Acknowledgments
This chapter is supported by Ministry of Science and Technical Development, Republic of Serbia, Project No. 175048.
Introduction
Patients suffering from gender dysphoria feel that their gender assigned at birth is in some way “wrong” and therefore seek to take the opposite gender role. Gender dysphoria is a state that requires a multidisciplinary approach to treatment, and the only real management consists of “adjusting the body to the mind.” A multidisciplinary approach includes psychiatric evaluation and ongoing care, hormonal therapy, and physical transition with gender affirmation surgery (also known as genital confirmation or sex reassignment surgery [SRS]), as the last possible step in treatment. In terms of readiness and eligibility for the surgery, it is generally recommended that surgeons and patients follow the criteria set forth by the World Professional Association for Transgender Health (WPATH). The Standards of Care advise that patients undergoing genital surgery should provide two letters of recommendation from qualified mental health professionals, provide confirmation that they have been on hormonal therapy for a period of 1 year minimum, and that the patient should be living full-time as their self-affirmed gender for that year as well. Gender reassignment surgery (GRS) refers to all surgical procedures that a patient wishes to undergo in an effort to become similar to the opposite gender. SRS is a part of GRS and refers only to the genital reconstruction.
When choosing to undergo genital confirmation surgery, the patient must bear in mind the desired postoperative result that they wish to achieve and be knowledgeable about the surgical options that are available to them. This is the point where the patient must undergo a thorough preoperative consultation and examination by the surgeon performing the surgery, as well as a detailed discussion with their referring mental health specialist to help them set realistic expectations and prepare for the postoperative recovery after surgery. Along these lines, it is important to preface that there is no single ideal surgical package that will suit every patient. We need to consider the patient’s anatomy as well as their personal concerns. This is why the decision to choose a particular surgical package rests not only with the surgeon, but with the patient as well.
The most commonly performed surgeries in female-to-male (FTM) patients are bilateral mastectomy with male chest contouring and genital reconstructive surgery which includes total hysterectomy with bilateral oophorectomy, vaginectomy, reconstruction of the neophallus, urethral reconstruction, and scrotoplasty with implantation of testicular prostheses. Phalloplasty and metoidioplasty are the two options for neophallic reconstruction and we will discuss these procedures in depth in this chapter.
History and Evolution of the Procedure
The goal of penile reconstruction has always been to create a neophallus of satisfactory appearance. The term “phalloplasty” was first mentioned back in the 19th century by Sprengler and referred to the reconstruction of the superficial penile tissue layers. Bogoras first reported the usage of a single abdominal tube for phalloplasty, naming his technique “penis plastic totalis.” Hoopes was one of the first surgeons to describe his phalloplasty technique using a simple abdominal tube pedicle flap to create the phallus with no urethral extension. He operated on the premise that most patients were strongly motivated by the “anxiety of discovery” and that functionality was not the primary goal of the surgery.
Conversely, Gilbert et al. believed in the functionality of the neophallus, and together with other authors defined the goals that should be met in penile reconstruction :
- 1.
One stage procedure
- 2.
Aesthetically appealing phallus with a normal appearance and adequate length and girth for sexual intercourse (after penile prosthesis implantation)
- 3.
Voiding while standing up with the urethral meatus opening at the tip of the phallus
- 4.
The phallus possessing adequate tactile and erogenous sensation
- 5.
Minimal scaring of the donor area or disfigurement
- 6.
The phallus reconstruction should preferably be reproducible.
From the experience of severely injured patients during the Second World War, new ideas and possibilities for phalloplasty arose. The first FTM GRS was performed in 1946 by Sir Harrold Gillies. This operation involved a pedicled groin flap with creation of a neourethra and subsequent implantation of rib cartilage into the penile shaft and became known as the Gillies’ technique and remained in common practice for years to come. The technique was later modified by Biber and later by Laub et al. from Stanford University to incorporate a neourethral reconstruction using an outside-in tubularized infraumbilical abdominal flap. Around this time, Bouman, who performed genital reconstruction in intersex patients, described the use of biaxial superficial inferior epigastric midline infraumbilical flaps for neophallic reconstruction in his FTM patients.
