent staged surgical excision for histologic evaluation of margins after imiquimod therapy. After a full treatment course, 25% of patients were found to have residual disease, with one patient having invasive disease. Reported recurrence is also present within the literature and must remain a concern, as follow-up in published studies is relatively short.
Aside from questions relating to efficacy, treatment with imiquimod does have associated morbidity and adverse effects. Patients typically develop a striking inflammatory response to treatment with superficial crusting, which can be disconcerting and create an environment for infection. Patients also are required to continue treatment for 4 to 6 weeks or more depending on response.
The efficacy of treatment of LM with imiquimod remains unproven and represents an off-label use of this drug. Early results have been encouraging, but long-term follow-up of treated patients is necessary before this therapy can be recommended for use.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree




