Key points
• Over the past two decades, success achieved by composite tissue allografts (CTA) in restoration of many functional and aesthetic deficits of the face has caused a paradigm shift in reconstructive surgery.
• Preoperative radiological imaging and intraoperative Doppler ultrasonography provide important information about the presence and patency of vessels that can save time and prevent failures of anastomoses postoperatively.
• In the authors’ first allotransplantation, all midface structures, including the skin, muscles, glandular tissue, and modified LeFort III anterior maxillary structures, were supplied by bilateral facial arteries alone.
• In addition to tissue immunogenicity, facial transplant donors and recipients will need to be matched on the basis of race, gender, age, skeletal dimensions, and skin color/tone.
• Biopsies of the transplanted skin are often necessary to later confirm episodes of rejection and grade their severity.
• A sentinel flap with similar antigenic composition serves as a biopsy site for monitoring purposes.
• Careful attention must be paid to allow the coronal, sagittal, and axial axes to be accurately positioned for facial width and projection.
• Previous face transplantations have shown that sensory nerve recovery occurred as early as 3 months postoperatively and that most patients achieved motor recovery in 6 to 18 months.
• The indications for facial CTA include only defects that are beyond reconstructon with conventional techniques.
Introduction
The face is one of the most complex parts of the body and harbors the most aesthetically sensitive organs, such as the lips, nose and eyelids. Reconstruction of these features often requires a combination of aesthetic and functional restorations. Severe facial trauma, extensive oncologic resections, and extremes of congenital disfigurements impair facial functions, profoundly decrease the quality of life, and preclude social integration. In these cases, restoration of the face, especially of the aesthetically sensitive organs, with autologous tissue and/or prosthetic alloplastic materials, cannot fully restore the unique form and function provided by the native tissues of the face and does not allow patients to assimilate into society. Reconstruction with conventional techniques in severely disfigured patients contradicts one of the main principles of plastic surgery—“replace like with like.”
Although currently used reconstructive procedures have advanced a great deal in recent years, they are still limited by poor functional and aesthetic outcomes, multiple revision procedures, prolonged rehabilitation resulting in patients not returning to work or social life and becoming dependent on family members and the health care system for care, high costs of multiple surgeries and hospitalizations, and donor site morbidity, and postoperative complications associated with implanted prosthetic materials (infection, ulceration, extrusion, altered healing, etc.) Furthermore, these procedures frequently result in functional deficits and a patchy appearance of the face due to mismatch of color, texture and thickness. Composite tissue allotransplantation (CTA) offers a solution to these shortcomings of autologous reconstruction. Over the past two decades, success achieved by CTA in restoration of many functional and aesthetic deficits of the face has caused a paradigm shift in reconstructive surgery.
In cases of severe and complex facial deformities, where conventional reconstructive methods significantly fall short, face transplantation has produced superior functional recovery and aesthetic restoration. However, facial CTA is still an emerging and developing concept in the field of plastic surgery. Its widespread clinical use currently has been limited by the risk of rejection and use of life-threatening immunosuppressive therapy for a nonlife-threatening condition. With the evolution of continued improvements in immunomodulation and refinements of surgical technique, CTA will gain widespread clinical acceptance in the field of reconstructive surgery.
Since the first face transplantation in 2005, at the time of this writing, 39 face composite tissue allotransplantations, three of which at the Cleveland Clinic, have been performed throughout the world. , In this chapter, the technical aspects of face transplantation will be highlighted in light of our own experience at the Cleveland Clinic and the recent literature on facial CTA. Most facial allotransplant candidates and donors are unique with regard to anatomic defects, skeletal measurements, and soft tissue characteristics. Therefore it is impractical to describe one surgical technique that is applicable to all face transplantation procedures. The aim of this chapter is to describe the technical and aesthetic principles of face transplantation that can be applied to achieve the most refined and superior outcomes.
The multidisciplinary transplantation team
Besides its technical complexity, face transplantation has certain inherent complexities requiring coordination of multiple disciplines. Face transplantation, therefore, should only be attempted at clinical institutions that have the necessary clinical experience and infrastructure that will allow for the coordination of the entire care, with the maximum benefit and the lowest risk to the patient. The face transplantation team, which includes a microsurgeon, a craniomaxillofacial surgeon, an anesthesiologist, an internist, an immunologist, a psychiatrist/clinical psychologist, an intensive care specialist, a pharmacologist, a dietitian, an occupational therapist, and a speech therapist, should be orchestrated by a leader who is responsible, from planning through execution, to analyzing the outcome. Each institution should establish, and strictly adhere to, a clinical protocol that may be tailored to the specific needs of the face transplant recipient.