In the early 1970s, McGregor and Jackson introduced the use of the groin flap based off of the superficial circumflex iliac artery and vein as a single pedicled flap. In 1978, Puckett similarly described a tubed groin flap for penile reconstruction, which had successful outcomes reported by many surgeons at that time. In Serbia, the groin flap technique was modified by extending the flap and lengthening the pedicle which led to very successful phallic reconstruction outcomes in adolescents and children.
The gracilis flap was also described as a possible option for phallic reconstruction, and Orticochea was the first to report success with its use in a five-stage neophallic reconstruction which resulted in a neophallus of normal size, color, micturition, with normal tactile, thermal, pain and erogenous sensitivity, and the ability for normal erections. Horton et al. also reported their experience with unilateral gracilis flaps in phallic reconstruction.
The radial forearm free flap (RFFF) was initially developed in 1978 by Chinese postgraduate doctors at Shenyang Military Hospital. They used the radial forearm flap as a free flap vascularized by the radial artery for the correction of severe neck burn scar contractures. Later in 1982, their teacher, Song et al., reported this flap in the English literature, naming it the “forearm flap.” Chang and Hwang first described the use of the radial forearm flap in single-stage phalloplasty surgery incorporating an autogenous cartilage stiffener in seven cases with satisfactory results and only one case of fistula. After their publication, it became known as the “Chinese flap.” Biemer later described a modification in which the neourethra was centered over the radial artery to provide it with the best possible perfusion. Semple placed the neourethra centrally over the vascular pedicle, reducing the width of the forearm flap, but resulting in a considerable reduction in neophallic length and a suboptimal donor site scar. Gilbert et al. described a modification to Biemer’s modification of the RFFF by centering the urethra on the ulnar forearm and allowing for creation of the glans by widening the “cricket bat” design. Gottlieb proposed a design incorporating a centrally placed neourethra in continuity with the neoglans, eliminating the need for a circumferential meatal suture line, thus avoiding meatal stenosis without sacrificing phallic length. Another modification of the RFFF was the introduction of the radial bone into the phallus as a stiffener. Unfortunately, this modification resulted in a high complication rate including urethral fistula formation (40% to 50%) and need for postoperative urethral reconstruction as well as fractures of the donor forearm in about 9% of the patients. While placement of this type of stiffener seems to be associated with a high complication rate, patients who do not experience complications seem to be satisfied. In recent studies looking at neophallus reconstruction with the use of an osteocutaneous RFFF, 70% of the patients report good to excellent stiffener function during intercourse. Currently, forearm flaps are one of the most popular methods for total penile reconstruction. The majority of centers performing the phallic reconstruction with RFFF are using penile implants to create phallus rigidity to allow for penetrative sexual intercourse.
In 1987, Upton reported the use of a lateral arm flap based on the posterior radial collateral artery for phallic reconstruction, which allowed for an inconspicuous donor site without compromise of the size of the neophallus. The technique includes a prefabricated neourethra which is positioned within the lateral arm to permit the coexistence of an erectile prosthesis alongside a fully vascularized urethra. Khouri et al. later reported 5-year outcomes after phalloplasty using the prefabricated lateral arm flap technique, and all patients were found to have erogenous and tactile sensibility of the neophallus.
The lateral thigh flap was first described by Baek in 1983. This is a fascial flap based on the smaller vessels that extend from the profunda femoris system to the skin. The anterolateral thigh (ALT) flap, first described in 1984 by Song et al., is a fasciocutaneous flap usually based on the musculocutaneous and septocutaneous perforators of the descending branch of the lateral circumflex femoral artery and its venae comitans. As a free flap, ALT was first mentioned by Felici and Felici in 2006, who reported their results in six FTM patients with the possibility of insertion of a penile prosthesis for penetrative sexual intercourse.
Outcomes from the pedicled latissimus dorsi flap were reported in the late 19th century in breast reconstruction following mastectomy. It was first described as a free flap in 1976 by Baudet et al. In 2006, our center in Belgrade reported on its use for neophallic reconstruction in eight boys with a diagnosis of micropenis as a result of exstrophy-epispadias complex. The latissimus dorsi flap has been widely used in reconstructive surgery because of its versatility and reliability. The free flap is used with great success in both adult and pediatric populations because it allows for the creation of a neophallus of adequate length and circumference.
Above we describe the most commonly used flaps. Many other free flaps have been described for phallus reconstruction, including the free deltoid flap, scapular free flap, sensate osteocutaneous free fibula flap, tensor fasciae latae, deep epigastric artery perforator (DIEAP) flap, and the dorsalis pedis flap.
As the above neophalloplastic techniques were being developed, an alternative to phallic reconstruction using the hormonally enlarged clitoris was also under study. This procedure was first reported by Durfee and Rowland. The term metoidioplasty, as the technique is known today, was introduced by Lebovic and Laub and is derived from Greek words “meta”—“toward” and “oidion”— “male genitalia.” Eicher later introduced the term “clitoral penoid” which actually refers to the same surgical procedure. Metoidioplasty involves the creation of a small penis, inadequate for penetration during intercourse, but otherwise involving all the phenotypic characteristics of male genitalia. The first big series of patients (70 FTM patients) undergoing this procedure was published by Hage and Turnhout and revealed that more than two surgical procedures were needed to obtain successful outcomes after surgery.
There are clearly many techniques for penile reconstruction and no true gold standard exists. This emphasizes the point that all techniques are fraught with disadvantages. In this chapter, we will emphasize the most commonly used surgical techniques for genital confirmation in FTM patients and we will elaborate on patient eligibility and readiness criteria for each procedure.
Phalloplasty Surgery: Current Operative Techniques
Phalloplasty involves creating a neophallus from extragenital tissue and is considered one of the most challenging and complex procedures in reconstructive surgery. Phallic reconstruction should ideally create an aesthetically pleasing phallus with sufficient length for vaginal penetration, with tactile and erogenous sensibility, with the ability to void in standing position, and with acceptable donor site morbidity.
Since total penile reconstruction was first described, there have been ongoing endeavors to develop the ideal technique for phalloplasty. Advancements in phalloplastic techniques have mirrored the improvements in reconstructive flap surgery. First experiences involved the use of local flaps, most commonly based off of the inferior epigastric vessels. In the 1980s microsurgical free flaps became very popular in reconstructive surgery, and these techniques are also applied to SRS phalloplasty surgeries. Today, there are many options for phalloplasty, including pedicled flaps (abdominal flap, groin flap, ALT flap) and free transfer flaps (RFFF and the latissimus dorsi flap). The most commonly used flaps in FTM patients are the RFFF and the free musculocutaneous latissimus dorsi (MLD) flap.
Abdominal flap phalloplasty
In this procedure, the donor site is located on the abdomen or waist. A “bird wing” incision is marked with its base in the suprapubic/mons pubic position with lateral extensions up to the lower abdominal skin crease, extending to both flanks. The base-to-limb ratio of the flaps is kept at 4 or 5:1, so that adequate blood supply is ensured to the most distal end. The depth of the incision reaches up to the anterior rectus sheath and the external oblique aponeurosis, from medial to lateral, with blood supply provided by superficial epigastric and circumflex iliac vessels. In abdominal “flap-apposition” and phalloplasty, the two lateral “wings” are approximated in the midline using subcuticular sutures. The clitoris is usually left in its native position to allow for sexual sensation. Urethral reconstruction cannot be performed in the first stage, but it is performed in one of the later stages. In cases where erectile function is desired, a penile prosthesis can be implanted.
Recently Bettocchi et al. published their technique of the pedicled pubic phalloplasty with the neourethra reconstructed from the hairless strip of skin from the clitoris and labia in 85 FTM patients. The authors reported complications in 75% of the patients related to urethral reconstruction, out of which 64% had urethral stricture and 55% had urethral fistula. They also reported that in cases where a two-staged urethroplasty was performed, there was a decrease in the overall complication rate.
