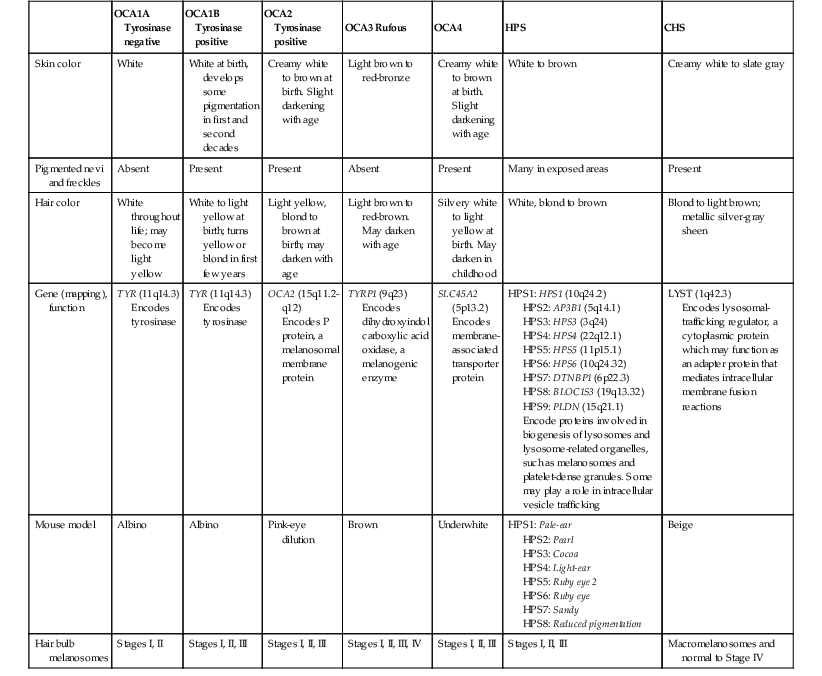
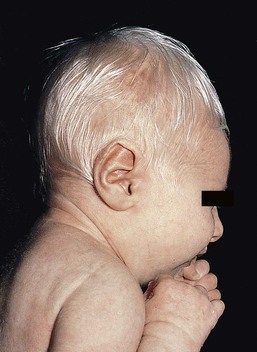
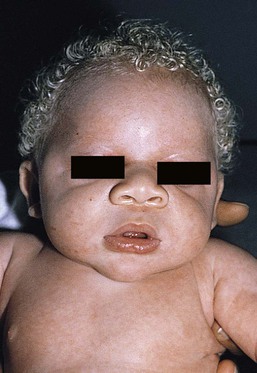
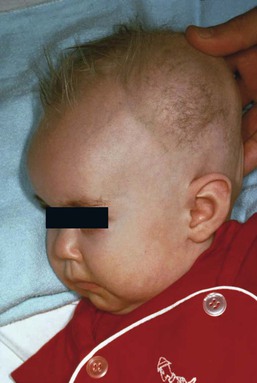
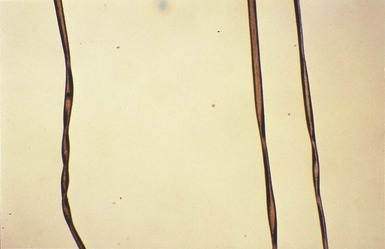
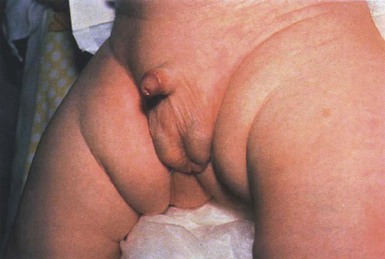
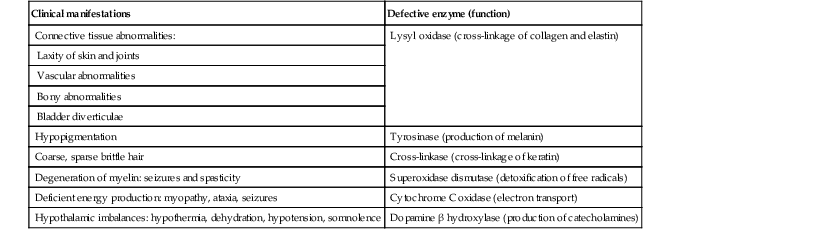
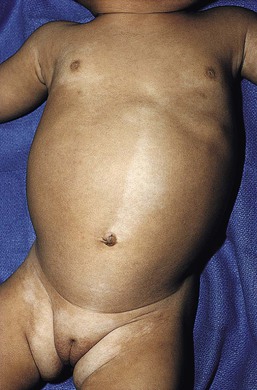
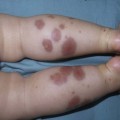
Yuin-Chew Chan, Yong-Kwang Tay A diverse group of conditions present with hypopigmentation in neonates and infants. A practical clinical approach would be to categorize them according to the distribution of the hypopigmentation: generalized, mosaic, or localized (Box 23.1). Some may be present at birth, but not noticed until later in infancy because neonates often have a lighter skin at birth than in later life. Any defect occurring in melanocyte development, melanin synthesis and transport, or distribution of melanosomes to keratinocytes can result in a hypopigmentary disorder. Melanocytes originate from the neural crest and are located in the epidermis, hair bulb, eye (choroid, ciliary body, iris), inner ear (cochlea) and central nervous system (leptomeninges). Melanin is synthesized in melanosomes, which are organelles that share characteristics with lysosomes. This chapter discusses a variety of clinical conditions causing hypopigmentation in neonates and infants, many of which have a genetic basis. Oculocutaneous albinism (OCA) refers to a group of autosomal recessive disorders involving abnormal melanin synthesis. Affected individuals have absent (type 1A) or reduced (type 1B, 2, 3, and 4) pigmentation in the skin, hair, and eyes from birth. The color varies from white to light brown, depending on ethnicity and the specific type of OCA. Ocular manifestations include nystagmus, photophobia, and decreased visual acuity. These are caused by decreased melanin within the eye or misrouting of optic nerve fibers. The affected infant is typically less pigmented than unaffected siblings. Historically, OCA was divided into two clinical types based on the presence or absence of tyrosinase, the rate-limiting enzyme in the melanin biosynthetic pathway.1 Advances in molecular genetics have given rise to a more accurate classification and better understanding of pathogenesis.2,3 In OCA, epidermal and follicular melanocytes are present in normal quantity and distribution but do not synthesize melanin adequately (Table 23.1). Oculocutaneous albinism type 1 (OCA1) is the second most common OCA worldwide. In OCA1A, there is marked generalized hypopigmentation at birth, with white hair and skin (Figs. 23.1, 23.2). As the child matures, the skin remains white. Nevi are not pigmented and sun tanning does not occur. The hair may acquire a slightly yellowish tint as a result of denaturing of hair keratins. In OCA1B, the variable decrease in tyrosinase activity results in several clinical phenotypes: yellow, minimal pigment, platinum, and temperature-sensitive. Individuals with OCA1B form pheomelanin, which requires less tyrosinase activity, and this results in some pigment production during the first two decades of life.4 Affected individuals have a similar appearance to those with OCA1A at birth, but with time develop some pigment. Hair color can change from white to light blond and even progress to light brown in adolescence. With sun exposure, some individuals with OCA1B may be able to tan, although it is more common to burn without tanning. Pigmented nevi and freckles may develop.5 An interesting subtype of OCA1B is the temperature-sensitive phenotype in which the tyrosinase activity is seen mainly on the extremities. In these patients, the enzyme has no activity at 37°C, but some activity at 35°C. These individuals have white or lightly pigmented hairs on the scalp and trunk (axillae, pubic area) and darkly pigmented hair peripherally (legs, arms). The pattern is similar to that observed in Siamese cats.4 In OCA1A, the irises are pale blue at birth and throughout life. In bright light, the entire iris can appear pink or red, which is caused by its translucency.5 Severe photophobia results from a lack of retinal pigment. Other ocular abnormalities include decreased visual acuity, nystagmus, and strabismus. In OCA1B, the irises can progressively darken to light tan or brown. In both subtypes, vision may remain stable or deteriorate with age. OCA1 is caused by loss-of-function mutations in the tyrosinase gene (TYR), which is mapped to chromosome 11q14.3. Tyrosinase is the key enzyme that catalyzes the first two steps in melanin synthesis, converting tyrosine to L-DOPA (L-3,4-dihyroxyphenylalanine), and L-DOPA to DOPAquinone. The OCA1A subtype is characterized by mutations that result in complete loss of tyrosinase activity, whereas OCA1B is caused by mutations that result in markedly reduced