12.4 Pre-operative Preparation and Organization
At first contact, a screening phone call is made providing medical history, and the previous medical records are collected. The patients attended a mandatory full-day course with information and education in group sessions held by a surgeon, anaesthesiologist, nurse, dietician and physiotherapist. The patient was fully informed about the fast-track set-up and what will happen on the day of surgery. Weight loss of 5 % and change in lifestyle and eating habits were strongly encouraged prior to surgery. At the end of the day of the first visit, patients were evaluated for indication for surgery, type of operation was decided and day of surgery was scheduled, usually 5–6 weeks later (Table 12.1).
Table 12.1
Enhanced recovery after surgery (ERAS) protocol
Pre-operative preparation | Screening by phone: criteria for surgery? Medical records collected A 1-day course on education and information by the multidisciplinary team Initiation of patient contribution: physical activities, diet, stop smoking |
Day of operation | Clear oral fluid up to 2 h before surgery, no solid food for 8 h Shower with antiseptic sponge 2 h before surgery, empty urinary bladder Single-dose cefuroxime (1.5 g iv) or 800 mg Bactrim® orally 2 h before surgery No thrombosis prophylactics unless history of thrombo-embolic diseases, or APC resistance in family history |
During operation | 8 mg i.v. dexamethasone at induction of anaesthesia Standardized anaesthesia Local anaesthetics in wounds |
In the recovery room | Observation (telemetry, oxygen saturation), pain medication and antiemetics when needed Free oral fluid (amount recorded ) Walking test (20 m) after 2 h and arrival to the ward |
At the ward – day of surgery | Standard multimodal analgesia and anti-emetic Intensive mobilization; out of bed every h, using the PEP whistle, “keep the patient busy” 5,000 IU low molecular heparin 6 h after surgery If tachycardia, more pain than expected or not getting out of bed the surgeon notified |
Day after surgery | Shower and changing the bandages Group meeting with the dietician (liquid and pure diet for 5 weeks) Going through pre-operative blood test and administering the supplements Discharge meeting with the surgeon emphasizing “early sign of complication” and the importance of early contact by phone Patients living within 90 min from hospital discharged home, the others to the neighbouring hotel for 1–4 days |
After discharge | Phone call 1 and 7 days and 3 weeks after discharge Self-administration of low molecular heparin s.c. for 10 days Omeprazole 40 mg daily for 3 months |
We developed a “length of hospital stay prediction system” based on comorbidity, BMI, psychosocial status and distance of residence from the hospital [17]. The ambition was to close the ward during most weekends, and this was facilitated by identifying and scheduling patients with anticipated longer hospital stay to the beginning of the week. Initially, patients were offered 3-day post-operative hospital care, but in 2008 this was reduced to 2 days and in 2009 patients were planned to be discharged on the first post-operative day. Patients living more than 1 hour of transport from the hospital were encouraged to stay near the hospital until 5 days after surgery.
The ward and one operating theatre were booked for 4 days at a time; the frequency of “bariatric weeks” depended on the number of patients cleared for surgery. The ward is usually closed on Friday evenings but remained open during the weekend only due to emergency cases or prolonged hospital stay.
12.5 Logistics and Fast-Track Principles
Medical audit for the patient flow was made. All elements of the treatment procedures before, during and after surgery were thoroughly standardized. The team in the operating theatre consisted of two surgeons, one senior bariatric surgeon and the other a surgeon skilled in laparoscopy but in training for bariatric surgery. There were also two operating nurses, one anaesthesiologist and one anaesthetist nurse. The time recordings were continuously registered in a database. The whole treatment was based on teamwork and all co-workers were primed on their role to get a streamlined logistics and avoid unnecessary waiting. An important principle at the operating theatre was “parallel working”, many staff members working with the patient at the same time, washing and covering abdomen before the patient was intubated, reflecting the fact that the “busy time in the process is the turnover-time”.
Standardization of the procedure makes the surgery safer, as it is performed precisely the same way by all surgeons of the team. Most authors agree that the experience and skill of the individual surgeon is the most important single factor for patient safety and good outcome [4–8]. Thus, training programmes for new surgeons are of great importance in order to avoid painful learning curves. Technical training in intracorporeal suturing and use of staplers needs numerous repetitions under guidance (Table 12.2).
Table 12.2
Safe surgery, teaching aspects and quality control
All members of the team | Continuous medical education of all staff Standardization Research and development projects for all categories |
Surgeon | Two surgeons procedure |
Five-step training programme for new surgeon | 1. Assisting in 50 operations 2. Starting operation and operating for 45 min with strict hands-on guidance (two of six operations that day) 3. When technically sufficient and good progression within 45 min, the trainee is allowed to complete the operation, but not exceeding 60 min 4. The trainee is performing 100 more operations still with hands-on guidance 5. The trainee becomes expert surgeon and starts performing surgery with a trainee |
Early sign of complication | Immediate re-operation |
Recordings of operation | If complication within 30 days, video recording of the operation examined in order to learn and improve |
Continuous database recordings | Individual surgeon’s complication rate can be followed |
All the instruments used for each operation were pre-packed into three standardized packages. One package contained the reusable surgical tools, packed and sterilized at our hospital. The other two packages were custom made for our purposes by surgical tools suppliers: one containing staplers and troacars (Ethicon Endo-Surgery or Covidien) and the other containing all other single-use items for one operation (Mölnlycke Health Care AB, Gothenburg, Sweden).
12.6 Surgical Procedure
The surgical procedure was meticulously standardized and the time used for different parts of the operation was recorded (Fig. 12.2). The purpose was to enhance technical improvements and shorten the surgical time. This also helps the anaesthesiologist to monitor the anaesthesia according to the progress of the surgery and to time the termination of the anaesthesia.
