Introduction
Gender confirmation surgery (GCS) for the male-to-female (MTF) individual fulfills a final, completive step in a process that, for most, has spanned years or decades. Completing this surgical journey is rate-limited historically by cost, by fears of complications, and by uncertainty over the final outcome. When the need to find anatomical wholeness overcomes these reservations, genital reassignment is chosen. Broadening of insurance coverage, reduction in complications, and clearer understanding of the anatomical details and outcomes have led to ever more transgender individuals seeking this major surgical option.
As chronicled in the recent film The Danish Girl , the first documented genital reassignment for an MTF individual took place in 1930 in pre-WWII Germany. At this time, the term transsexual had been coined by the famous sexologist Magnus Hirschfeld, who cared for and studied transgender individuals. The genital reassignment surgical process was the culmination of 2 decades of research and early scientific interest in the phenomenon of gender incongruence. Although the previously mentioned film’s subject, Lili Elbe, died of complications related to her surgical process, the door had been opened, and generations of gradual surgical innovations have followed. Wide publicity and attention was brought to the United States in the 1950s when Christine Jorgensen, a former GI, was the first American to undergo sex reassignment surgery in Europe. Later in the 1950s, the French gynecologic surgeon Georges Burou developed one of the versions of the modern penile inversion vaginoplasty technique. Modifications of this technique are still commonly used today.
Although consensus regarding a definitive surgical approach has not been reached among surgeons, there is broad agreement that hormonal and/or surgical treatment of individuals treated for gender dysphoria is of benefit to the individual in general. Documented cases of true regret are rare and usually are due to the difficulty of the transition process itself.
World Professional Association for Transgender Health Standards of Care
Patients who seek to undergo MTF GAS do so by following a general set of guidelines and recommendations established by the World Professional Association for Transgender Health (WPATH) in its standards of care (SOC). Currently in its seventh edition, the SOC are a series of flexible guidelines for clinical practice set forth by the society, based on evidence and expert consensus, and may be modified to meet the needs of an individual. The guidelines provide a framework for those individuals in the health-care community who provide assessment, mental and physical health care, hormone therapy, and surgery for transgender individuals.
These following criteria are recommended by WPATH for genital surgery: (1) well-documented, persistent gender dysphoria, (2) two letters of referral from mental health professionals well versed in the care of transgender patients, (3) capacity of the patient to engage in informed decision making and consent, (4) well-controlled comorbid medical and mental health conditions, (5) 12 continuous months of hormone therapy, and (6) 12 continuous months of experience living in the gender role consistent with the patient’s gender identity. In addition, regular follow-up with a mental health or medical care provider is recommended.
In general, individuals who seek to undergo genital reassignment do so after living at least 1 year in their desired gender role. They are expected to have completed a process of social transition which makes the surgical transition more of a formality than a singular process that defines gender. Instead, it is an affirmation of a role already well adopted.
Preoperative Planning and Care of Patients Undergoing Vaginoplasty
The overall approach to transgender care should be undertaken in a multidisciplinary approach when possible, incorporating input from mental health and medical health-care professionals to ensure that patients are and remain appropriate candidates for surgery. Surgical consultation prior to vaginoplasty should involve an extensive discussion between the surgeon and patient on the individual’s history and rationale for surgery, as well as extensive counseling on the types or routes of surgeries available, the risks and benefits related to surgery, and the limitations and expected outcome of the surgery.
Permanent depilatory procedures (e.g., electrolysis or laser hair removal) are recommended (or required by some surgeons) for patients undergoing vaginoplasty via the penile inversion technique, which involves use of a peno-scrotal graft to line the neovaginal cavity. Preoperative depilation optimizes surgical results by preventing future hair growth in the neovagina. Hair removal is recommended on the scrotum, perineum, and proximal penile shaft, all of which are often used to create the graft. Laser hair removal uses a laser light source to target pigmented hair, destroying the follicle with heat while electrolysis destroys the hair follicle through the use of an electric current directed through a needle or probe, and can be used on pigmented or unpigmented hair. These procedures may be uncomfortable, and lidocaine jelly or lidocaine/prilocaine cream may be prescribed to reduce the pain associated with the procedure. For optimal results, several treatments are necessary, and hair removal should be stopped 4 to 6 weeks prior to a scheduled surgery.