tyrosinase activity (5–10% of the normal level). Oculocutaneous albinism type 2 (OCA2) is the most common form of OCA. It is most prevalent in people of African descent.6 There is a spectrum of clinical phenotypes, depending on the ethnic background and the dilution of hair and skin pigment, which may be minimal to moderate. Comparison with a first-degree relative may be necessary to distinguish the degree of lightening.4 Most individuals are born with creamy white skin, and light yellow or blond hair. Depending on the individual’s ethnic background, hair may also be reddish blond or brown. Pigmented birthmarks may be present. With maturity, the amount of pigment in the skin and hair tends to increase. In sun-exposed areas, pigmented nevi and freckles can develop. The dilution of iris pigment may be mild to moderate. With age, the amount of pigment in the eyes tends to increase. Ocular manifestations are generally not as severe as those seen in OCA1A.2 Visual acuity and nystagmus tend to improve with age. OCA2 can be found in 1% of individuals with Prader–Willi and Angelman syndromes.6 OCA2 results from loss-of-function mutations of the OCA2 gene (previously called the P gene), which is mapped to chromosome 15q11.2–q13.7 The OCA2 gene encodes the P protein, a melanosomal membrane protein. The specific function of the P protein is currently not known, but is believed to be involved in tyrosine transport within the melanocyte, regulation of melanosome pH and tyrosinase processing and transport.8 The melanocytes of affected individuals are able to synthesize some melanin, but the majority is yellow pheomelanin rather than black-brown eumelanin. Prader–Willi syndrome involves deletions of the 15q region, including the OCA2 gene, on the paternally inherited copy of chromosome 15, whereas Angelman syndrome involves loss of the maternally inherited allele. Deletion of one copy of the OCA2 gene associated with a mutation in the second copy results in OCA2 in these patients.6 OCA3, previously called ‘rufous OCA’, is most commonly seen in people of African and Puerto Rican descent.9,10 At birth, individuals with this tyrosinase-positive OCA have light brown to red-brown skin and hair. With age, the hair becomes more pigmented. Mild sun tanning is possible. At birth, the irises are light brown and become more pigmented with age. Ocular manifestations are present, but less severe. Red reflex on transillumination of the iris and nystagmus are important clues to the diagnosis in dark-skinned people. OCA4 may be one of the most common types of OCA in Japan, with solute carrier family 45, member 2, SLC45A2 (previously called ‘membrane-associated transporter protein’, MATP) gene mutations found in 24% of 75 unrelated Japanese patients with OCA.13 Ocular manifestations include nystagmus, decreased iris pigment with iris translucency, reduced retinal pigment, foveal hypoplasia associated with reduction in visual acuity, and strabismus. OCA4 results from mutations in the SLC45A2 gene, which is mapped to chromosome 5p13.2. The gene encodes a melanosomal membrane protein that is likely to function as a transporter.13 The similar functions of the OCA2 and SLC45A2 genes may explain the phenotypic resemblance of OCA2 and OCA4. The diagnosis of OCA is usually made clinically and can be confirmed by DNA mutation analysis. Historically, the hair bulb incubator test for tyrosinase activity was used to differentiate between tyrosinase-positive and tyrosinase-negative OCA. In tyrosinase-negative albinism, there is the lack of pigment formation in hair bulbs when incubated with tyrosine, whereas in tyrosinase-positive albinism, pigment is produced. Prenatal diagnosis of OCA using DNA mutation analysis is available. OCA can be differentiated from other disorders with cutaneous and ocular albinism by the absence of neurological defects, immunodeficiency, and bleeding diathesis. No specific treatment is available for OCA. The importance of photoprotection, including sun avoidance, broad-spectrum sunscreen, protective eyewear and clothing, should be stressed to reduce the risk of photodamage and cutaneous malignancies. Early ophthalmologic evaluation and management is important. As squamous cell carcinomas and basal cell carcinomas have been known to develop in all types of OCA, yearly examination by a dermatologist is recommended. Hermansky–Pudlak syndrome (HPS) comprises nine genetically different autosomal recessive disorders characterized by tyrosinase-positive OCA, a bleeding diathesis, and a lysosomal ceroid storage disease affecting the viscera.16–18 The majority of individuals affected are of Puerto Rican or Dutch descent. HPS type 1 is the most common. The degree of pigmentary dilution in the skin and hair is highly variable. The color of the skin and hair