The main advantage of the abdominal flap phalloplasty is that it is a relatively easy procedure to perform, with acceptable scaring of the donor area and of the neophallus. The main disadvantage of the procedure is the creation of a relatively small neophallus with poor erotic sensation and undesirable hair growth.
Groin flap phalloplasty
The base of this flap lies over the femoral artery approximately 2 cm below the inguinal ligament. The flap is then designed to be approximately 22 × 12 cm. The surface of the flap is outlined in three parts: (1) the lateral thin hairless part (approximately 2 cm) for the neourethra; (2) the thick, partially hairy medial part for the neophallus shaft; and (3) the base of the flap (approximately 4 to 5 cm) which is to be de-epithelialized. The flap is elevated beneath the deep fascia, leaving out the rectus or the external oblique fascia, starting from its distal part and proceeding to its base. Dissection is performed medially and the sartorius fascia is lifted together with the deep branch of the superficial iliac circumflex artery. The base of the flap is then mobilized from the lower and medial margin by blunt dissection of the subcutaneous tissue.
De-epithelization is performed at about 4 to 5 cm from the base of the flap, excluding approximately 2 cm of the lateral part, which is to be used for the neourethra. About 2 cm of the lateral part of the flap is separated from the medial part by de-epithelializing a 1 cm margin of skin which is then tubularized over a stent, then sutured with running and interrupted absorbable sutures. The medial part is tubularized over the newly formed urethra in a tube-within-a-tube fashion. After the flap is elevated with a long pedicle, a subcutaneous tunnel measuring 3 fingerbreadths in width is created between the donor and recipient site and the arch of the pedicle is rotated between 90 and 180 degrees without compromising the blood supply of the flap. The neophallus is sutured to the skin of the recipient region and the neourethra is anastomosed to the native urethra after it has been lengthened using the labia minora, while the clitoris is incorporated into the base of the neophallus. Urethral anastomosis may be performed simultaneously, or in a second procedure, following a complete formation of the neourethra. The donor site skin defect can be easily closed by undermining and direct approximation.
Advantages of the groin flap phalloplasty are that it is a relatively short procedure to perform, with a satisfying postoperative result for the patient and acceptable morbidity and scarring of the donor area. Disadvantages are similar to those of the abdominal phalloplasty: poor erogenous sensation, smaller and usually hairy neophallus. In our experience, complications occurred in 5 out of the 24 patients who underwent this procedure. All of these complications were resolved with a minor surgical repair (two urethral fistulas, one stenosis of the urethral anastomosis, and two patients with partial flap necrosis).
Anterolateral thigh flap phalloplasty
The mobilization of this flap is performed medial to lateral, with careful mobilization anteriorly between the rectus femoris and vastus lateralis muscles to preserve the perforators through the vastus lateralis muscle. The ALT flap has a rectangular shape with one side shorter and wider and the other longer and narrower, for the neophallus and neourethra, respectively. After the major perforator vessel is identified, pedicle dissection goes through the septum to the descending branch of the lateral circumflex femoral artery. The flap is then marked eccentrically to obtain the maximal pedicle length possible. The lateral cutaneous nerve of the thigh is identified, mobilized, and dissected at about 5 cm proximal to the superior edge of the flap, to preserve an adequate length for the neurorrhaphy part of the procedure.
The main advantage of the ALT phalloplasty is the avoidance of a microvascular anastomosis, good sensation of the neophallus due to the anastomosis of the lateral cutaneous nerve with the clitoral nerve, as well as low morbidity and “invisible” scar at the donor site, adequate size of the neophallus, as well as better skin tone matching with the genital color compared to the RFFF phalloplasty. Disadvantages include presence of hair on the donor area, which in some cases requires prior laser hair removal, and a large donor defect that requires a split thickness graft for closure. Another limitation of the technique is that the ALT flap phalloplasty can only be done in patients with no excessive fatty tissue. Several authors have published their results of ALT flap phallic reconstruction with the neourethra constructed using the tube-within-a-tube technique with different success rates.
