Key Points
- ▪
Current major surgical options for lymphedema treatment are liposuction, lymphatic grafting, lymphovenous shunt, and vascularized lymph node transfer.
- ▪
The surgical outcomes for primary lymphedema are not as good as those of secondary lymphedema.
- ▪
The timing of surgical treatments for various stages of lymphedema is important.
- ▪
Liposuction is effective for immediate volume reduction, but it commits the patients to wearing compression garments.
- ▪
Lymphovenous shunts are effective in the early stages but have limited use for advanced lymphedema.
- ▪
Vascularized lymph node transfer is a promising option, but its scientific rationale needs to be clarified.
- ▪
The role of surgical treatment should be redefined as complimentary to complex decongestive treatments.
Introduction
Surgical treatment for lymphedema has existed in some form since Charles reported the first debulking surgery for scrotal lymphedema in 1901. Despite this long history, treating lymphedema remains a challenging task for plastic surgeons. Surgical procedures have been refined over the decades, but we have yet to define a standard algorithm to treat lymphedema in its various stages.
Early clinical applications of microsurgery to treat lymphedema were reported in lower extremity lymphedema cases in 1967. In this procedure, called a lymphovenous shunt operation, a lymph node in the inguinal region was transected, the internal surface of this node was faced toward the lumen of the femoral vein, and the capsule of the lymph node was sutured to the venous wall. The lymphovenous shunt procedure was modified and popularized by O’Brien et al. in the 1970s but it was not recognized as a standard procedure because of variable patient outcomes.
Several new surgical techniques have been developed and applied to lymphedema patients over the past half century. Brorson and Svensson refined the liposuction technique for lymphedematous extremities and achieved reduction of limb volume to normal. Baumeister and Siuda harvested the lymph-collecting vessels from the medial thigh region and transposed them to the contralateral lymphedematous lower extremity or grafted them to the lymphedematous upper extremity. Campisi replaced the obstructed portion of the lymphatic vessels with a vein graft to bridge proximal and distal lymphatic vessels. Becker et al. reported that vascularized lymph node transfer was effective to improve lymphedema symptoms, including pain and deformity, in breast cancer-related lymphedema patients. Koshima et al. applied supermicrosurgical techniques, which are able to anastomose vessels less than 0.8 mm in diameter, to refine conventional lymphovenous shunt operations and named the new procedure lymphaticovenular anastomosis. The indications for each of these procedures, however, are unclear.
The ultimate goals of surgical treatment for lymphedema are to eliminate progressive increase of limb volume, diminish emotional distress and anxiety, and release patients from daily self-management procedures such as skin care, compression garment use, and self-decongestive therapy. However, despite extensive efforts by pioneering microsurgeons, surgical treatment for lymphedema does not fully address patient needs. This chapter focuses on current controversies in the surgical treatment of lymphedema that hamper the establishment of standard of treatment and must be overcome for the field to progress.
Etiology of Lymphedema
Lymphedema is categorized as primary or secondary. Primary lymphedema is attributable to a variety of etiologies including functional and/or anatomic deficit of the lymphatic vessel. The onset of primary lymphedema varies from congenital (birth), praecox (after birth and before 35 years of age), to tarda (after the age of 35 years). Several specific gene mutations have been identified in patients with primary lymphedema (e.g., mutations of the forkhead box C2 gene ( FOXC2 ) and the SRY (sex-determining region Y)-box 18 gene ( SOX18 ) ), and these mutations are often associated with congenital vascular malformations. Abnormal lymphatic vessel anatomy has been detected in primary lymphedema, and imaging studies demonstrate various conditions from aplasia to hyperplasia of the lymphatic vessel. Lymphoscintigraphy reveals reduced speed of lymph flow in primary lymphedema.
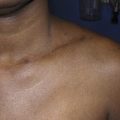
Secondary lymphedema is attributed to obstruction of lymphatic channels and an imbalance between the production of lymph fluid and its outflow from affected body regions. Lymphostasis causes reflux of lymph fluid into the dermal region; this reflux, ‘dermal backflow sign’ ( Figure 3.1 ), can be demonstrated with lymphoscintigraphy or indocyanine green (ICG) lymphography.