ranges from white to brown. The degree of pigmentary dilution in the eyes is highly variable. Other ocular findings include nystagmus, reduced retinal pigment, and foveal hypoplasia with significant reduction in visual acuity. The nystagmus is most obvious during periods of fatigue or emotional change. The bleeding diathesis is caused by platelet storage pool deficiency and results in epistaxis, gingival, menstrual, colonic or post-surgical bleeding. Platelet numbers, prothrombin time, and partial thromboplastin time are normal but bleeding time is prolonged. The absence of dense bodies on electron microscopy of platelets is pathognomonic of HPS.19 Lysosomal ceroid accumulation can result in interstitial pulmonary fibrosis, granulomatous colitis, cardiomyopathy, and renal failure. These life-threatening complications usually develop in adulthood. Some patients with HPS type 2 have persistent neutropenia and suffer from recurrent bacterial infections.20,21 Most of the HPS-related genes encode proteins involved in the biogenesis of lysosome-related organelles. Ceroid is produced by degradation of lipids and glycoproteins within lysosomes. Ceroid accumulation in HPS suggests a defect in the elimination mechanisms of lysosomes.22 The diagnosis of HPS is made on clinical findings of oculocutaneous albinism, bleeding diathesis, and absence of dense bodies on electron microscopy of platelets. Molecular genetic testing of some HPS types, e.g. HPS1, HPS3, is available on a clinical basis. The differential diagnosis includes the Griscelli, Elejalde and Cross syndromes. Photoprotection is important, as patients have a predisposition to develop basal cell carcinoma and squamous cell carcinoma. An examination by a dermatologist should be performed annually. Patients should avoid aspirin and trauma to minimize the chance of a bleeding episode. Platelet transfusions may be considered prior to surgical procedures. Cigarette smoking should be avoided, as this reduces pulmonary function and may hasten progression of pulmonary fibrosis. Chediak–Higashi syndrome (CHS) is an autosomal recessive disorder characterized by OCA, immunodeficiency, progressive neurological deterioration, a mild bleeding tendency and abnormal inclusions present in a wide variety of cells. Compared with unaffected family members, the skin and the hair of affected individuals are lighter in color. Cutaneous pigmentation is often slightly to moderately decreased. The hair is blond to light brown, often with a silvery tint (Fig. 23.3). Recurrent skin and systemic pyogenic infections occur in early childhood. Cutaneous involvement usually manifests as a pyoderma, and there are a few reports of deeper involvement resembling pyoderma gangrenosum.23 Loss of ocular pigmentation results in a translucent iris and pale retina, leading to photophobia and an increased red reflex. Visual acuity is normal, but strabismus and nystagmus are common. Infections typically involve the skin, lungs, and upper respiratory tract. These intractable infections are often fatal before the age of 10 years. Common culprits include Staphylococcus aureus, Streptococcus pyogenes and S. pneumoniae. Periodontitis is an important manifestation of the immunologic dysfunction.24 Progressive neurologic deterioration with clumsiness, abnormal gait, paresthesias, and dysesthesias is often apparent later in childhood. Other neurologic abnormalities include peripheral and cranial neuropathies, spinocerebellar degeneration, ataxia, seizures, decreased deep tendon reflexes, cranial nerve palsies, and motor weakness.25–30 Most patients with CHS eventually develop a lymphoproliferative syndrome (‘accelerated phase’) characterized by fever, hepatosplenomegaly, lymphadenopathy, pancytopenia, bleeding, and generalized lymphohistiocytic infiltrates.26 Viral infections, particularly with the Epstein–Barr virus, have been implicated in causing the accelerated phase.31,32 CHS results from mutations in the lysosomal trafficking regulator gene (LYST) gene. The LYST gene (1q 42.1–q42.2) encodes a large cytoplasmic protein that appears to function as an adapter protein to mediate intracellular membrane fusion reactions.33 Natural killer (NK) cell function is drastically decreased. Diminished chemotaxis of granulocytes, monocytes, and lymphocytes has also been reported, as well as decreased antibody-dependent cytotoxicity and reduced suppressor T-cell function.34–36 The resulting susceptibility to infections is caused by the combination of these factors.37 The diagnostic hallmark of CHS is the finding of giant lysosomal granules within leukocytes, melanocytes, platelets, and other cells due to uncontrolled fusion of lysosomes. In bone marrow myeloid cells, the giant granules appear prominent. In melanocytes, giant melanosomes result from