Radial forearm free flap phalloplasty
Starting in the late 1980s, the RFFF phalloplasty was considered the gold standard for neophallic reconstruction. This procedure involves the construction of a neophallus from forearm tissue. The neophallus is usually formed from tissue taken from the inner forearm skin (on the patient’s nondominant side) as well as vaginal tissue to form the neourethra. The forearm tissue includes nerves and vasculature that are grafted after the neophallus is constructed into a tube around a catheter. The neourethra is anastomosed to the native urethra and allows for urination while standing. The clitoris is left intact beneath the neophallus or within the constructed neoscrotum so that it can be stimulated independently of the neophallus. The use of the RFFF is well described by many surgeons in the genital reconstructive surgery literature.
The RFFF is perfused by the radial artery and venous drainage is provided by the paired venae comitantes which accompany the artery as well as the subcutaneous veins of the forearm including the cephalic vein. The radial artery arises from the brachial artery just distal to the antecubital fossa and travels between the brachioradialis and flexor carpi radialis as it runs distally into the forearm. It runs in the lateral intermuscular septum, which separates the flexor and extensor compartments of the forearm. It supplies the skin over the volar aspect of the forearm from the elbow to the wrist as well as portions of the radius. Generally, the skin is thin, pliable, and usually hairless while the vascular pedicle may be up to 18 cm in length and the vessel diameters are usually large (2 to 4 mm). Medial or lateral antebrachial cutaneous nerves can also be included in this flap and are commonly used when a sensate flap is created.
At the preoperative evaluation of the patient, it is necessary to examine the forearm to see where a hairless skin paddle can be taken. An Allen test should be performed with compression of the radial artery to ensure collateral circulation via the ulnar artery.
The flap is raised in the usual fashion. A tube-within-a-tube is created from a single, folded, 13-cm-long radial forearm flap. A 4.5-cm-wide area along the ulnar border of the forearm, without the presence of hair, is tubed inward around a Foley catheter, creating the neourethra. The adjacent radial strip of skin is de-epithelialized to allow the flap to be rolled up. The remaining 12-cm width of the radial part of skin is then used to provide an external cover as it is wrapped around the inner tubed portion. The radial artery, venae comitantes, lateral cutaneous nerve of forearm, and cephalic vein are dissected. The pedicle of the flap is left attached while a second surgical team prepares the recipient groin vessels.
To achieve a normally positioned phallus, inferior to the pubic symphysis, the urethral meatus must be advanced. This lengthening is achieved by raising a labia minora flap in continuity with the external urethral meatus which is tubularized around a Foley catheter. The new urethral opening is brought out onto the mons pubis, and this can be done either as part of a one-stage procedure or as an initial procedure some months prior to the phalloplasty. The clitoris remains undisturbed at the base of the new phallus, retaining its sensation. Once the recipient site is ready, the radial artery flap is transferred to the groin. The neourethra is anastomosed to the advanced female urethra. Vascular anastomosis of the radial artery end to side with the common femoral and of the cephalic vein end to end with the long saphenous vein is performed. The lateral cutaneous nerve of the forearm is joined to the ilioinguinal nerve ( Fig. 11.1 ).