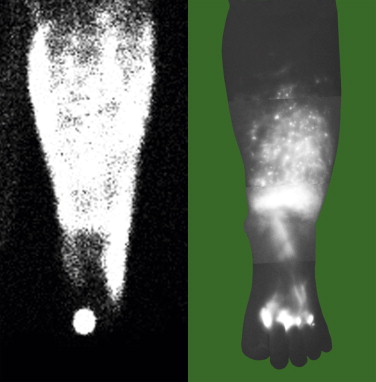
Although the volume increases seen in the affected body regions are similar in primary and secondary lymphedema, the functional and anatomic features of the associated lymphatic vessels differ. In primary lymphedema, anatomic defects of the lymphatic system are detected at various structural levels. Although the structures of the lymph collecting vessels are maintained, the ability of the vessels to convey lymph fluid centripetally is impaired. In secondary lymphedema, obstruction of the lymphatic vessel is considered the initial insult. In general, surgical outcomes for primary lymphedema are not as favorable as those of secondary lymphedema, probably because lymph vessel function is maintained in the early stages of secondary lymphedema, but systemically impaired in primary lymphedema.
Timing of Surgical Treatment
The International Society of Lymphology (ISL) classification system is used to stage lymphedema, and the stages reflect the progression of the disease. Lymphedema starts with a subclinical period called the latent phase (stage 0). It then progresses to swelling with fluid collection that is reversible with a positive pitting test (stage I). The next stage is spontaneously irreversible, with negative pitting test (stage II), and the final stage is elephantiasis with cutaneous complications (stage III). Different stages of lymphedema should require different surgical treatments.
Decongestive physiotherapy has been recognized as the standard of care for all stages of lymphedema. Some of the current practical indications for surgical treatment are progressive lymphedema that is resistant to physiotherapy, severe deformity with fibrosis, and patient choice.
Several groups have described prophylactic lymph reconstructive surgery. Boccardo et al. proposed simultaneous prophylactic lymphovenous shunt creation during breast cancer oncologic surgery to prevent future lymphedema. Koshima et al. proposed prophylactic lymphaticovenular shunt creation for unilateral lower extremity lymphedema patients who underwent subsequent gynecological cancer treatment with intrapelvic lymph node dissection. The rationale for this prophylactic procedure was that intrapelvic lymph node dissection bilaterally impairs lymphatic drainage from the lower extremities and therefore is likely to cause lymphedema in the asymptomatic limb if the contralateral limb already suffers from lymphedema. These prophylactic approaches have not become popular because patients who undergo lymph node dissection do not always develop lymphedema. However, in our experience, this surgery may be justified for asymptomatic limbs to prevent future lymphedema because it can achieve larger volume reductions in early-stage lymphedematous limbs before the lymphatic vessels lose their function.
In a study of early diagnosis of risk factors for lymphedema, Akita et al. used ICG lymphography of the lower extremities to follow postoperative gynecologic cancer patients. The authors found that dermal backflow patterns in the early postoperative period could distinguish between patients whose lymphatic condition was reversible and patients whose lymphatic condition was worsening based on types of dermal backflow pattern as an indicator of severity of lymphedema. Further follow-up will be required to understand the meanings of different dermal backflow patterns, but it is clear that imaging studies must play a key role in the evaluation of lymphedema status in addition to volume measurements.
Lymphatic microsurgery has the potential to change paradigms in lymphedema treatment. A growing body of evidence indicates that surgical treatment of lymphedema is more effective in the early stages, before extensive fat deposition and fibrosis occur. Complete decongestive therapy (CDT) is widely accepted as the first-line lymphedema treatment and demonstrates excellent outcomes in the management of skin care. However, there is currently insufficient supporting evidence to indicate that this therapy prevents lymphedema progression. Lymph reconstructive surgery is most effective in early-stage lymphedema patients, and combinations of lymphatic microsurgery with CDT for maintenance are expected to prevent lymphedema progression.
Significance of Early Detection
Early detection of lymphedema symptoms is a key to achieve better surgical outcomes. Secondary lymphedema is caused initially by lymphostasis following obstruction of lymphatic vessels, but the condition becomes irreversible after fibrosis develops and the lymphatic vessels deteriorate. It is crucial to identify patients predisposed to lymphedema during the subclinical phase and patients with very early stage lymphedema.
Cooperation between medical oncologists, oncologic surgeons and reconstructive surgeons is therefore crucial to detect early symptoms of lymphedema in cancer survivors. Besides physical examination, sensitive, specific, noninvasive methods to detect lymphedema in the subclinical phase are needed. Diagnostic protocols that are suitable to detect those predisposed to lymphedema need to be established. Identified predisposed patients may be able to skip a trial period of CDT and go directly to surgical treatment during the early stages of lymphedema, thus preventing disease progression. Surgical treatment should be considered as a first-choice therapy instead of waiting until later disease stages, when lymphedema becomes irreversible. Additionally, patients that fail CDT should be referred to surgical evaluation expeditiously.