Several factors place MTF patients seeking vaginoplasty at risk for venous thromboembolic events (VTE) during the perioperative period. These factors include the use of high-dose exogenous estrogen, a prolonged surgical procedure, and need for postoperative bed rest and decreased mobility. Most surgeons request that patients stop their estrogen regimen 2 to 6 weeks prior to surgery, and allow patients to restart it once they have returned to light activity, generally 1 to 4 weeks after surgery. Some surgeons do not require complete cessation and only ask that their patients modify their estrogen use to a low-dose regimen perioperatively. This may lower the emotional angst and physiologic changes that some patients experience as a result of complete cessation. Patients should receive appropriate VTE prophylaxis in the perioperative and postoperative periods.
Guidelines for prophylactic antibiotic use do not currently exist for vaginoplasty in the transgender population, and therefore appropriate prophylaxis should be administered for urogenital procedures. The specific antibiotic selection and length of administration is based on expert opinion. Many practices use intravenous second-generation cephalosporins such as cefazolin. Some surgeons also choose to provide patients with postoperative prophylaxis to prevent urinary tract infection while an indwelling Foley catheter and vaginal packing are in place.
The use of a mechanical bowel preparation prior to penile inversion vaginoplasty is surgeon dependent. Many experts may choose to perform some degree of modified bowel preparation prior to surgery to avoid fecal contamination in the rare event of a rectal injury, and to delay the immediate return of postoperative bowel movements while a vaginal packing is in place. Modified bowel preparation regimens may involve a combination of clear liquid diet the day prior to surgery and mechanical bowel preparation or enemas. Rectal wash can be performed at the time of surgery with povidone/iodine solution. For intestinal segment vaginoplasty, full mechanical bowel preparation and oral antibiotics are recommended to reduce the risk of surgical site infection and anastomotic leak.
Vaginoplasty: Techniques and Special Considerations
Standardization and consensus among surgeons is rare and, until recently, uncommonly shared. The surgical process itself has remained largely shrouded by proprietary and guarded thinking among surgeons. Patients are wise to compare outcomes between surgeons, communicate with other patients, and in general, act as their own patient advocates in choosing a surgeon.
Penile inversion vaginoplasty
The most commonly performed technique for primary MTF transgender vaginoplasty is the penile inversion vaginoplasty. The technique has been widely researched and described, and modifications have been made over time to improve aesthetic outcome and functional results. Penile inversion vaginoplasty is irreversible and generally includes orchiectomy (if not previously performed), penile deconstruction, formation of a sensate neoclitoris from a portion of the glans penis on its dorsal neurovascular pedicle, creation of a neourethral meatus, creation of a vaginal cavity and lining of the neovagina with local peno-scrotal skin flaps, and labiaplasty to create an aesthetic and feminine external appearance of the genitalia. Surgery may be performed by a variety of surgical specialists including plastic surgery, urology, gynecology, and urogynecology.
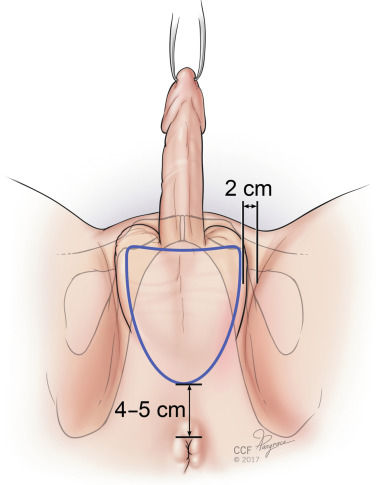
The procedure is performed once the patient is positioned in lithotomy, prepped, and draped. To start the procedure, the scrotal flap, which will later be used to line the neovagina, is marked. Surgeons mark out this graft differently. Some refer to it as “butterfly markings,” while other graft markings are more oval ( Fig. 10.1 ). It is imperative that the markings be symmetric with the principle of symmetry maintained throughout the surgery. The flap is incised sharply, then excised from the underlying subcutaneous tissue using electrosurgery. To prepare the graft, all excess subcutaneous tissue is removed sharply. If hair removal is suboptimal, remaining hair follicles can be electrosurgically coagulated. The graft is kept moist with saline soaked sponges.
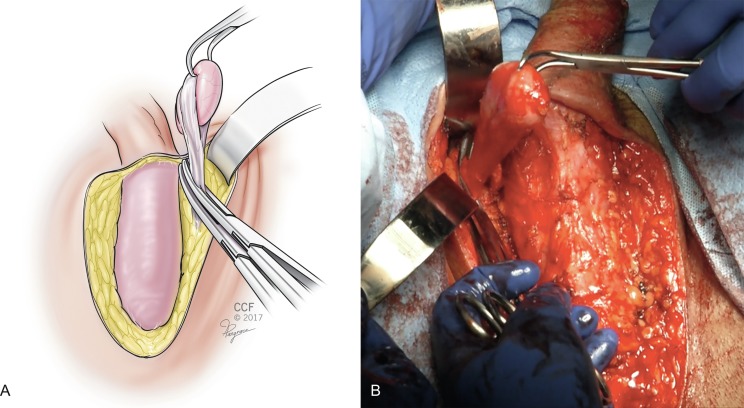
Next, a bilateral orchiectomy is performed by skeletonizing the spermatic cord and isolating the gonadal vessels, which are cross clamped and suture ligated at the level of the external inguinal ring ( Fig. 10.2 A and B ). Excess subcutaneous tissue is then either pinned laterally or removed, depending on how bulky it is, and the penile structures down to the deep fascia (Buck’s fascia) are exposed ( Fig. 10.3 ). A circumferential incision is made around the glans of the penis, freeing the epithelium from the underlying tunica albuginea. The penile structures are then sharply de-epithelized, leaving a penile tube ( Fig. 10.4 A and B ). The suspensory ligament of the penis is released. A urinary catheter is placed to drain the bladder.
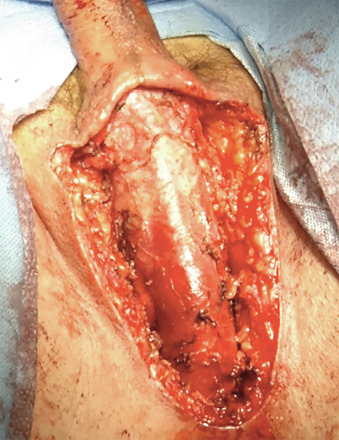
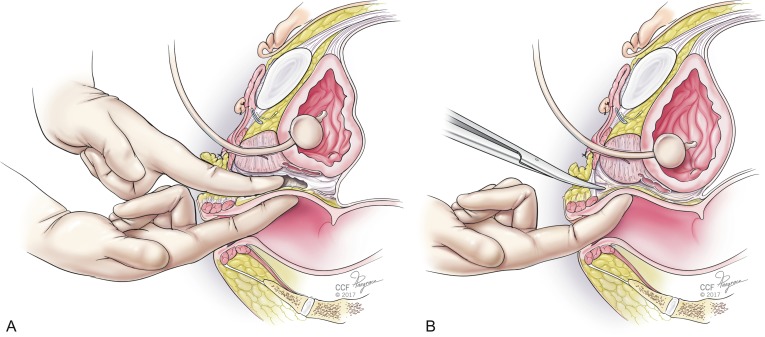
Attention is then turned to creation of the neovagina. In our opinion, the perineal dissection to create the neovaginal cavity is the most technically difficult portion of the procedure. Injuries to the rectum, urethra, or bladder are possible with eventual fistula formation if this step of the surgery is not skillfully performed. Avoiding rectal injury is the most crucial goal. Proper positioning, lighting, and assistance are key to avoiding this. There are three basic landmarks that are traversed in the perineal dissection, and the first 4 cm are the most crucial. Injuries to the rectum in this area are most common, and because of the high-pressure status of the rectum and relative lack of repairable layers in this area, these injuries are the most likely to fistulize. A transverse incision is made in the perineum below the bulbous urethra, transecting the perineal tendon (aponeurosis of the bulbospongiosus muscles) and creating a cavity by separating the levator ani muscles laterally. The midline central tendon is a constant and is a gateway to safety if the dissection remains as anterior as possible once the urethra is safely palpated and protected by retraction. The cathertheterized urethra is palpable and allows progress superiorly until the prostate capsule is entered. Once the prostate capsule has been entered, the risk of rectal injury is markedly lowered. Sharp dissection is then performed through the lower pole of the prostate until Denonvilliers fascia is reached, always with the urethra intermittently palpated behind the retractor. Once the bilateral superior prostatic arteries are encountered near the superior aspect of the prostate capsule, blunt dissection can normally complete the dissection. Dissection can be done with or without a finger in the rectum, depending on the surgeon’s preference and choice ( Fig. 10.5 A and B ). Achieving a depth of at least 15 cm is ideal. Once adequate caliber and depth are achieved, meticulous hemostasis is obtained to avoid hematoma formation. A bubble test may be performed to assess for rectal injury by filling the neovagina with irrigation solution and then insufflating the rectum using a large syringe. If bubbles are seen in the neovaginal cavity, further investigation for rectal injury should be performed. Once no injury is confirmed and hemostasis is satisfactory, the neovagina is packed.