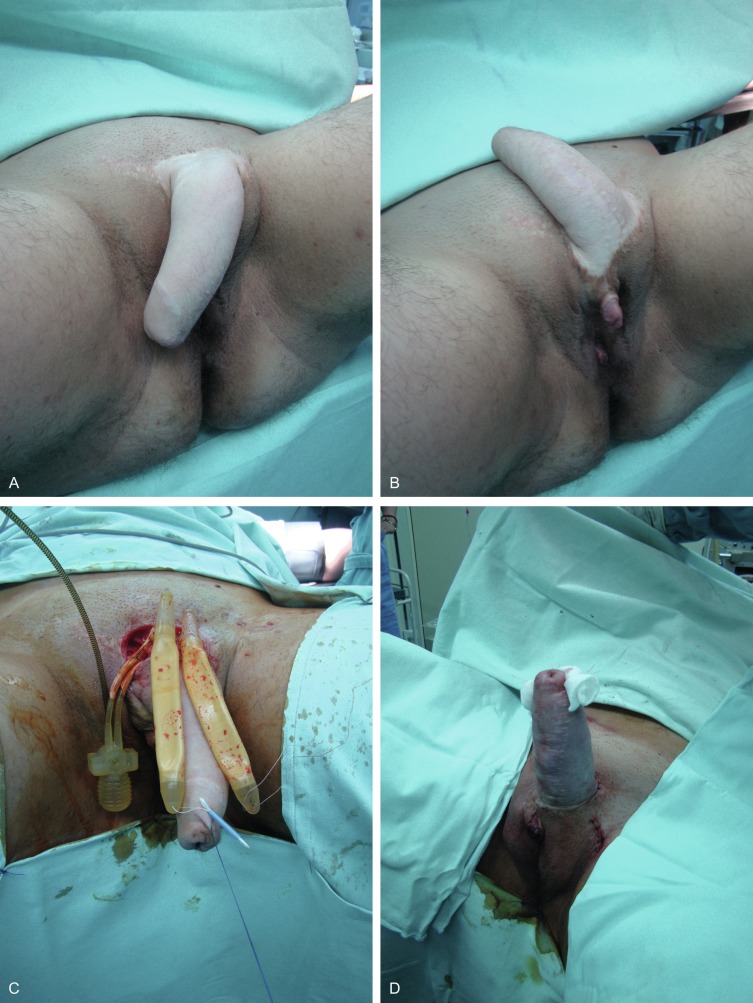
The Ghent group published their results using the RFFF for phallus reconstruction in 316 FTM patients. Their most common complications were related to urethral reconstruction (40%), with urethral fistula appearing in 17% of the patients requiring revision surgery, 16.7% developing fistulas that healed spontaneously, and 6% resulting in urethral strictures that were treated conservatively. Authors reported erectile prosthesis implantation in 143 patients with a high complication rate of 41% with need for revision surgery. In their study, the authors concluded that a complete penile reconstruction with an erection device can never be performed in a single operation, and that the penile prosthesis can only be placed after healing of the reconstructed neophallus is complete.
The London group has also published their results with RFFF phalloplasty with urethral reconstruction in 115 patients. Of these patients, 112 were satisfied with their postoperative results and 86% reported sensation in the neophallus. Urethral fistula and stricture occurred in 9 and 20 of the patients, respectively, which were resolved with revision surgery. Success of voiding in standing position occurred in 99% of their patients.
The advantages of the RFFF procedure include the creation of a sensate neophallus with the creation of the neourethra at the same stage. The main advantage of this flap is consistent arterial anatomy and a long vascular pedicle with vessels of a large diameter, which facilitates microsurgical anastomoses. However, disadvantages of this flap are related to issues that can occur at the donor site, such as delayed healing, need for skin grafting for closure, unsightly scars, and limited wrist motion. Additional limitations include the size and width of the neophallus, especially in thin patients, which can sometimes hinder the implantation of a penile prosthesis or lead to complications after implantation.
Latissimus dorsi flap phalloplasty
Our group uses the MLD flap because of its reliable and suitable anatomy (good size, volume, and length of neurovascular pedicle) to meet the aesthetic and functional requirements of phallic reconstruction. We report excellent results with MLD flap use in penile reconstruction in children and adolescents with congenital aphallia and penile trauma.
In preoperative preparation, patients are advised to have a special massaging treatment of the donor area (the nondominant side is usually chosen to be the donor site) for at least 3 months preoperatively, to make the donor site skin elastic and soft for the direct closure after flap harvesting. Patients are also advised not to work out to build muscle, as extra muscle bulk can cause problems with vascularization during the tubularization of the flap.
This procedure consists of two or three separate surgical procedures: designing and harvesting a latissimus dorsi musculocutaneous flap from the nondominant side with thoracodorsal artery, vein, and nerve; and, insertion of a penile prosthesis and staged neophallic urethroplasty. The steps of the surgery are described below.