Liposuction
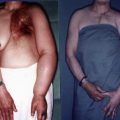
Liposuction, or suction-assisted lipectomy, was initially developed as a surgical procedure for improving body contours. In this procedure, a small metal cannula connected to a vacuum machine is inserted in the subcutaneous tissue and used to remove extra fat tissue with a sweeping motion. This motion may, however, cause damage to the lymphatic system. The possibility of this damage was investigated in a cadaver model, and investigators found that traumatic damage to the lymphatic system could be avoided if the aspiration procedure was performed in the leg’s axial direction.
Secondary lymphedema initiates as collection of extra interstitial fluid. When the disease is progressive, accumulation of adipose tissue occurs between the skin and deep fascia and fibrosis follows. Liposuction does not improve lymphatic function but is the surgical technique that can achieve complete and instantaneous reduction of volume in the affected limbs. One major drawback of liposuction as a treatment for lymphedema is that the patients have to wear custom-made compression garments for the rest of their lives to avoid recurrence. Although liposuction is categorized into the excisional procedures, it is ineffective in the advanced stage of lymphedema in which extra fat is replaced by firm fibrous tissue.
Adipogenesis in lymphedema is a major area of study. If the mechanisms of adipose tissue accumulation are elucidated and can be controlled, liposuction may be combined with other therapies to maintain good outcomes without requiring long-term compression garment use.
Lymphatic Grafting
Baumeister and Siuda were the first to describe harvesting intact lymphatic vessels from the medial thigh region as lymphatic graft material for both upper and lower extremity lymphedema treatment. The graft was either transferred as a free graft and lymphatic vessels within the graft were anastomosed to stumps of the lymphatic vessels in the neck and upper arm in patients with a history of surgical lymph node excision and upper extremity lymphedema or transposed into the medial thigh in the contralateral limb by pivoting in the ipsilateral inguinal region in patients with lower extremity lymphedema. Patency of the transposed lymphatic vessels in lower extremity cases was confirmed by lymphoscintigraphy with demonstration of functional lymph nodes in the contralateral inguinal region.
Lymphatic grafting attempts to surgically treat lymphedema by supporting or recreating the lymphatic system in lymphedematous limbs. Placing the healthy lymphatic vessels may create a new lymphatic drainage pathway. However, there are some drawbacks to this surgical approach. The major cause of secondary lymphedema in the lower extremity is oncologic treatment for gynecological cancers, and most gynecological cancer patients have undergone intrapelvic lymph node dissections and radiation treatment. Although lymphedema symptoms may be unilateral in these patients, degeneration of the lymphatic vessels can be expected in both lower limbs.
Consequently, the lymphatic vessels in the non-lymphedematous limb may not be intact, and harvesting a lymphatic graft may increase the risk of donor-site lymphedema. Furthermore, a long axial incision is required for harvesting the lymphatic graft because the surrounding lymphatic vessels must be preserved to avoid creating iatrogenic lymphedema, and this long scar may compromise cosmetic outcomes.
Lymphovenous Anastomosis
Since O’Brien popularized lymphovenous anastomosis (LVA) in the 1980s, LVA has been the hallmark of the surgical treatment for lymphedema. In the initial lymphovenous shunt procedure, three horizontal incisions were placed at the distal, proximal, and middle regions of the medial aspect of the lymphedematous limbs. Patent blue violet dye was injected a few centimeters distal from the incisions to stain the lymphatic vessels during the dissection. The incisions were placed over areas with a high-density of lymphatic vessels under normal conditions. However, the anatomic structures of the lymphatic system in lymphedematous limbs can be dramatically altered. The dye often did not work in the lymphatic vessels in lymphedematous limbs because they had lost their ability to absorb interstitial foreign substances. Thus, it was often difficult to find suitable lymphatic vessels to be anastomosed to the veins without any imaging guidance.
As a solution to this identified problem, ICG lymphography was used by Ogata et al., who reported its usefulness for LVA. In ICG lymphography, small amounts of ICG are injected intradermally; the ICG is specifically taken up by the lymphatic system and reflects infrared light, which outlines the lymphatic vessels. ICG lymphography can be used for real-time imaging and works as a beacon for surgeons to map the lymphatic vessels prior to LVA.
Koshima developed a supermicrosurgical technique that allows for anastomosis between vessels smaller than 0.8 mm. LVA is a superb lymphatic reconstructive surgery concept to reroute extra interstitial fluid to the venous circulation. However, LVA is sometimes ineffective, and the following matters need to be addressed for it to become a reliable standard of care:
Selection of Functioning Lymphatic Vessels
In LVA, a lymph collecting vessel running through the subcutaneous tissue is anastomosed to a vein. In secondary lymphedema, the lymph collecting vessels have degenerated and lost their ability to propel lymph fluid with spontaneous contractile movement.
A histological study with electron microscopy demonstrated that smooth muscle cells in the outer layer of the lymph collecting vessels in lymphedematous limbs were fragmented or had disappeared and been replaced by collagen fibers. These structural changes in the lymph-collecting vessels were more likely in the proximal regions of the extremities near the node clearance sites, and the changes in the distal region were generally minor. It is expected that the functional capacity of the lymph vessel to be anastomosed will affect the outcome of LVA.
In a prospective analysis of LVA in 100 consecutive patients 12 months after LVA, we found a mean volume reduction of 61% in early-stage upper extremity lymphedema and 17% in advanced-stage lymphedema. This study demonstrated the importance of selecting functioning lymph vessels for this procedure. We found ICG lymphography useful to select functioning lymph vessels because some lymph vessels lose their ability to absorb ICG despite having patent lumens.
Pressure Gradients Between Lymphatic Vessels and Venules
Pressure gradients in LVAs are another concern. Lymph fluid does not contain coagulation factors such as platelets. If the anastomosis is patent and there is no valve in the recipient venule, blood reflux can occur because venous pressure is higher than lymph pressure.
Olszewski et al. measured intralymphatic pressure in the lower extremities using a T-shaped cannula inserted into the proximal and distal stumps of lymphatic vessels. In his study of 25 volunteers without any lymphatic diseases, the mean systolic pressure of the lymphatic vessels was13.5±8.01 mmHg, and the mean pulse amplitude was 8.80±4.6 mmHg in a prone position. On the other hand, the mean peripheral venous pressure has been reported to be 12.4±3.6 mmHg. Intralymphatic pressure may be lower than peripheral venous pressure in the diastolic phase but slightly higher in the systolic phase. Furthermore, lymphatic pulse amplitude decreases by about 2 mmHg in patients with advanced-stage lymphedema. This physiological study suggested that LVA may not be feasible for the advanced stage of lymphedema.
Long-Term Patency of LVA
Lymphovenous shunt operations require delicate maneuvers because the lymphatic vessels have small diameters and thin walls. The walls of lymphatic vessels are thinner than those of veins, and the lumens are more prone to collapse. LVA surgery requires extreme care to not catch the lymphatic vessel’s back wall with the suture needle. Narushima et al. reported a stenting technique for making reliable anastomoses with a lower risk of lymphatic vessel collapse.
The patency of LVAs can be tested by injecting dye into the skin a few centimeters distal from the anastomotic site during the surgery. However, to date, there are no effective methods to evaluate LVA patency in a postoperative setting, and it is also difficult to confirm continued drainage of excess interstitial fluid to the anastomosed vein.
There is relatively little information available on the late patency of lymphovenous shunts. Gilbert et al. studied the patency of LVAs in non-lymphedematous dogs and observed 74% patency at one week after surgery, and 66% patency between six weeks and three months. Similarly, Puckett et al., in a study of LVA operations in lymphedematous dogs, found 100% patency at one week after surgery, 21% patency at two weeks, and 0% patency at three weeks. Thus, experimental studies using large animals have proven contradictory. Patients who have undergone LVA often have reasonable long-term volume reductions, but late patency of the lymphovenous shunts has not been confirmed in clinical settings.
Location and Number of LVA
There is no consensus regarding suitable locations for LVAs. If the anatomy of the lymphatic system is preserved in lymphedematous limbs, it is feasible to place the anastomosis in the proximal region because the lymph flow in the lumen of the lymphatic vessel is unidirectional from the peripheral toward the proximal region and one anastomosis can drain a large territory. However, valves in the lymphatic vessels become incompetent to various extents in lymphedema, and lymph reflux toward the skin (dermal backflow) is always observed in lymphedematous limbs with ICG lymphography and lymphoscintigraphy. The lymphatic vessels are more easily found in the distal limb because the subcutaneous fat is thinner and the structure of the lymphatic vessels is more preserved than in the proximal limb. LVA in the distal region may be effective in lymphedematous limbs because incompetency of valves and influence of gravity push lymph fluid toward the distal region.
There is no standard guideline for a recommended number of LVAs. Use of ICG lymphography allows for real-time scanning of the lymphatic vessels prior to LVA in the operating room. Greater numbers of anastomoses could be beneficial for reducing interstitial fluid levels and ensuring adequate numbers of patent anastomoses. Conversely, there may be a risk of worsening lymphedema if functioning lymphatic pathways are sacrificed. Despite these concerns, we have not experienced any negative postsurgical impact from multiple LVAs.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree