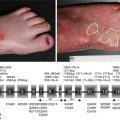
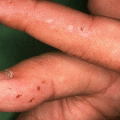
Acquired immunobullous diseases (AIBD)
Antigen mapping (AgM)
1. Level of split formation
Intraepidermal, junctional, dermolytic
Autoantibody mapping (AbM) = salt split skin test (SSST)
2. Location of binding of autoantibodies
(a) In vivo bound autoantibodies (direct SSST)
(b) In vitro binding of autoantibodies (indirect SSST)
Dermal, epidermal, or both sides of artificial (lamina lucida) split
EB hereditaria (EBH)
Antigen mapping (AgM)
1. Level of split formation
Intraepidermal, junctional, dermolytic
2. Expression of structural proteins
Normal, reduced, lacking
For adequate storage until further processing, the biopsy is placed in a tube without any liquid and transferred immediately to a freezer at −20 °C. If the biopsy is to be sent to a specialized IF laboratory, it has to be placed in a tube containing Michel’s medium and mailed at normal (room) temperature within a time limit of 1 month [6–9].
18.2.2 Blood Sample
A blood sample should be taken whenever possible to detect and classify circulating autoantibodies in AIBDs by routine IIF (detection on monkey/guinea pig esophagus or normal human skin, NHS), alternatively by other methods such as ELISA, immunoblot, or immunoprecipitation. In addition, a blood sample is necessary to perform the indirect salt split skin test (SSST = indirect AbM) on cryocut sections of NHS previously separated within the lamina lucida by incubation in 1 M sodium chloride [10, 11]. A 2–5 ml sample of whole blood (without any additives) is centrifuged for 15 min at 200 g, and the serum is stored in small aliquots in a freezer at −20 °C or can otherwise be sent (also frozen) to a specialized IF laboratory [5].
Likewise, according to the results of the AgM in EBH subtypes, a 2–5 ml EDTA blood sample should be taken for DNA preparation for mutation analysis. This sample also could be sent at room temperature to a reference laboratory. Alternatively and if possible, it is recommended to send extracted DNA.
18.2.3 Staining Procedure
The IFM — AbM or AgM — is technically performed according to the routine procedures for DIF and IIF as described in Chap. 16 [see also 1–5].
Basically in DIF, cryostat sections of the tissue sample are covered with an appropriate dilution of an antiserum (e.g., against immunoglobulins, complement factors, or fibrinogen) that is covalently bound to a fluorescent dye (most commonly fluorescein isothiocyanate, FITC), and the results are subsequently read with a fluorescence ultraviolet microscope at 450–490 nm (e.g., Axioskop, Carl Zeiss GmbH).
For IIF, cryostat sections from different animal or human tissues are incubated with patient serum suspected to contain circulating (auto)antibodies. The slides are then rinsed in PBS and incubated again with an antiserum (second antibody, e.g., immunoglobulin bound to FITC). Typically these second antibodies are directed against the (auto)antibody immunoglobulin that has bound in vitro in the first step.
The technique of AgM could be viewed as a combination of DIF and IIF: DIF by means of detecting a specific structure (protein, antigen) within the skin of the patient and IIF with regard to the method itself which is a two-step staining procedure. Cryostat sections of a patient biopsy — together with cryocuts of NHS that serve as positive control — are incubated with the respective first antibodies against specific structural proteins. Subsequently, staining of the sections is performed with different specific poly- or monoclonal second antibodies depending on the source of the first antibody.
The method of AbM — direct or indirect SSST — is performed on tissue sections previously separated in 1 M sodium chloride. The direct AbM (direct SSST, DIF) is applied on patient skin to localize in vivo bound autoantibodies within the artificial blister (epidermal or dermal). The indirect AbM (indirect SSST, IIF) shows the in vitro binding and location of circulating autoantibodies on a previously separated NHS.
18.3 Results
18.3.1 Immunofluorescence Mapping (IFM) for Acquired Immunobullous Diseases (AIBD)
In AIBDs, the IFM aids in the differential diagnosis of disorders such as the pemphigoid diseases and various subtypes of epidermolysis bullosa acquisita (EBA) or bullous systemic lupus erythematosus (BSLE).
The technique allows to specify the level of blister formation in AIBDs with the common linear staining pattern with antibodies to immunoglobulins and/or complement components (immune deposits) along the basement membrane zone (BMZ) by the conventional immunofluorescence method. Two major types of the method are applied (see Table 18.1).
With AgM, the basal lamina (lamina densa) in a biopsy of an in vivo blister is visualized by staining with antibodies directed against, for example, type IV collagen. The level of tissue separation is then assessed with reference to the location of the basal lamina, i.e., either below or above the lesional split (junctional or dermolytic).
In the absence of in vivo blistering, AbM of a patient skin specimen (clinically normal-appearing skin of, for example, the inner aspect of the upper arm — non-sun exposed!) is the diagnostic method of choice. By overnight incubation in 1 M NaCl, an artificial split formation is induced within the lamina lucida. The sample is devoid of secondary changes which may interfere with regular IFM and thereby modify the diagnosis. In the subsequent DIF test (direct AbM), in vivo bound autoantibodies are visualized either on the epidermal, dermal, or both side(s) of the artificial blister (i.e., on the blister roof and/or blister floor), whereas the basal lamina, as a reference in this assay, is constantly found by AgM on the dermal side, i.e., blister floor.
In addition to studies on patient’s skin, the same principle is used modified with an IIF test performed with patient’s serum (indirect AbM) on cryostat sections of salt-separated normal human skin (obtained during surgical procedures with informed consent!). This technique demonstrates the location of binding of circulating autoantibodies either to the epidermal (i.e., bullous pemphigoid, BP) or dermal (i.e., EBA) side of the artificially created blister. This is particularly helpful for making the diagnosis in patients where a biopsy is not available. Indirect SSST is also markedly more sensitive than routine IIF.
The staining profile of immune deposits in SSST provides essential information for the differential diagnosis between those subtypes of AIBDs which are characterized by autoantibodies directed against BMZ proteins (Table 18.2). For instance, it allows to differentiate unequivocally between BP (epidermal fluorescence above the basal lamina = lamina densa) and EBA (exclusive dermal fluorescence below the basal lamina) which clinically may appear to be identical.
Table 18.2
IFM profile of “subepidermal” AIBDs
AIBD | Autoantibody mapping profile |
|---|---|
Bullous pemphigoid | Linear epidermal (blister roof) fluorescence (IgG, IgA) |
Mucous membrane pemphigoid | Linear epidermal (target antigens: BP180, α[alpha]6/β[beta]4-intergin) and dermal (blister floor; target antigens: laminin 332, type VII collagen) fluorescence (IgG, IgA) |
Gestational pemphigoid | Linear epidermal fluorescence (IgG; C3 by complement binding test) |
Lichen planus pemphigoides | Linear epidermal fluorescence (IgG) |
Anti-laminin-γ[gamma]1 pemphigoid | Linear dermal fluorescence (IgG) |
Linear IgA dermatosis | Linear epidermal (target antigens: BP180 and LAD-1, BP230) and dermal (target antigens: type VII collagen) fluorescence (IgG, IgA) |
Epidermolysis bullosa acquisita | Linear dermal fluorescence (IgG, IgA) |
Bullous systemic lupus erythematosus | Linear dermal fluorescence (IgG, IgA, IgM) |
Epidermal fluorescence in SSST is a characteristic finding in several types of pemphigoid besides classical BP. Circulating autoantibodies binding to the dermal side (floor of the artificial blister) are seen in EBA, but also in BSLE, a subset of patients with mucous membrane pemphigoid and autoantibodies against laminin 332 within the lamina lucida, a bullous pemphigoid-like disease with autoantibodies to a 105 kDa lamina lucida glycoprotein unrelated to laminin 332, as well as anti-laminin-γ[gamma]1 pemphigoid [12–15]. Fluorescence on both – the epidermal and the dermal sides of the blister – is observed in mucous membrane pemphigoid and linear IgA dermatosis, partly reflecting the consequences of “epitope spreading.”
One major drawback of direct SSST on routinely obtained IF samples should be considered, i.e., that the induction of the artificial split means the original sample is lost for reinvestigation by routine DIF.
18.3.2 Immunofluorescence Mapping (IFM) for Hereditary Bullous Diseases (Epidermolysis Bullosa Hereditaria, EBH)
For a patient suspected to have one of the various subtypes of EBH, the immunofluorescence AgM technique supports to reach the primary diagnosis. It demonstrates (1) the level of split formation (intraepidermal, junctional, dermolytic) as well as (2) the expression of a specific structural protein in the skin (normal, reduced, or lacking; see Table 18.1).
The level of split formation can be determined on a cryosection of a lesional (blister) biopsy (see above). In addition, it is possible to gain information on the expression of specific structural proteins in the epidermis, the BMZ, or the dermis: in the latter situation, cryostat sections of a sample of normal-appearing skin (inner aspect of the upper arm) are incubated with different antibodies by IIF (Table 18.3). Taking the biopsy, attention has to be paid that the often minute and fragile epidermis is not kept in the punch instrument or gets lost during the final embedding procedure of the sample!
Table 18.3
List of antibodies and dilutions currently used for IFM of EBH in the laboratory of the Department of Dermatology, Paracelsus Medical University Salzburg, Austria, and respective EB subtypes
Dilution | Host | Company | Catalog no | EB subtype | |
|---|---|---|---|---|---|
First antibodies | |||||
Transglutaminase 5 | 1:20 | Mouse | Biomedical Tech. Inc | BT-621 | EBS |
Desmoplakin I + II | 1:10 | Mouse | Abcam | ab16434 | EBS
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|