uncontrolled fusion of melanosomes, and this failure to disperse melanin to adjacent keratinocytes accounts for the decrease in pigmentation.38 On a cellular level, abnormal intracellular transport to and from the lysosomes has been detected.39 Giant granules within the phagocytic cells cannot discharge their lysosomal and peroxidative enzymes into phagocytic vacuoles.40 Prenatal diagnosis has been successfully performed using light microscopy by examining fetal hair shafts for characteristic clumping of melanosomes.41 Hematopoietic stem-cell transplantation (HSCT) is the only definitive treatment for this disorder.42 The NK cell defects and immunodeficiencies can be reversed, but the neurological deterioration and pigmentary dilution are not altered.43,44 Without allogenic HSCT, patients who develop an accelerated phase usually die in childhood, usually from pyogenic infections or hemorrhage.26 Patients who do not develop an accelerated phase tend to have fewer or no infections, but usually develop progressively debilitating neurologic manifestations.45,46 Supportive treatments in early infancy include antibiotics for infections, and intravenous γ-globulin. Nonsteroidal anti-inflammatory drugs can exacerbate the bleeding tendency and should be avoided. Ascorbic acid has been shown to partially correct the granulocytic function in some patients.34,35 Cross syndrome, or oculocerebral syndrome with hypopigmentation, is an oculocutaneous albinism associated with ocular anomalies, postnatal growth retardation, and neurological defects.47–49 The inheritance is probably autosomal recessive. The affected neonate has cutaneous generalized hypopigmentation and silvery hair. Ocular defects include microphthalmos, a small opaque cornea, and nystagmus. Neurological defects include mental retardation, ataxia, and spasticity. Treatment is supportive. Phenylketonuria (PKU) is an autosomal recessive disorder that results from the impaired conversion of phenylalanine to tyrosine, which is caused by the absence of hepatic phenylalanine hydroxylase (PAH) activity. The absence of this enzyme leads to a build-up of the amino acid phenylalanine and its byproducts in the bloodstream and spinal fluid. The incidence in the USA is estimated at 1 in 10 000 among Caucasians.50 It is most commonly observed in individuals of Scandinavian, Turkish and Irish descent, with males and females equally affected. PKU without treatment results in mental retardation and oculocutaneous pigment dilution. Most affected individuals have blond hair, blue eyes, fair skin, photosensitivity, a musty body odor, and neurologic disturbances.51 At birth, the neonate appears normal but may have a musty odor secondary to urinary and sweat phenylacetic acid or phenylacetaldehyde. Caucasian children with PKU almost invariably have blond hair, blue eyes, fair skin, and photosensitivity. African-American and Asian children tend to be lighter in color than their parents and unaffected siblings. The ability to tan is normal. Endogenous eczema often develops in these patients. In affected babies, serum phenylalanine levels begin to rise on the third or fourth day of life. Newborn screening with the Guthrie card bacterial inhibition assay test was implemented in the USA beginning in 1963, testing all newborns for PKU. Prenatal diagnosis is also possible by performing amniocentesis or chorionic villus sampling, with identification of the gene.51 Untreated PKU results in neurologic defects, including mental retardation, seizures, psychosis, hyperreflexia, and growth retardation. Phenylalanine hydroxylase deficiency is caused by mutations in the PAH gene (mapped to 12q23.2), with more than 400 different mutations identified so far.52,53 Hypotheses to account for the decrease in skin and hair pigmentation include a competitive inhibition of the binding of tyrosine to tyrosinase by excess phenylalanine or a decreased amount of tyrosine.54 With a low phenylalanine diet, the skin color, photosensitivity, odor, and eczema are reversible. Implementing a diet low in phenylalanine early in infancy can also dramatically reduce the mental retardation.51 Although children with treated PKU typically have a lower IQ than the general population, affected individuals can be expected to have a low-normal to normal intelligence if blood phenylalanine is maintained at a reasonable level in early childhood.55 Supplementation with tyrosine or tryptophan in the diet may be necessary. For women with PAH deficiency who are considering pregnancy, dietary restriction must be started before conception and continued throughout pregnancy.56 Aspartame, an artificial sweetener that contains phenylalanine, should be avoided. Griscelli first described this syndrome in 1978.57 It is a rare autosomal recessive syndrome that results