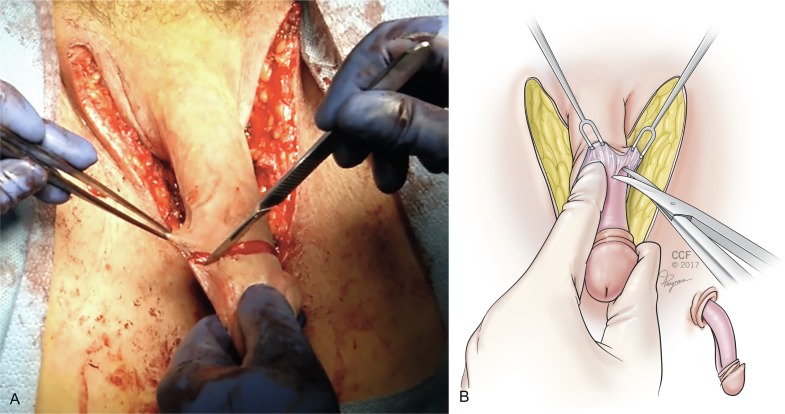
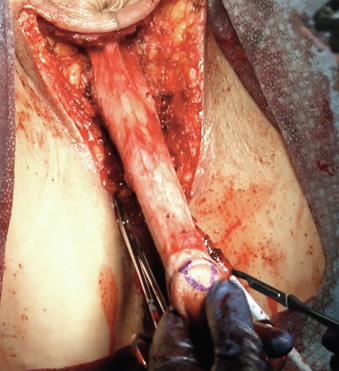
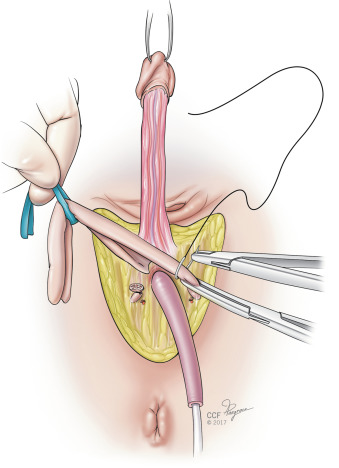
Attention is returned to the penile structures. The clitoris is marked in a triangular or heart-shaped fashion on its dorsal tip ( Fig. 10.6 ). A dorsal flap containing the dorsal penile neurovascular blood supply to the neoclitoris (created from a portion of the dorsal glans penis) and a ventral urethral flap are created. The ventral corpora cavernosa are divided with electrocautery from the symphysis to the distal glans, and the ventral portions are discarded ( Fig. 10.7 ). Erectile tissue from the ventral sheath is aggressively removed with scissors or electrocautery and discarded. Some surgeons dissect the neurovascular structures completely along their length, discarding the tunica—the chief advantage being the lack of bulk and erectile tissue potentially left behind, with perhaps greater risk of clitoral necrosis if there is any compromise to the blood supply.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree