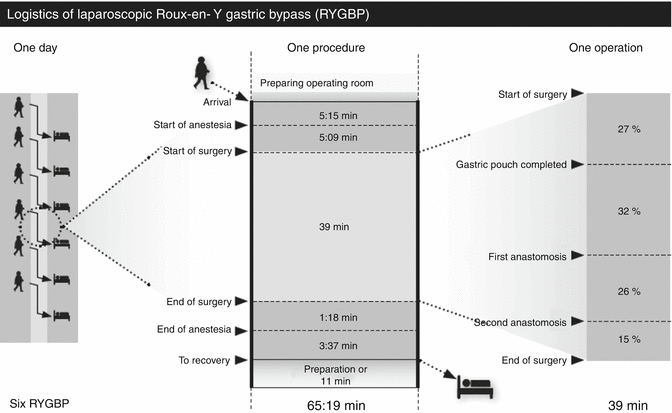

Fig. 12.2
Logistic of the treatment process from when a patient walks into the operating theatre until next patient arrives for 261 patients operated during second half of 2010. Non-operative time is mean time (minutes: seconds). The operating time is referred as median (From Jacobsen et al. [17], with permission)
The surgical procedure has been described in detail previously [17, 19]. The patient was placed in a supine, split-leg, reversed Trendelenburg position. Five ports were used: two 5-mm and two 12-mm ports for instruments and one 10-mm port for the camera. A 45° scope and intra-abdominal pressure at 18 mmHg were used. In brief, a small gastric pouch (15 ml) was created with the bowel in an antecolic and antegastric position. The gastroenteric (GE) anastomosis and the enteroenteric (EE) anastomosis were stapled linearly and the staple holes handsewn. The bowel was approximated to the gastric pouch as an omega loop, subsequently divided by stapling between the two anastomoses. The last step was to test the integrity of the GE anastomosis by inflation with methylene blue–dyed saline via a nasogastric tube. The nasogastric tube was inserted and removed during the period of anaesthesia. Until June 2010, LRYGBs were performed without closing the mesenteric defects, but since July 2010 the mesenteric defects were stapled as described previously [20].
During the period from 2007 to 2013, five skilled laparoscopic surgeons went through a specific training programme in bariatric surgery. In order to minimize the negative consequences of the learning curve, the training programme was strictly organized in five steps as shown in Table 12.2.
12.7 Anaesthesia
The patient walked into the OR after receiving a peripheral venous cannula and IV antibiotic prophylaxis with single dose of cefuroxim 1.5 g. The anaesthetic method has been described in detail [10, 17]. In brief, induction of anaesthesia was performed in semi-reversed Trendelenburg position, using fentanyl and target controlled infusion of propofol and remifentanyl based on the patient’s ideal weight. Muscle relaxation was facilitated with a small dose of vecuronium. After tracheal intubation, propofol was substituted with desflurane to ensure rapid recovery from anaesthesia after the surgery.
12.8 Post-operative Care (Table 12.1)
The patients were observed post-operatively and monitored at the recovery unit for approximately 2 h. Within the first 2 h, the patients were mobilized out of bed and were allowed to drink freely. The patients were encouraged to drink at least 1 L of water until the next day.
Post-operative pain was controlled with paracetamol, parecoxib and oxycodone at fixed intervals. Post-operatively, 1,000 mL Ringer’s acetate was prescribed as IV fluid until the next morning.
If the patient had more pain than expected, experienced tachycardia or was not able to move out of bed, the surgeon and/or anaesthesiologist were consulted. Threshold for early re-laparoscopy was low in order to reduce the consequences of a possible complication.
At the ward, observation and mobilization were continued. Patients were instructed to get out of bed at least once every hour, drinking small sips and blowing the PEP whistle. Early the day after surgery patients took a shower and got new bandages. They also attended a group meeting with the dietician to discuss fluid and pureed food, and with the surgeon for instruction about possible complications and how to react. After discharge the patients had open access for readmission to the hospital and were carefully instructed to contact the ward or surgeon in case of any adverse events. The patient also had the telephone numbers of the surgeon on call at any time, of the ward and the outpatient clinic.
A questionnaire was sent to all the patients 4 weeks after surgery in order to evaluate patient satisfaction. This questionnaire consisted of questions about the patient’s perception of the different parts of the treatment process.
12.9 Follow-Up Routines
Our patients also entered a 5-year interdisciplinary follow-up programme. This includes live-style courses consisting of a group of 16–18 patients at 3 months, 1 and 3 years. Metabolic follow-up is done twice a year in the first 3 years and then once a year, most often in co-operation with their general practitioner. Abdominal problems that could be related to the obesity surgery are a part of the treatment package and taken care of by the surgeons of the team.
12.10 Data Collection and Statistics
Data were prospectively collected and registered in our database (FileMaker 11), which is a part of our routine patient record system. The surgeons filled in data regarding the operation and post-operative care, as well as primary data concerning patient characteristics and discharge status. The anaesthesia personnel registered the time consumption for different tasks in the operation theatre and turnover time between operations. The theatre procedure was divided in six different parts for this purpose (Fig. 12.2).
All data regarding weight loss, metabolic status and post-operative changes in comorbidity as well as complications were registered continuously. All patient contacts with personnel from the obesity team were recorded and relevant information registered in the database. All patients operated in Sweden had their data entered into the database of the Scandinavian Obesity Surgery Registry (SOReg) that covers >98 % of bariatric surgery in Sweden. This enabled us to make frequent comparisons with other specialist centres.
Proportions are referred to as numbers (%). All continuous data are presented as median (range) unless otherwise stated. Differences were estimated by Student’s t test, with a p-value <0.05 being regarded as significant. Weight loss was expressed as percentage excess BMI loss (%EBMIL).








