Reconstruction of severe facial deformities poses a unique surgical challenge: restoring the aesthetic form and function of the face. Facial transplantation has emerged over the last decade as an option for reconstruction of these defects in carefully selected patients. As the world experience with facial transplantation grows, debate remains regarding whether such a highly technical, resource-intensive procedure is warranted, all to improve quality of life but not necessarily prolong it. This article reviews the current state of facial transplantation with focus on the current controversies and challenges, with particular attention to issues of technique, immunology, and ethics.
Key points
- •
Computed tomography angiography is helpful preoperatively for assessment of recipient vessel targets to plan for typically 2 arterial and 2 venous anastomoses to support the transplant.
- •
Skin is highly antigenic; acute rejection of facial transplants is common, if not universal, but chronic rejection is rare.
- •
Typical 3 drug immunosuppression exposes patients to risks of infection and development of malignancies, both of which can be deadly.
- •
Careful patient selection, thoughtful informed consent, and consideration of the psychosocial impact of facial transplant are necessary to keep the procedure ethically sound.
Introduction
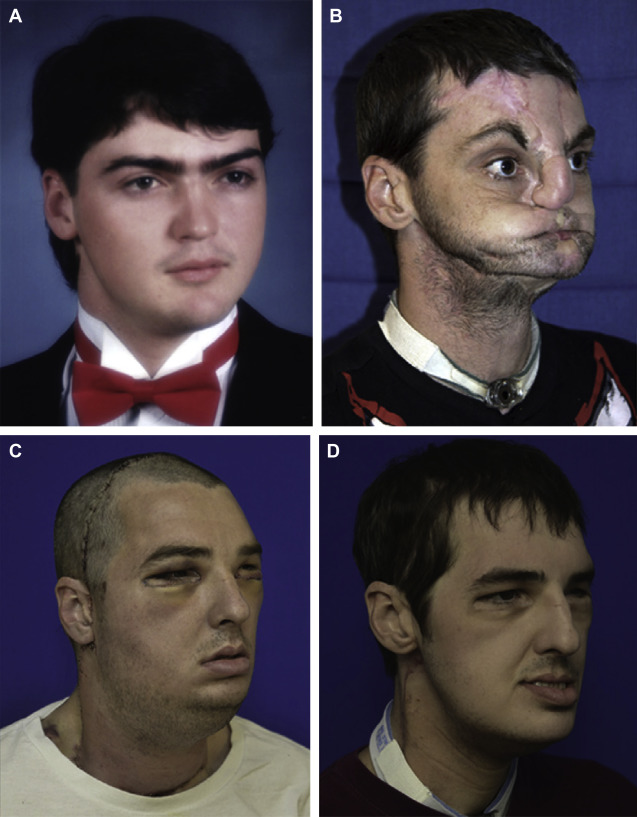
Conventional approaches to facial reconstruction are largely dictated by the extent of the defect. Although smaller defects may be amenable to local flaps, more extensive defects often require free tissue transfer or large split-thickness skin grafts. These techniques may suffice to provide coverage and occasionally restore function. However, in terms of both cosmesis and functionality, traditional reconstruction options fall short for patients with the most severe whole face deformities, often resulting in a patchwork appearance that reflects the sometimes dozens of surgeries these patients endure in pursuit of the elusive goal of achieving an acceptable appearance and quality of life ( Fig. 1 ).
Over the last 10 years, facial transplantation, a form of composite tissue allotransplantation (CTA), has emerged as a viable option for reconstruction of the most severe facial deformities in carefully selected patients. There have been 31 cases of facial transplantation reported in the world literature since the first case in 2005, with good results overall ( Table 1 ). However, facial transplantation was controversial at its inception; despite growing experience with this procedure, significant controversies persist.
| Number | Month/Year | City & Team Leader | R Sex, Age (y) | D Age (y) | Indication | Type | SD (h) |
|---|---|---|---|---|---|---|---|
| 1 | 11/2005 | Devauchelle, Amiens, France | F, 38 | 46 | Animal attack | Partial myocutaneous | 15 |
| 2 | 04/2006 | Guo, Xian, China | M, 30 a | 25 | Animal attack | Partial osteomyocutaneous | 13 |
| 3 | 01/2007 | Lantieri, Paris, France | M, 29 | 65 | NF 1 | Partial myocutaneous | 11 |
| 4 | 12/2008 | Siemionow, Cleveland, United States | F, 45 | 44 | Gunshot injury | Partial osteomyocutaneous | 22 |
| 5 | 03/2009 | Lantieri, Paris, France | M, 27 | 43 | Gunshot injury | Partial osteomyocutaneous | 19 |
| 6 | 04/2009 | Lantieri, Paris, France | M, 37 a | 59 | Burns | Partial myocutaneous | 13 |
| 7 | 04/2009 | Pomahac, Boston, United States | M, 59 | 60 | Burns | Partial osteomyocutaneous | 17 |
| 8 | 08/2009 | Lantieri, Paris, France | M, 33 | 55 | Gunshot injury | Partial osteomyocutaneous | 16 |
| 9 | 08/2009 | Cavadas, Valencia, Spain | M, 42 a | 35 | Radiotherapy | Partial osteomyocutaneous | 15 |
| 10 | 11/2009 | Devauchelle, Amiens, France | M, 27 | — | Burns | Partial osteomyocutaneous | 19 |
| 11 | 01/2010 | Gomez-Cia, Seville, Spain | M, 35 | 30 | NF 1 | Partial myocutaneous | 22 |
| 12 | 04/2010 | Barret, Barcelona, Spain | M, 31 | 41 | Gunshot injury | Full osteomyocutaneous | — |
| 13 | 07/2010 | Lantieri, Paris, France | M, 37 | — | NF 1 | Full myocutaneous | 14 |
| 14 | 03/2011 | Pomahac, Boston, United States | M, 25 | 48 | Burns | Full myocutaneous | 17 |
| 15 | 04/2011 | Lantieri, Paris, France | M, 45 | — | Gunshot injury | Partial osteomyocutaneous | — |
| 16 | 04/2011 | Lantieri, Paris, France | M, 41 | — | Gunshot injury | Partial osteomyocutaneous | — |
| 17 | 04/2011 | Pomahac Boston, United States | M, 30 | 31 | Burns | Full myocutaneous | 14 |
| 18 | 05/2011 | Pomahac, Boston, United States | F, 57 | 42 | Animal attack | Full osteomyocutaneous | 19 |
| 19 | 01/2012 | Özkan, Antalya, Turkey | M, 19 | 39 | Burns | Full osteomyocutaneous | 9 |
| 20 | 01/2012 | Blondeel, Gent, Belgium | N/A | N/A | N/A | Partial osteomyocutaneous | 20 |
| 21 | 02/2012 | Nazir, Ankara, Turkey | M, 25 | 40 | Burns | Full face transplant | — |
| 22 | 03/2012 | Özmen, Ankara, Turkey | F, 20 | 28 | Burns | Partial face transplant | — |
| 23 | 03/2012 | Rodriguez, Baltimore, United States | M, 37 | 21 | Gunshot injury | Full osteomyocutaneous | 36 |
| 24 | 05/2012 | Özkan, Antalya, Turkey | M, 27 | 19 | Burns | Full face transplant | — |
| 25 | 01/2013 | Pomahac, Boston, United States | F, 44 | — | Burns | Full myocutaneous | 15 |
| 26 | 05/2013 | Özkan, Antalya, Turkey | M 27 | 19 | Gunshot injury | Partial osteomyocutaneous | — |
| 27 | 07/2013 | Maciejewski, Warsaw, Poland | M, 33 | 42 | Crush trauma | Partial osteomyocutaneous | 27 |
| 28 | 07/2013 | Özkan, Antalya, Turkey | M, 27 | — | Ballistic trauma | Full osteomyocutaneous | — |
| 29 | 08/2013 | Özkan, Antalya, Turkey | M, 54 a | — | Ballistic trauma | Partial osteomyocutaneous | — |
| 30 | 12/2013 | Özkan, Antalya, Turkey | M, 22 | — | Ballistic trauma | Partial osteomyocutaneous | — |
| 31 | 12/2013 | Maciejewski, Warsaw, Poland | F, 26 | — | Neurofibromatosis | Full myocutaneous | — |
In this article, the authors focus on the current controversies, challenges, and questions that confront facial transplantation while highlighting the lessons learned and challenges overcome through experience thus far. The discussion focuses on 3 main topics: technical issues, issues of facial transplantation immunology, and ethical concerns.
Technical issues
Facial transplantation is undoubtedly a highly technical and complex surgical undertaking that requires a cohesive team approach. Because of the complexity of the undertaking, only a handful of centers worldwide have the expertise and infrastructure necessary to perform facial transplantation. As the worldwide experience has grown over the last decade, technique has been refined, though some controversies and challenges remain.
Because facial CTA requires revascularization via microvascular anastomoses, a detailed understanding of the recipient vascular anatomy is paramount to determine if a given individual is an acceptable facial transplantation candidate and, if so, to create a viable surgical plan. This understanding is especially important because most patients who are considered for facial transplantation have undergone numerous prior reconstructive surgeries, usually including prior free tissue transfer, which may limit the availability of viable recipient vessels and can make dissection of these vessels challenging and time consuming because of scarring and tissue fibrosis. Computed tomography (CT) angiography of recipients before facial transplantation is advocated to identify potential vascular targets and to better understand their relationship to other facial structures in the interest of shortening already long surgical times, minimizing intraoperative blood loss, and increasing the chances of having robust perfusion to the transplant. CT angiography also helps to identify the specific branching patterns of the external carotid system and the caliber of these vessels for a given facial transplant candidate so that a specific surgical plan can be made preoperatively.
There has been controversy regarding the number of microvascular anastomoses necessary to support a face transplant. Each anastomosis increases surgical time and complexity, but loss of a CTA due to arterial insufficiency or venous congestion would be devastating; thus, redundancy in the feeding blood supply is typically sought. Studies have demonstrated that one vascular anastomosis is sufficient to perfuse a facial tranplant. However, to minimize the risk of catastrophic graft lost, it is advisable to plan for at least 2 arterial and 2 venous anastomoses. Choosing recipient vessels with large caliber to ensure reliable flow is important. Caution must be exercised, however, when considering bilateral end-to-end anastomoses directly to the external carotid as ischemia of the hypopharynx and to the eyes (via loss of external carotid to ophthalmic collaterals) are potential complications.
There are also questions on the donor side as to which vessels will adequately perfuse the transplanted tissue. The answer to this question undoubtedly depends on the specific tissues included in the transplant. There is evidence that the facial arteries alone can supply both the mandible through submental and periosteal perforators and the maxilla via perforators from the oral mucosa. This allows for composite partial facial transplants of the midface and lower face, including bone, to be supplied by the facial arteries. The overarching principle is vascular supply via angiosomes in which particular territories of facial soft tissue are supplied predictably by certain vessels, with the understanding that there are often collaterals between angiosomes. Although portions of the upper face and periorbital region are within angiosomes supplied by branches from the ophthalmic artery, itself a branch of the internal carotid system, full face transplants as well as partial face transplants of the upper face can be reliably supplied by collaterals from the superficial temporal arterial system, which is fed by harvesting and anastomosing the external carotid. It has also been advocated to leave the recipient’s forehead skin intact until after the vascular anastomoses have been performed and the forehead portion of the CTA is observed to be well perfused (the most likely site of ischemia). To standardize the transplant harvest and to maximize vessels available to robustly supply and drain a full face CTA, Lantieri and his team routinely harvest 6 vessels (2 external carotids, 2 external jugular veins, and 2 thyrolingofacial trunks).
Most face transplants have been harvested from heart-beating, brain-dead donors, though harvest from non–heart-beating donors has also been performed. Coordination is required with other surgical teams who are harvesting solid organs from the same donor to determine the timing of facial harvest relative to harvest of organs, which will result in loss of perfusion to the remaining body tissues.
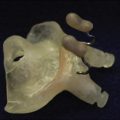
Another potentially controversial aspect of harvesting of the face, as compared with other transplants, is the resultant external disfigurement to the donor. To lessen the impact that facial harvest will have on the families of donors, it is common to take a mold of the donor’s face before harvest so that a painted resin mask can be fabricated during the harvest for placement on the donor before returning the body to the family ( Fig. 2 ).