The face transplantation surgical team should include individuals who have expertise in reconstruction of the head and neck region, an understanding of the microvascular anatomy of the face, and experience in microvascular anastomosis, microscopic repair of nerves, and craniofacial osteosynthesis.
Face transplantation—anatomic considerations
The extent of transplantation and the tissues to be included vary, depending on the extent of the recipient’s injury. Burn injuries usually involve the more superficial structures, such as the skin and muscles, whereas gunshot injuries or oncologic resections often require more complex and extensive reconstructions of the skeletal and soft tissue components of the face. From the reconstruction standpoint, the face can be divided into three regions: (1) upper third (scalp and skull); (2) middle third (midface); and (3) lower third (mandible). By considering the anatomic and functional aspects of each subunit to be reconstructed, the ideal facial allograft can be designed containing multiple tissue layers. The design of the allograft should also take the individual blood supply of tissues into consideration.
Detailed preoperative radiologic imaging and careful intraoperative dissection of donor and recipient vessels are required. In addition, comprehensive medical and surgical histories of the donor and the recipient may provide essential clues that will aid in the planning of surgery or in changing strategies during the operation.
Tracheostomy of the donor and/or the recipient may be performed before allograft harvest to allow for full access to the neck and the facial structures. In each facial allograft harvest, the donor vessels are dissected, beginning at the common carotid artery and extending superiorly to reach the carotid bifurcation. In certain cases where the mandible is not part of the allograft, mandibular osteotomy can be performed for exposure of the facial vessels.
The selection of recipient vessels depends on the region of the face or scalp to be reconstructed and the availability of vessels. Some studies recommended the superficial temporal vessels for upper third (scalp and skull) reconstruction; the facial and superficial temporal vessels for middle third (midface) reconstruction; and the ipsilateral neck vasculature for lower third (mandibular) reconstruction. Alternative recipient vessels may be needed in patients who have undergone previous microvascular reconstructive procedures. Some of the alternative vessels include (1) adjacent small vessels in the area of the first-choice vessel; (2) major neck vessels (external carotid artery or internal jugular vein); and (3) distant vessels (thyrocervical trunk or contralateral neck). Preoperative radiologic imaging and intraoperative Doppler and/or color Doppler ultrasonography may provide important information about the presence and patency of vessels that can save valuable time intraoperatively and prevent failures of anastomoses postoperatively.
Knowledge of facial angiosomes is important for the planning and harvesting of the allograft. Initial angiosome studies suggested that multiple facial arteries would be necessary to adequately perfuse an entire panfacial allograft. However, clinical cases of facial allotransplantation and experimental data have proven otherwise. In the Cleveland Clinic’s first allotransplantation, all midface structures, including the skin, muscles, glandular tissue, and modified LeFort III anterior maxillary structures, were supplied by bilateral facial arteries alone. This case and the relevant literature support the idea that periosteal blood supply to the maxillary bone is enough for bone survival. Anatomic studies further supported these findings by showing that maxillary segments receive periosteal blood supply from the ascending palatine and superior labial branches of the facial artery. Therefore during allograft harvesting, it is critical that the periosteum remains attached to the maxilla for bone viability. The pedicle of choice should be the bilateral internal maxillary arteries in allografts that include the maxilla without the overlying soft tissues because the periosteum is excluded in such flaps and midline crossover at the palate is poor. In cases where the forehead, ear, and scalp are parts of the allograft, it would be necessary to include the superficial temporal artery and the external carotid artery as the source vessels. Additionally, occipital arteries need to be identified and protected for perfusion of the grafts containing the entire scalp.
Another surgical consideration is the performance of bilateral orbital exonerations to provide adequate exposure of the lower orbital floor in preparation for LeFort III osteotomies. Periosteal blood supply of the maxilla allows for a “veneer” approach, where the anterior, lateral, and mediolateral maxillary walls are harvested, leaving behind the posterior maxillary bone. With this approach, it is important not to disrupt the pterygopalatine branches of the internal maxillary artery, and the flap is easily placed into the recipient bed without removing scar tissue near the middle cranial fossa skull base. Another technical consideration is to perform osteotomies as one of the last steps in flap harvesting. Ligation of the distal external carotid artery before zygomatic osteotomies prevents sudden hemorrhage from the branches of the internal maxillary artery; such hemorrhages may threaten the hemodynamic stability of the donor during harvesting of solid organs by other transplantation teams.
