I. DEFINITION
A. Composite tissue consists of a “composite” of nonsolid organ tissues including skin, bone, muscle, nerve, and fat.
B. May also be referred to as a vascularized composite allograft (VCA). This is defined by the American Society of Transplant Surgeons (ASTS) as nonautologous vascularized peripheral tissues including skin, muscle, nerve, tendon, blood vessels, and/or bone as a functional unit.
C. Allotransplantation refers to the fact that these tissues are transplanted from one individual to another. This is in contrast to autologous transplantation, where tissues of an individual are moved to another location on the same individual.
II. UPPER EXTREMITY/HAND TRANSPLANTATION
A. Background
1. First hand transplantation was performed in 1964 in Ecuador, which resulted in rejection requiring reamputation.
2. Subsequent hand transplantation was reported 35 years later in France. Graft was lost after 2.5 years in this instance.
3. Now over 60 upper extremity transplants and over 150 composite tissue allo-transplants have been performed including cases of bilateral upper extremity transplantation.
B. Indications
1. Upper extremity transplantation remains experimental.
2. Patient compliance is extremely important. Patients must go through significant rehabilitation and must be adherent to immunosuppression medications.
3. Patients should be otherwise generally healthy given the health risks associated with immunosuppression and the need for intense postoperative rehabilitation
4. Patients must find use of prosthesis insufficient.
5. Body image is additionally cited as an indication for candidacy.
6. One report cites exclusion of patients with known psychiatric disorders, blindness, recent malignancy, hepatitis B/C infection, or lack of economic support.
C. Surgical procedure
1. Surgical procedure consists of procurement from the donor and transplantation to the recipient. These require large teams consisting of plastic and orthopedic surgeons, anesthesiologists, nurses, and solid organ transplant surgeons.
2. Donor procedure
a. Matching of gender and blood type is well documented, although further matching based on human leukocyte antigen (HLA) is not well reported.
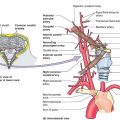
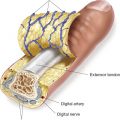
b. Upper extremity procurement from deceased donor requires amputation of the allograft proximal to the level of the defect in the recipient. The integrity of the structures vital for survival and function of the graft must be ensured, including bone, arteries/veins, nerves, and tendons.
c. A prosthesis is fitted to the deceased donor in order to avoid donor disfigurement after amputation of the allograft.
d. Must consider that deceased donor will also likely be donor for solid organs. Careful coordination with solid organ transplant procurement team(s) is required.
e. The allograft is then perfused with cold solution to avoid warm ischemia.
f. Recent trials have demonstrated that bone marrow transplantation (BMT) from donor vertebral bodies combined with immunosuppression prolongs allograft survival.
3. Recipient procedure
a. The recipient stump must be prepared for the allograft with opening of skin and preparation of the bony stumps.
b. Transplantation then proceeds in the general order of replantation, with bony fixation, extensor and flexor tendon repair, nerve repair, vein repair, and finally arterial repair.
D. Immunosuppression and rejection
1. Immunosuppression medications are required to prevent rejection of the allograft. Rejection can occur either acutely or chronically.
2. Induction immunosuppression consists of antithymocyte globulin and basil-iximab; these are anti-T cell antibodies. Maintenance immunosuppression consists of tacrolimus, mycophenolate mofetil, and prednisone.
a. Tacrolimus is associated with elevated serum creatinine levels.
b. Prednisone is associated with hypertension, diabetes, dyslipidemia, and risk of malignancy.
3. Acute rejection appears to affect the skin first. It manifests as erythematous macules and diffuse redness. Histologic analysis shows lymphocytic infiltrate consisting of T cells. It is graded according to the Banff score.
a. Grade 0 (no rejection): No or rare inflammatory infiltrates.
b. Grade 1 (mild rejection): Mild perivascular infiltration. No involvement of overlying epidermis.
c. Grade 2 (moderate rejection): Moderate-to-severe perivascular inflammation with or without mild epidermal and adnexal involvement. No epidermal dyskeratosis or apoptosis.
d. Grade 3 (severe rejection): Dense dermal inflammation associated with epidermal involvement (keratinocyte vacuolization, apoptosis, and necrosis).
e. Grade 4 (necrosis): Necrosis of epidermis or other skin structures.
4. Treatment of acute rejection consists of increased oral steroids or IV steroids, polyclonal or monoclonal antibodies, and local immunosuppressants (topical).
5. The efficacy of generic versus brand immunosuppression medications has not been determined and is of question in the solid organ literature.
6. Acute rejection can be monitored by visualization; biopsies of skin can be taken as needed.
7. Single-drug immunosuppression (tacrolimus) has been used in patients treated with donor bone marrow cells on postoperative day 14.
8. Chronic rejection has not been defined in the literature. However, it would likely include intimal proliferation of vessels, skin and muscular atrophy, and fibrosis of deep tissues.
E. Rehabilitation
1. Rehabilitation is similar to that after replantation. Passive mobilization is continued for 1 year. Patients may be started on active motion within weeks after surgery.
2. Steroid use as part of immunosuppression impedes healing and may place tendon repair at risk if active motion is performed too early postoperatively.
F. Outcomes
1. Immediate outcomes of interest include postoperative graft survival.
2. Immunologic outcomes of interest include acute or chronic rejection.
3. Long-term outcomes of interest include sensation, motor recovery (strength, function), and patient satisfaction.
4. Immunosuppression-related outcomes include development of any long-term sequelae (e.g., diabetes, hypertension, malignancy, infection).
A. Background
1. Seventeen face transplantation procedures documented from 2005 to 2012.
2. First face transplant in November 2005 in France, has since been performed in France, the United States, Spain, and China.
B. Indications
1. Significant facial deformity, although clear indications have not been delineated.
2. Facial deformity has been due to trauma (animal bites, weapons), congenital deformities, burns, and tumors.
C. Surgical procedure
1. Donor procedure
a. Surgical procedure is dependent on the structures required for transplantation.
i. May include bone, muscle, nerve, mucosa, skin.
ii. May include various aesthetic units, including nose, lips, cheeks.
b. Facial vessels or external carotid arteries reported as pedicles.
c. Facial nerve is transected and preserved distally to perform neurorrhaphy with the recipient facial nerve.
d. Consideration has been given to skin color and HLA matching.
2. Recipient procedure
a. Identify missing structures, as this guides donor operation.
b. Recipient vessels are identified.
c. Facial nerve trunks are identified.
d. Microvascular anastomosis performed between donor vessels and the recipient vessels.
e. Facial nerves are connected.
f. Sensory nerves (e.g., trigeminal branches) are connected if available.
D. Immunosuppression and rejection
1. HLA matching is reported in many of the facial transplant reports.
2. Induction regimen consisting of antilymphocyte serum (thymoglobulin) and methylprednisolone; thymoglobulin, prednisone, tacromlimus, and mycophenolate mofetil.
E. Rehabilitation
1. Rehabilitation is similar to that after replantation. Passive mobilization is continued for 1 year. Patients may be started on active motion within weeks after surgery.
2. Steroid use as part of immunosuppression impedes healing and may place tendon repair at risk if active motion is performed too early postoperatively.
F. Outcomes
1. There are multiple outcome measures. While initially interest was focused on immediate postoperative graft survival and immunologic outcomes, there has been an increasing interest in functional outcomes.
2. Two of the 17 patients have died due to lack of compliance with immunosuppression and posttransplant complications. No graft loss or GvHD.
IV. ABDOMINAL WALL TRANSPLANTATION
A. Background
1. Involves transplantation of abdominal skin and/or muscle and/or fascia for abdominal wall support.
2. Described in both pediatric and adult patients in conjunction with solid organ transplants.
B. Indications
1. Described in pediatric multivisceral transplant recipients to provide abdominal wall support.
2. Described in adult patients with small bowel transplants.
3. May avoid donor site morbidity associated with autogenous free flaps, or risk of infection with open abdomen or prosthetics.
1. May be taken with rectus muscle, fascia, subcutaneous tissue, and skin (vascularized myocutaneous abdominal wall flap).
a. Bloody supply is from the deep inferior epigastric arteries.
b. Flap can be elevated on either the DIEA or more proximal femoral or iliac vessels.
c. These are then anastomosed to the recipient’s common iliac arteries.
d. Soft tissues are then closed with the flap in place.
2. May be taken only as posterior rectus sheath fascia
a. Perfused by the falciform ligament. May be taken en bloc with liver if liver transplantation is being performed.
b. Posterior rectus sheath fascia may be used to bridge the recipient’s fascia.
D. Immunosuppression
1. Induction therapy with alemtuzumab and maintenance with tacrolimus described for vascularized myocutaneous abdominal wall.
2. Acute rejection treated with steroids.
3. Posterior rectus sheath fascia immunosuppression dictated by liver requirement and included methylprednisolone induction followed by prednisone and tacrolimus maintenance.
E. Outcomes
1. Posterior rectus sheath fascia
a. Five (out of six) posterior rectus sheath fascia recipients have survived with complete allograft incorporation.
b. No documented rejection of the posterior rectus sheath fascia.
2. Vascularized myocutaneous abdominal wall flap
a. A total of 14 patients received 15 abdominal wall grafts.
b. Two grafts loss due to vascular thrombosis.
c. Five patients alive to date, one with failed abdominal wall graft; four with intact grafts. Deaths unrelated to abdominal wall transplant.
d. Rejection treated in four patients.
V. BASIC SCIENCE RESEARCH
A. Immunology
1. Dendritic cells
a. Antigen-presenting cells (APCs) include Langerhans cells in the skin.
b. Acquire antigen in peripheral tissues.
c. Drain to lymph nodes via lymphatics.
d. Mature dendritic cells interact with T cells to induce an effector phenotype.
e. Immature dendritic cells interact with T cells and are believed to induce a tolerogenic phenotype.
2. T regulatory cells
a. CD4+CD25+FoxP3+ cell population.
b. May be induced or native (derived in thymus).
c. Inhibit effector function of T cells, mitigating rejection or graft versus host disease.
i. Express cytotoxic lymphocyte antigen 4 (CTLA4), which inhibits activity of APCs, preventing development of effector T cells.
ii. CTLA4 can bind to co-stimulatory molecules (CD80 and CD86) to activate indoleamine 2,3-dioxygenase (IDO) to deprive local environment of tryptophan and produce kynurenines, attenuating T cell proliferation.
iii. Produces IL-10 (immunosuppressive cytokine), which inhibits APC activity and promotes T cell conversion to T regulatory cells.
d. Believed to exert their effects in the lymph node and allograft.
3. Mesenchymal stem cells (MSCs)
a. Found in bone marrow, adipose tissue, umbilical cord.
b. Multipotent, for example, may develop into multiple other tissue types.
c. May inhibit generation of T inhibitor 17 (TH17) cells from naïve CD4+ T cells.
d. Mediate effects via paracrine factors and cell contact.
a. Complication associated with hematopoietic stem cell transplantation for malignancies.
b. Donor allogeneic T cells attack the host.
c. May be associated with vascularized BMT.
5. Chimerism
a. Full chimerism refers to the complete replacement of an organism’s hematopoietic cells (HSCs) with those of the donor.
b. Mixed chimerism refers to the presence of both donor and recipient hematopoietic cells in the recipient.
c. Donor hematopoietic stem cells are isolated from the donor bone marrow. The recipient is conditioned using whole-body irradiation, T cell depletion, co-stimulatory blockade, and immunosuppression followed by injection of the donor HSCs.
d. Bone marrow cells have been injected into peripheral circulation in hand transplant patients with no evidence of chimerism. Patients have been maintained on single-drug immunotherapy (tacrolimus).
6. Co-stimulatory blockade
a. T cell activation is dependent on antigen presentation by APC and interaction between B7-1/CD80 and B7-2/CD86 on APC with CD28 on T cells.
b. Results in IL-2 binding receptor and mTOR pathway activation T cell clonalization.
c. B7-1 and B7-2 have affinity for CTLA4 (see above) in addition to CD28.
d. CTLA4-Ig (belatacept) is a fusion protein between the extracellular domain of CTLA4 and IgG which may inhibit co-stimulation by binding to B7-1 or B7-2.
e. CTLA4-Ig has shown efficacy in kidney transplantation clinical trials.
f. CD40/CD40L pathway involved in B cell activation. CD40 is on B cells while CD40L is on activated T cells.
g. Leukocyte function-associated antigen (LFA)-1 expressed on memory T cells interacts with intercellular adhesion molecules (ICAMs) on APCs to stabilize interface between two cells.
B. Ischemia-reperfusion injury
1. Prolonged ischemia followed by reperfusion results in production reactive oxygen species.
2. This results in oxidization of proteins and upregulation of heat shock proteins (HSPs).
3. HSPs activate Toll-like receptors and may mediate innate immune response (e.g., neutrophils, macrophages, natural killer cells).
4. Some experimental models have demonstrated more severe rejection following longer ischemia times.
VI. ECONOMIC CONSIDERATIONS
A. Cost of disability
1. This is an emerging area of research with respect to general reconstruction. Understanding the costs of specific disabilities is complicated by various factors specific to the defect and the patient characteristics.
2. Hand/upper extremity
a. Does level of amputation associate with economic disability?
b. How does type of employment of recipient affect economic cost of transplantation (e.g., labor vs. desk job)?
3. Face
a. How is economic burden of facial disfigurement assessed? What is this economic burden?
b. What is the cost associated with multiple reconstructive procedures?
4. Abdominal wall: what is the economic burden of hernia in terms of lost productivity and reoperation?
1. What is the cost of immunosuppression? What are the costs of generic versus brand immunosuppression medications?
2. What are the costs of managing complications associated with immunosuppression?
3. What are the costs of single-drug therapy (and associated complications) versus multidrug therapy (and associated complications)?
C. Cost of surgery: these costs are related to time in the operating room and anesthesia. Cost comparisons looking at cost of surgery must also look at the cost of multiple operations if traditional reconstructive methods are used.
D. Cost of rehabilitation: analyses of rehabilitation cost should again look at the cost of rehabilitation in the case of traditional reconstructive methods as well.
E. Cost of complications: we need to first define complications, and what the rates of these complications are. This requires further long-term investigations of the current experience with each particular type of composite tissue allotransplantation (CTA) (e.g., hand vs. face). Following this, the cost of management strategies related to each of these complications must be assessed.
VII. ETHICAL CONSIDERATIONS
A. Immunosuppression and rejection
1. Do the benefits of a non-life-saving transplant (e.g., hand/face) outweigh the risks associated with immunosuppression (e.g., hypertension, diabetes, dyslipidemia, and malignancy)?
2. How do you define quality of life before and after transplantation?
3. What are the options if a patient has rejection which necessitates graft removal? Consider the implications of removing the face.
B. Patient selection
1. How should patients be selected for transplantation? What are the screening mechanisms in place?
2. Who can we justify for exclusion from candidacy for this procedure? Should economically disadvantaged individuals be excluded?
3. Should patients be screened based on employment? Are patients who perform labor-intensive jobs better candidates for extremity transplantation?
4. How can we screen patients to avoid transplanting patients who are motivated by a desire for financial gain or publicity from having this procedure?
C. Informed consent
1. As most surgeons are aware, it is difficult to inform patients about an operation with all of its potential complications. How can we ensure that patients understand the full scope of this procedure?
2. Informed consent must include not only information regarding the procedure and its immediate postoperative complications but also the need for intense physical rehabilitation (especially for upper extremity), and lifelong immunosuppression. Patients must be aware of the potential complications from noncompliance and should understand the challenges of compliance as well.
D. Procurement
1. How can we coordinate successfully with solid-organ transplant surgeons to ensure that procurement of composite tissue allografts does not interfere with solid organ procurement?
2. Will procurement of composite tissue affect an individual’s likelihood of becoming a solid organ donor upon death?
3. How can we avoid disfigurement to the deceased donor?
4. How can we inform patients and deceased donor families about the benefit they will provide with donation of composite tissue allografts?
E. Economics
1. Who should shoulder the costs associated with CTA? Should it depend on the type of allograft performed (e.g., hand vs. face vs. abdomen)?
2. Should different economic costs of patient care be shouldered by separate institutions (e.g., surgery vs. immunosuppression vs. complications vs. rehabilitation)?
3. Who are the potential payers for this procedure (e.g., patient, worker’s compensation/government/Medicare/Medicaid, hospital, employer, private insurers, NIH, military)?
PEARLS
1. Treatment of acute rejection consists of increased oral steroids or IV steroids, polyclonal or monoclonal antibodies, and local immunosuppressants (topical).
2. Prednisone is associated with hypertension, diabetes, dyslipidemia, and risk of malignancy
3. Induction immunosuppression consists of antithymocyte globulin and basiliximab; these are anti-T cell antibodies. Maintenance immunosuppression consists of tacrolimus, mycophenolate mofetil, and prednisone.
4. Acute rejection appears to affect the skin first. It manifests as erythematous macules and diffuse redness. Histologic analysis shows lymphocytic infiltrate consisting of T cells.
QUESTIONS YOU WILL BE ASKED
1. What is the most immunogenic tissue for transplantation?
Skin.
2. What is the difference between allotransplantation and autotransplantation?
Allotransplantation refers to the fact that these tissues are transplanted from one individual to another. This is in contrast to autotransplantation, where tissues of an individual are moved to another location on the same individual.
Recommended Readings
Cavadas PC, Landin L, Thione A, et al. The Spanish experience with hand, forearm, and arm transplantation. Hand Clin. 2011;27:443–453. PMID: 22051386.
Chung KC, Oda T, Saddawi-Konefka D, Shauver MJ. An economic analysis of hand transplantation in the United States. Plast Reconstr Surg. 2010;125:589–598. PMID: 19910847.
Kaufman CL, Breidenbach W. World experience after more than a decade of clinical hand transplantation: update from the Louisville hand transplant program. Hand Clin. 2011;27:417–421. PMID: 22051383.
Morelon E, Kanitakis J, Petruzzo P. Immunological issues in clinical composite tissue allotransplantation: where do we stand today? Transplantation. 2012;93:855–859. PMID: 22538449.
Rohrich RJ, Longaker MT, Cunningham B. On the ethics of composite tissue allotransplantation (facial transplantation). Plast Reconstr Surg. 2006;117:2071–2073. PMID: 16651986.
Schneeberger S, Landin L, Jableki J, et al. Achievements and challenges in composite tissue allotransplantation. Transpl Int. 2011;24:760–769. PMID: 21554424.
Siemionow M, Ozturk C. Face transplantation: outcomes, concerns, controversies, and future directions. J Craniofac Surg. 2012;23:254–259. PMID: 22337420.
Wood KJ, Bushell A, Hester J. Regulatory immune cells in transplantation. Nat Rev Immunol. 2012;12:417–430. PMID: 22627860.
< div class='tao-gold-member'>