in pigmentary dilution of the skin and hair, the presence of large clumps of pigment in hair shafts, and an accumulation of melanosomes in melanocytes. There are three types: • Type 1 (GS1): association of hypopigmentation and neurological abnormalities, due to mutations of the MYO5A gene (15q21.2)58,59 • Type 2 (GS2): association of hypopigmentation and immunological abnormalities, due to RAB27A gene (15q21.3) mutation60 • Type 3 (GS3): only hypopigmentation, due to melanophilin (MLPH) gene (2q37.3) mutation.61 In early childhood, individuals with all three types of GS have silvery gray hair, eyebrows, and eyelashes (findings that may also be present in the neonatal period), and skin hypopigmentation.62–65 Histologically, the hair shafts reveal uneven clumps of melanin, mainly in the medulla. Skin biopsy specimens reveal hyperpigmented oval melanocytes and poorly pigmented adjacent keratinocytes. On electron microscopic examination, epidermal melanocytes are found to contain perinuclear stage IV melanosomes. Adjacent keratinocytes contain only sparse melanosomes.66 Prenatal diagnosis of Griscelli syndrome has been accomplished by examination of hair from fetal scalp biopsies performed at 21 weeks’ gestation, with confirmatory postabortion examination of the fetus revealing silvery hair and identical microscopic findings.41 Neurological defects in GS1 include intracranial hypertension, cerebellar signs, encephalopathy, hemiparesis, peripheral facial palsy, spasticity, hypotonia, seizures, psychomotor retardation, and progressive neurologic deterioration.64–68 Immunological abnormalities in GS2 result in severe pyogenic infections, due to defective release of cytotoxic lysosomal contents from hematopoietic cells, and a hemophagocytic syndrome.66,69 There is combined T- and B-cell immunodeficiency. Frequent pyogenic infections, acute febrile episodes, neutropenia, and thrombocytopenia usually begin between 4 months of age and 4 years.57,64,65,67,70–72 The hemophagocytic syndrome is characterized by acute onset of uncontrolled lymphocyte and macrophage activation, resulting in infiltration and hemophagocytosis in multiple organs and death. Differentiation from Chediak–Higashi syndrome can be made by pathognomonic light and electron microscopic features. Griscelli syndrome lacks the large cytoplasmic inclusions and granulocyte abnormalities that are characteristic of Chediak–Higashi syndrome. Both diseases, however, are associated with an accelerated phase and carry a poor prognosis without bone marrow transplantation. Elejalde syndrome, also called neuroectodermal melanolysosomal disease, is a rare autosomal recessive disorder characterized by silvery hair, hypopigmented skin, severe central nervous system dysfunction, and abnormal intracytoplasmic inclusions in fibroblasts, histiocytes, and lymphocytes.76–78 Neonates have silvery hair and generalized hypopigmentation of the skin, which may develop a bronze color after sun exposure.79 In homozygotes, abnormal melanolysosomes are found in melanocytes and keratinocytes, cultured fibroblasts, and histiocytes of bone marrow. Extracutaneous features include mental retardation, hypotonic facies, plagiocephaly, nystagmus, diplopia, micrognathia, crowded teeth, a narrow high palate, pectus excavatum, and cryptorchidism. Neurological abnormalities range from severe hypotonia and the almost complete absence of movements, to seizures and spasticity. The age of onset of neurologic signs ranges from 1 month to 11 years.78 Several authors have suggested that subtypes of Elejalde syndrome and Griscelli syndrome type 1 are the same entity.80–83 However, the absence of immunologic defects allows Elejalde syndrome to be distinguished from the Griscelli syndrome type 1. Chediak–Higashi syndrome should also be considered in the differential diagnosis. Treatment is supportive and prognosis is poor. Classic Menkes disease is a multisystem disorder that manifests with hypopigmentation, hair abnormalities, failure to thrive, connective tissue changes, seizures, neurological degeneration, and death by the age of 3 years.84 The disorder has a prevalence of 1 in 250 000–350 000 live births.85 Most infants born with Menkes disease appear normal for the first few months of life before showing a rapid decline in growth and neurologic development. The cutaneous manifestations include alterations in hair, pigmentation, and elasticity of the skin.86 The scalp hair may appear normal at birth, but by about 3 months of age, it becomes sparse, light colored, lusterless, with a ‘steel wool’ quality. The hair is fragile and fractures easily, resulting in generalized alopecia (Fig. 23.4). Pili torti is the most common hair shaft abnormality (Fig. 23.5), demonstrating a flattened appearance under light microscopy with multiple twists of 180° around the long axis of the shaft.87 The twisted hairs result from excessive free sulfhydryl groups and a decrease in copper-dependent disulfide bonds. Cutaneous hypopigmentation is common, may be generalized or localized to the skin folds, and is caused by decreased tyrosinase, a copper-containing enzyme. Scaly dermatitis in a seborrheic distribution occurs frequently88 and transient neonatal erythroderma has been reported as an initial manifestation of Menkes disease.89 The skin is lax, and this doughy laxity is most prominent over the posterior neck, eyebrows, and leg folds (Fig. 23.6).86 Generalized puffiness of the cheeks and feet has been noted. Progressive neurodegeneration begins at about 2 months of age as a result of gliosis and demyelination of the cerebrum and cerebellum.84 Patients present with seizures, hypothermia, developmental retardation, spontaneous subdural hematomas, muscle hypotonia, and feeding difficulties.90 Urogenital problems include undescended testes, hydronephrosis, hydro-ureter, recurrent urinary tract infections, diverticula of the ureters and bladder, and rupture of the bladder. Chronic intractable diarrhea resulting in malnutrition88,91 and increased frequency of congenital heart defects are seen.92 Skeletal abnormalities are manifested as wormian bones of the skull and spurring of long bone metaphyses. Connective tissue changes are evidenced by loose joints and tortuous blood vessels, such as the carotid and cerebellar arteries, which may cause intracranial hemorrhages. This increased tortuosity is secondary to fragmentation of the internal elastic lamina of the arteries. Low copper and ceruloplasmin levels in the serum and high copper levels in cultured fibroblasts are useful in the diagnosis.93 Menkes disease can be considered a disorder of copper maldistribution. Menkes disease and occipital horn syndrome are rare, allelic, X-linked recessive copper deficiency disorders caused by mutations in the ATP7A gene (mapped to Xq21.1), which encodes a copper-transporting P-type ATPase involved in transport of copper to copper-requiring proteins.94,95 ATPase is localized in the trans-Golgi membrane of cells. The clinical features are due to malfunction of one or more copper-requiring enzymes, such as lysyl oxidase, tyrosinase, cytochrome C oxidase, and dopamine beta-hydroxylase, caused by the deficiency of the ATP7A protein (Table 23.2).86 Most patients are males, although a few female patients have also been reported. Most of the female patients have an X; autosome translocation, where the normal X-chromosome is preferentially inactivated.96 Occipital horn syndrome, formerly classified as Ehlers–Danlos syndrome type IX or X-linked cutis laxa, is now recognized as a milder form of Menkes disease and is caused by mutations in the same gene. Occipital horn syndrome is characterized primarily by connective tissue abnormalities, including skin laxity, hyperextensible joints, urinary tract diverticuli, hernias, and bony changes such as osteoporosis, arthrosis, and exostoses, such as the presence of a spike of ossification within the occipital insertion of the paraspinal muscles trapezius and sternocleidomastoid muscles at their attachments to the occipital bone (occipital horns), which gives the syndrome its name.97 Intelligence is normal or borderline, and patients can survive into adulthood. The milder phenotype results from the presence of low levels of functional ATP7A, unlike Menkes disease, in which no normal ATP7A activity exists.85,98 Daily subcutaneous administration of copper-histidine has been shown to be helpful in preventing the severe neurodegenerative problems in some patients with Menkes disease when the treatment is initiated early in life before the onset of significant neurological symptoms.99,100 This treatment, however, does not prevent the development of connective tissue problems, and cannot be regarded as a cure for Menkes disease.100–102 Mosaicism refers to the presence of two or more genetically distinct cell lines within an individual. These cell lines may be due to X-inactivation, as is normal in all human females, or to postzygotic somatic mutation. When mosaicism affects the skin, the affected skin may show patchy hypopigmentation or hyperpigmentation in a linear or segmental distribution (Figs. 23.7, 23.8). (Pigmentary mosaicism associated with hyperpigmented disorders is discussed in Chapter 24, and other mosaic conditions in Chapter 29.) Segmental hypopigmented lesions may be seen as an isolated cutaneous skin condition or as part of a genetic syndrome. The presence of mosaicism can sometimes be documented by the karyotyping of lymphocytes from peripheral blood or by genomic evaluation of both involved and uninvolved skin. In 1901, Blaschko characterized the distribution of segmental and linear skin abnormalities by examining patients with linear lesions and formulating a patterned composite diagram. He described these patterns as V-shaped or fountain-like over the spine, S-shaped or whorled on the anterior and lateral aspects of the trunk, and linear over the extremities (see Chapter 3). These lines should not be confused with dermatomes, which are the segments of skin that correspond to sensory innervation.103 Hypopigmentation that follows the lines of Blaschko and segmental patterns is thought to reflect cellular migration during embryogenesis affecting pigmentation.104 In 1952, a Japanese dermatologist named Ito described a 21-year-old woman with hypopigmented cutaneous whorls and streaks.105 As the distribution of the hypopigmentation was analogous to that of the hyperpigmented streaks observed in incontinentia pigmenti, he called the disorder ‘incontinentia pigmenti achromians’. To avoid confusion of these two unrelated entities, the preferred terminology later became ‘hypomelanosis of Ito’ (HI).106 HI is a descriptive term, rather than a diagnosis.107 It has been used for a phenotype with unilateral or bilateral hypopigmented streaks and whorls that follow the lines of Blaschko and which are present at birth or become apparent within the first 2 years of life (Figs 23.7, 23.8).108 There may be associated systemic findings. Recently, the terms ‘nevoid hypopigmentation,’ ‘mosaic hypopigmentation’ or ‘segmental pigmentary disorder’ with or without systemic anomalies, were adopted to better reflect the heterogeneous nature of this group of disorders. Hypopigmented whorls and streaks are distributed along the lines of Blaschko. They tend to be stable, although there are reported cases in which the pigmentary changes become more or less pronounced over time.109 In some cases, both hypopigmented and hyperpigmented streaks are evident. Wood’s lamp examination may help to determine the extent of the lesions in fair-skinned patients. Extracutaneous findings are variable, and include central nervous, musculoskeletal, and/or ocular abnormalities.110 Defects of teeth, hair, nails, and sweat glands, as well as aplasia cutis, fibromas, and generalized or focal hypertrichosis, have been reported.109,111,112 Additional abnormalities reported include limb-length discrepancies, facial hemiatrophy, scoliosis, sternal abnormalities, dysmorphic facies, genitourinary and cardiac anomalies. Nearly all of the defects are detectable by a thorough physical examination and regular follow-up. Infants should be observed for evidence of CNS involvement, reflected by developmental delay or seizures.109 Most children with CNS involvement manifest with neurological abnormalities before 2 years of age. Embryonic somatic mutations are the likely pathogenesis, with distribution and pattern of lesions determined by the type of progenitor cell affected and the timing of mutation during embryogenesis. Multiple chromosomal abnormalities have been associated with HI, and most cases are sporadic and have negligible risk of recurrence.106,111,113 On histologic examination, the hypopigmented areas have either normal or reduced numbers of melanocytes, and those melanocytes that are present demonstrate a reduction in the number of melanosomes.114 This includes nevus depigmentosus (also known as nevus achromicus) and Goltz syndrome. Patients with Goltz syndrome have both hyper- and hypopigmentation, as well as depressed areas of depigmentation following Blaschko’s lines. Cosmetic cover-up products can be used to conceal the hypopigmented areas but are usually not needed. The use of sunscreens can prevent or lessen the accentuation of pigmentary differences.109 Nevus depigmentosus (nevus achromicus) is an uncommon birthmark occurring in 0.4% of newborns.115 It is a well-circumscribed area of hypopigmentation with an off-white color that may occur as a small isolated (circular or rectangular) patch, or develop in a unilateral segmental distribution or follow the lines of Blaschko.116 Hair within a nevus depigmentosus may also be hypopigmented, and the margins may be irregular or serrated. The isolated form is the most common (Fig. 23.9) and the lesions do not usually cross the midline.117 The term depigmentosus is a misnomer because the lesions are actually hypopigmented, not completely depigmented, and become more prominent under Wood’s lamp. They are usually present at birth or become evident shortly thereafter, and remain stable in size and shape. Increase in size is in proportion to the growth of the child. Some lesions may appear during the first 3 years of life,117–119 with the trunk being the most commonly affected site.117,119,120 Occasionally, lesions may appear after the age of 3 years.117,120 In particular, the back and buttocks are most commonly affected, followed by the chest and abdomen.117 Males and females are affected equally and there is no distinct pattern of inheritance.116 The hypothesis is that during embryogenesis a clone of cells with a reduced melanogenic potential arises via a post-zygotic somatic mutation.103 Systemic manifestations are rare in patients with nevus depigmentosus. Neurologic abnormalities such as seizures and mental retardation have been reported,121 as well as ipsilateral hypertrophy of the extremities.122 In a survey of 50 patients with nevus depigmentosus, none had any extracutaneous features on examination.123 In two studies involving 29 patients with nevus depigmentosus, extracutaneous abnormalities were present in about 10% of the children.118,124 Occasionally, lentigines can develop within the achromic nevi, and this could be explained by the reversion of a mutation in one of the genes involved in pigmentation.125,126 There is a report of an infant developing multiple primary milia within a nevus depigmentosus.127 With dopa-staining, normal melanocytes are seen in nevus depigmentosus, and electron microscopic studies suggest a reduced synthesis of melanosomes and also a defect in their transfer to the keratinocytes, which could account for the hypopigmentation.128 Transfer of melanosomes from melanocytes to keratinocytes is essential for normal pigmentation. Other entities with which nevus depigmentosus is sometimes confused include nevus anemicus, segmental vitiligo, hypopigmented lesions of tuberous sclerosis, and hypomelanosis of Ito. The distinction between nevus depigmentosus and hypomelanosis of Ito may be artificial, as many patients with segmental hypopigmented macules also have linear pigmentary anomalies, similar to those seen in hypomelanosis of Ito, and underlying mosaicism is the common factor.109 Patients currently diagnosed with either of these conditions might simply be categorized as having nevoid hypopigmentation with or without extracutaneous anomalies. Genetic analysis may be considered in all patients with segmental or linear pigmentary abnormalities, and those with extracutaneous abnormalities to investigate cytogenic anomalies.109 There has been a report of spontaneous resolution of nevus depigmentosus after a 6-year period.129 One patient with a nevus depigmentosus had partial repigmentation following autologous melanocyte grafting.130 The use of noncultured epidermal cellular grafting has been reported to be successful in the treatment of nevus depigmentosus and a mottled repigmentation of 78% was observed.131 Repigmentation however, may not be permanent and recurrence of a successfully repigmented nevus depigmentosus 6 years after blister roof grafting has been noted.132 Twice weekly narrow-band UVB for 32 sessions was helpful in repigmenting a nevus depigmentosus lesion,133 as was the 308 nm excimer laser.134 Cosmetic camouflage may be helpful.
Hypopigmentation Disorders
Introduction
Generalized hypopigmentation of skin, hair, and eyes
Oculocutaneous albinism
Oculocutaneous albinism type 1
Cutaneous findings
Extracutaneous findings
Etiology and pathogenesis
Oculocutaneous albinism type 2
Cutaneous findings
Extracutaneous findings
Etiology and pathogenesis
Oculocutaneous albinism type 3
Cutaneous findings
Extracutaneous findings
Oculocutaneous albinism type 4
Extracutaneous findings
Etiology and pathogenesis
Differential diagnosis
Treatment and care
Hermansky–pudlak syndrome
Cutaneous findings
Extracutaneous findings
Etiology and pathogenesis
Differential diagnosis
Treatment and care
Chediak–higashi syndrome
Cutaneous findings
Extracutaneous findings
Etiology and pathogenesis
Treatment and care
Cross syndrome
Cutaneous findings
Extracutaneous findings
Treatment and care
Phenylketonuria (phenylalanine hydroxylase deficiency)
Cutaneous findings
Extracutaneous findings
Etiology and pathogenesis
Treatment and care
Generalized hypopigmentation involving skin and hair
Griscelli syndrome
Cutaneous findings
Extracutaneous findings
Differential diagnosis
Elejalde syndrome
Cutaneous findings
Extracutaneous findings
Differential diagnosis
Treatment and care
Menkes disease (and occipital horn syndrome)
Cutaneous findings
Extracutaneous findings
Etiology and pathogenesis
Treatment and care
Mosaic hypopigmentation
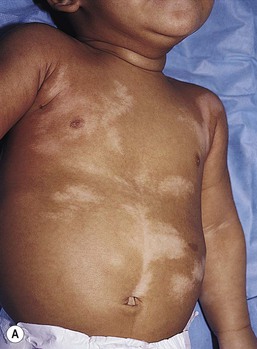
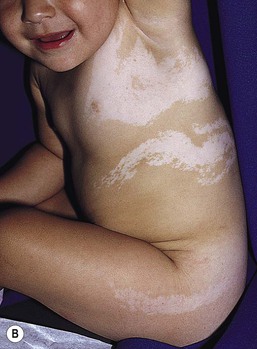
Nevoid hypopigmentation
Cutaneous findings
Extracutaneous findings
Etiology and pathogenesis
Differential diagnosis
Treatment and care
Nevus depigmentosus
Cutaneous findings
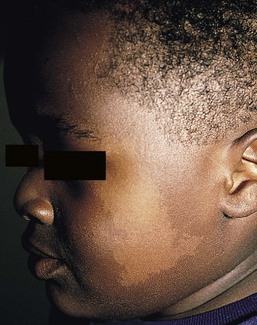
Extracutaneous findings
Etiology and pathogenesis
Differential diagnosis
Treatment and care
Localized hypopigmented disorders
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Hypopigmentation Disorders
23