Preoperative workup for recipient
Selection of the potential candidate for face transplantation is probably the most important and challenging part of the preparation process. Informed consent should be obtained after the patient understands all of the potential risks of face transplantation, including rejection or loss of allograft, metabolic complications, opportunistic infections, malignancy, and death. Psychiatric evaluation should be performed to identify preexisting psychiatric conditions and to assess for postoperative compliance. All candidates on transplant lists should undergo preoperative testing to evaluate the feasibility of face transplantation and to individualize face transplantation protocol. These tests include computed tomography (CT) of the head/neck, chest, abdomen/pelvis, CT angiography (vessel mapping, arterial and venous phase), magnetic resonance imaging (MRI) of the face, nerve sensory testing, and electromyography (EMG) of the facial nerve. Malignancy should be ruled out to prevent any detrimental results in the postoperative period due to chronic immunomodulation. All patients older than 50 years of age should undergo upper/lower gastrointestinal (GI) endoscopy to rule out undiagnosed carcinoma. Women older than 40 years of age must also have a recent mammography. Each patient, regardless of age, should undergo a complete dental–oropharyngeal examination to rule out dental cavities, abscesses, periodontal disease, and oropharyngeal carcinoma.
Another recent development in the process of preparation for face transplantation is the use of virtual surgical planning (VSP). It has been shown that computer-aided design, modeling and intraoperative surgical navigation significantly reduce surgical time and help to achieve more successful allograft placement. , In our experience, the use of computer-aided design and stereolithic models has yielded similar results. However, unexpected bony discrepancies and major revision surgeries have still been reported after use of computer aided surgical planning. ,
Donor allograft recovery
Donors and recipients of solid organs should be matched on the basis of similar blood type, viral serology, and immunologic profile (lymphocytotoxic, human leukocyte antigen [HLA], B- and T-cell matching tests); facial transplant donors and recipients will need to be matched additionally on the basis of race, gender, age, skeletal dimensions, and skin color/tone.
An impression of the donor’s face is taken to fabricate the donor mask before procurement of the donor face. Tracheostomy is usually the first step in the harvesting procedure. The allograft is harvested with its pedicle, according to the preoperative plan. In cases where the upper and lower jaws are transplanted, performing maxillomandibular fixation (MMF) before the osteotomies can be advantageous for preservation of the occlusion. The extra skin should be included in the graft design to allow for tension-free closure. The harvest team should work in concert with the solid organ transplantation teams involved. The sequence of harvesting organs and clamping of the aorta needs to be discussed preoperatively because harvesting of other organs will result in loss of perfusion to the remaining body tissues. Barret et al. described a synchronous procurement method whereby the face and other internal organs were harvested after the heart and lungs. During harvesting, cannulas should be placed into the facial artery or external carotid artery for constant infusion with cold (4°C) University of Wisconsin (UW) solution during transport. A facial prosthesis (donor mask), which is fabricated during harvesting, should be placed at the end of the operation to lessen the impact of the facial transplantation on the families of donors. The facial/scalp allotransplant is wrapped in sterile moist gauze and kept on ice in a secure, sterile compartment for rapid transport. Cold ischemia time for face allotransplants is currently unknown due to paucity of data. But studies on hand CTA showed that cold ischemia time for successful hand transplantation ranges from 30 minutes to 13 hours.
A surplus of skin, oral mucosa, and tarsus are harvested from a donor and trimmed during allograft placement. Some of the extra skin may be placed peripherally and used later for skin biopsy purposes. Additionally, as reported by Devauchelle et al. and Dubernard et al., a sentinel vascularized myocutaneous flap can be recovered from the donor and used for postoperative monitoring. , Biopsies of the transplanted skin are often necessary to later confirm episodes of rejection and grade their severity. Repeated biopsies may lead to scarring and compromise the aesthetic value of the face transplant. Thus a sentinel flap with similar antigenic composition serves as a biopsy site for monitoring purposes. Kueckelhaus et al. found similar grades of rejection between the sentinel flap and facial allograft–based skin biopsies.
Face transplantation procedure
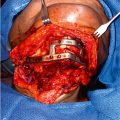
A second team simultaneously prepares the recipient vessels and removes the cicatricial tissue to minimize ischemia time. The recipient’s head and neck region are dissected carefully, and all viable vessels, nerves, and bone endings are identified and labeled. Excision of scar tissue and preparation of skin flaps are done according to the preoperative plan. Before implantation, all UW fluid in the allograft is flushed and replaced with cool lactated Ringer’s solution.
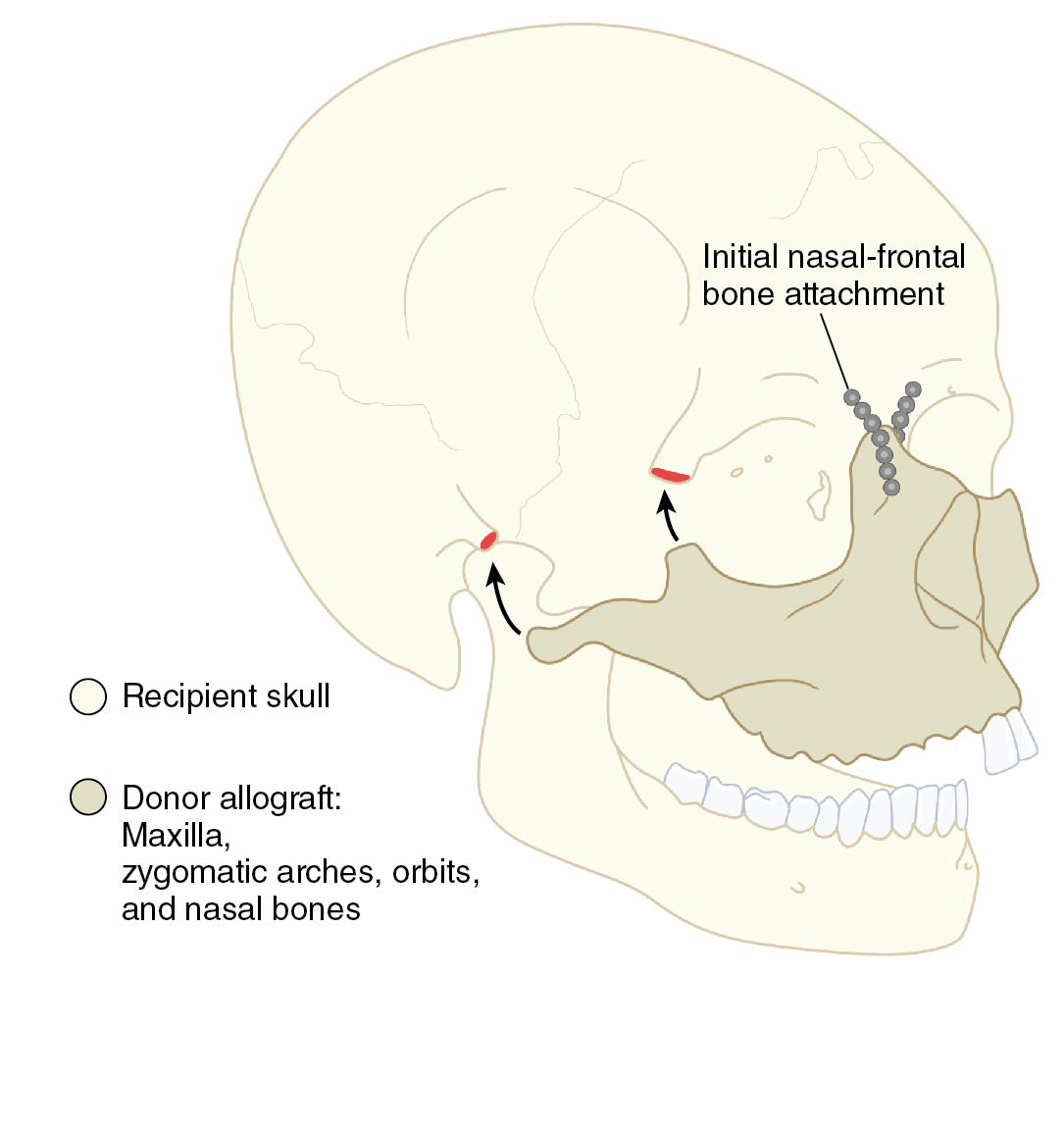
As in extremity transplantation, face transplantation involving bone begins with the osteosynthesis ( Fig. 43.1 ). Careful attention must be paid to allow the coronal, sagittal, and axial axes to be accurately positioned for facial width and projection. Surgical modification of bone components may be required in cases where there is a width discrepancy between donor and recipient bones. Dental occlusion should be checked before starting affixation of plates and wires in the bone segments. This can be somewhat challenging in the presence of poor dental hygiene and periodontal disease of the donor or the recipient. Intraoperative dental extraction in the donor may be necessary to prevent any infectious complications after the start of immunosuppressive therapy. Routine extractions of healthy teeth are not necessary. Extractions of decayed teeth may be delayed till the jaws are aligned because this will aid in maxillary and/or mandibular positioning. Because of the limited ischemia time, bony osteosynthesis can be temporized by using a limited number of plates and screws till the anastomosis of vessels is completed.