First Stage
Removal of Female Reproductive Organs
The procedure is usually performed by three teams: gynecologists, plastic surgeons, and urologists. Hysterectomy and bilateral salpingo-oophorectomy can be performed either before or at the same time as the first stage of phalloplasty. In the majority of cases, the transvaginal approach is used for the removal of female reproductive organs. Complete vaginal mucosa removal is performed by colpocleisis except for the distal anterior vaginal wall close to the native urethral meatus, which is later used for the proximal part of neourethral reconstruction.
Clitoral Lengthening and Repositioning
Lengthening of the clitoris starts with a circular incision between the inner and outer layer of the clitoral prepuce and continues around the urethral plate and native urethral orifice. After complete degloving, the fundiform and suspensory clitoral ligaments are dissected from the pubic bone to advance the clitoris, mobilizing it to enable its fixation in a new position at the base of the neophallus.
Urethral Lengthening
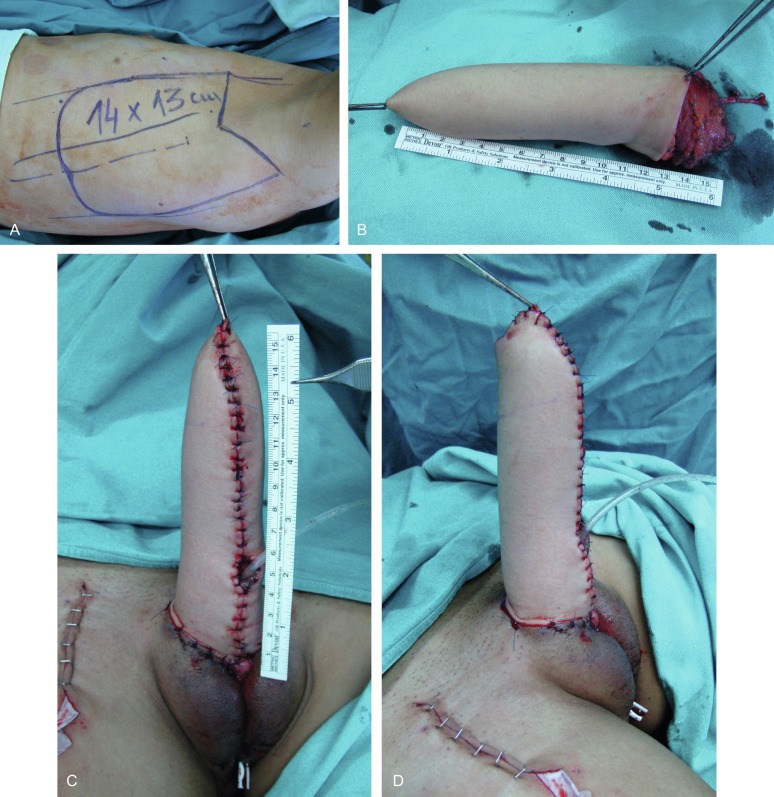
Reconstruction of the proximal part of the neourethra begins with the reconstruction of its bulbar part. A vaginal flap is harvested from the anterior vaginal wall with its base close to the female urethral meatus. This flap is joined with the remaining part of the divided urethral plate, forming the bulbar part of the neourethra. Further urethral reconstruction includes the use of all available hairless vascularized tissue to lengthen the neourethra to the maximum extent, preventing postoperative complications. In this way, the new urethral opening is placed at the base or in the first third of the neophallus, depending on the availability of the hairless tissue. Both varieties of flaps, clitoral, and labial, have fine supportive tissue that prevents fistula formation and yields satisfactory aesthetic results. A suprapubic cystostomy tube is introduced into the bladder and a silicone catheter is placed into the neourethra for a period of 10 days ( Fig. 11.2 A–E ).
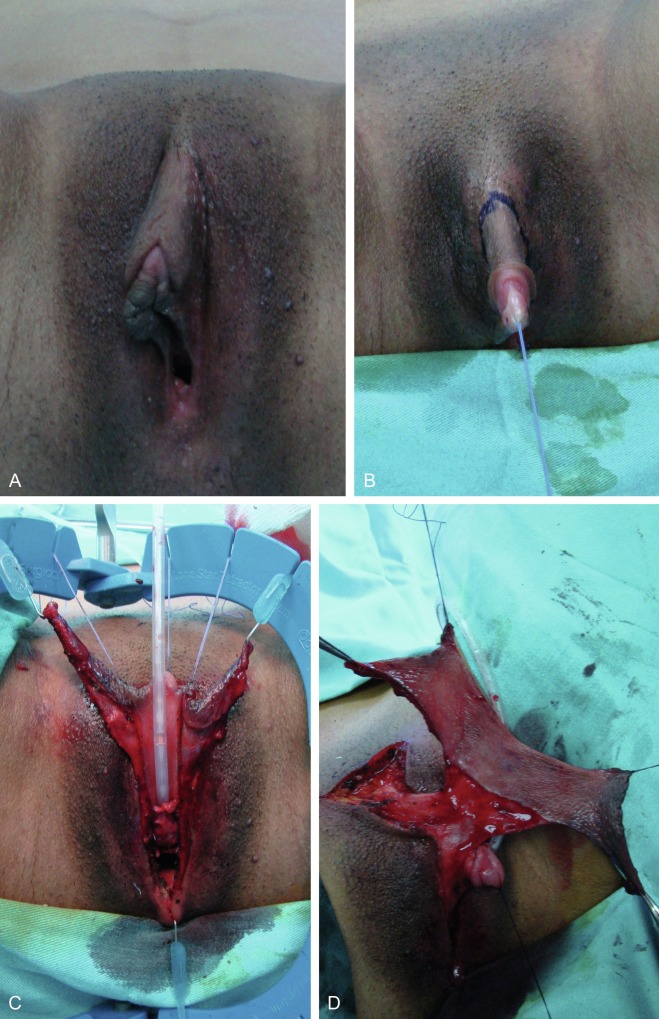
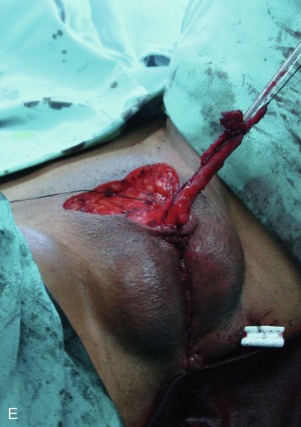
Reconstruction of the Perineum and Scrotum
Following colpocleisis, the vaginal space is closed and the perineum is reconstructed to have a male appearance. The two labia majora are joined in the midline over the neourethra creating a one-sac scrotum. Silicone testicular prostheses are inserted into the previously created pockets in the two labia majora, completing the scrotoplasty part of the procedure.
Flap Design
The patient is placed in the lateral position (upper torso placed in a full lateral position at 90° and the pelvis at a 30° angle) to provide access to the groin. The flap consists of two parts: a rectangular part for the neophallic shaft and a semicircular component for glans reconstruction. Flap dissection starts with an incision along the anterior skin margin down to the deep fascia. The plane is developed between the latissimus dorsi and serratus anterior muscles, using sharp and blunt dissection. The flap is divided inferiorly and medially, cauterizing the large posterior perforators of the intercostal vessels, and then lifted to expose the neurovascular pedicle. The pedicle, surrounded by fatty tissue, is identified and dissected proximally up to the axillary vessels. The thoracodorsal nerve is identified and isolated proximally for 3 to 4 cm, preserving its vascularization. The flap is elevated completely, except for the neurovascular bundle, which is not transected until the recipient vessels and nerve have been prepared for microanastomosis. The latissimus muscle is fixed to the edges of the skin at several points to prevent layer separation during further dissection. The flap is tubularized creating the neophallus, while still perfusing on its vascular pedicle. The circularized terminal part is rotated back over the distal body and sutured to create a neoglans. The completely constructed neophallus is detached from the axillar region after the subscapular artery, vein, and thoracodorsal nerve are clamped and divided at their origins, to achieve maximal pedicle length ( Fig. 11.3 A and B ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree




