Key Points
- ▪
Lymphedema is caused by partial or total obstruction of lymphatic collectors.
- ▪
Lymphovenous shunts have their established position in therapy of lymphedema.
- ▪
Control of infection of lymphedematous tissues, innovations of optical devices, and improved design of fine surgical instruments and sutures contribute to maintaining patency of shunts.
- ▪
Debulking of lymphedema tissue mass with normal healing can be accounted for by control of infection with antibiotics, preoperative compression therapy and better knowledge of blood vascularization of skin at various limb levels.
- ▪
Postoperative therapy after lymphovenous shunting and debulking must be a combined modality of long-term penicillin, elastic support and intermittent pneumatic compression application.
Introduction
This chapter is taking us back to the historical period of the 1960s, when the first ever surgical lymphovenous shunts in humans for the treatment of obstructive lymphedema were performed by Olszewski and Nielubowicz in Poland. At that time, it was not easy to convince the vascular surgeons that this type of surgical procedure had a future. But dynamic lymphography delineating lymphatic pathways, new operating microscopes, and fine atraumatic sutures helped us to successfully perform the first five operations in patients with lymphedema of lower limbs after radiotherapy for uterine cancer. Our results allowed us to convince others. Today, lymphovenous shunts are carried out all over the world. In addition, studies of bacteriology and immunohistochemistry of lymphedematous tissues have allowed for improvements to debulking surgeries mitigating previously encountered complications such as skin necrosis and fluid leakage. Also, this chapter will discuss the latest trends in surgical treatments for lymph stasis, which are based on the current knowledge of anatomy and physiology.
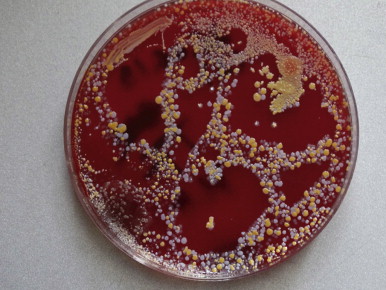
Lymphedema of the limbs is characterized by increases in the volume of the extremity caused by the accumulation of tissue fluid, proliferation of fibroblasts and adipocytes, and excessive production of collagen. There are also increases in infiltrating immune cell mass. Bacterial colonization ensues as the result of an inadequate lymphatic clearance of microbes that routinely penetrate the palmar skin of the foot and hand. Under physiological conditions, capillary filtrate-tissue fluid flows into the lymphatics and is transported via the collecting lymphatic trunks to the blood circulation. The transported fluid volume for one lower limb ranges from 20 to over 200 mL during a 24-hour period. Obliteration of the transporting lymphatic channels and sinuses of regional lymph nodes cause stasis of intercellular water, proteins, and migrating immune cells. Contractility of the lymphatics eventually disappears. These alterations lead to tissue changes, such as hyperkeratosis, fibrosis, accumulation of tissue fluid/lymph under the epidermis, and occasionally lymphorrhea. The most common complication of tissue fluid stasis is bacterial dermato-lymphangio-adenitis (DLA) and affects over 50% of patients. Early on, there was no awareness of the progressive tissue changes in lymphedema leading to the development of elephantiasis. The accumulation of mobile fluid was considered the primary process in lymphedematous tissues.
The Importance of Lymphatic Fluid Outflow
For centuries, lymphedema was considered as an accumulation of excess water with proteins that should be treated by total excision of the diseased tissue or drainage procedures comprising various types of tissue flaps bridging the lymphedematous and healthy regions. Operations designed by Charles, Sistrunk, Thompson, and Goldsmith were widely practiced with rather unsatisfactory results. Commonly occurring events included: delayed wound healing, leakage of tissue fluid from the denuded surfaces, and chronic inflammation of the tissues with continued penetrance by environmental microbes.
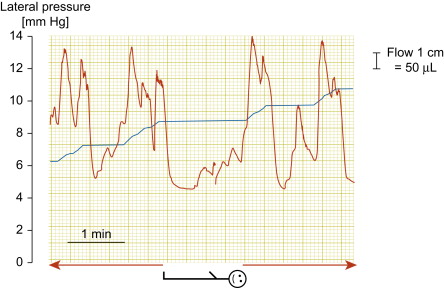
The actual understanding of the mechanism of the development of lymphedema, based on contemporary human studies, drastically changed the surgical approach to lymphedema. Modern imaging techniques of the lymphatic vascular system (X-ray contrast lymphography, lymphoscintigraphy, near-infrared lymphography, computed tomography angiography and magnetic resonance imaging (MRI)) allowed for the discovery of spontaneous lymphatic contractility that is imperative for lymphatic flow ( Figure 2.1 ), visualization of stagnant tissue fluid, and evidence for the presence of bacterial flora in stagnant tissue fluid/lymph to be controlled by antibiotics ( Figure 2.2 ). These factors made it easier not only to re-design the old types of debulking surgery, but also to propose new procedures restoring lymph flow by microsurgical anastomoses. Development of these techniques was further aided by improvements in the optics of operating microscopes and the production of ultra-thin atraumatic sutures. Additionally, long-term lymphangiographic observations highlighted the progressive and gradual obliteration of peripheral segments of collecting trunks after skin infections (erysipelas) and/or their proximal obliteration after lymphadenectomy, termed the ‘die-back’ phenomenon. This prompted surgeons to surgically intervene earlier and perform anastomoses of patent fragments of collectors with the neighboring veins.
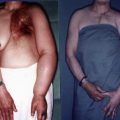
Debulking surgery, still indicated for millions of patients, has become more effective due to pretreatment with antibiotics suppressing colonizing microbes, which results in faster wound healing. Moreover, antibiotic prophylaxis can protect distal limb tissues, deprived of lymphatic drainage from chronic inflammation. High-efficiency diathermy scalpels facilitate tissue resection without using ligatures, which are frequently expulsed from the debulking wounds for months following surgery. Various types of external compression (bandages, stockings and intermittent pneumatic compression devices) have improved the long-term results of surgery, forcing tissue fluid to flow through newly formed pathways, either to veins or to non-swollen parts of the body.
Progress of Lymphedema Therapy in the Twentieth Century
The modern era of clinical lymphology and the development of treatment methods based on new knowledge of physiology of the lymphatic system dates from the introduction of oil-contrast X-ray lymphography in the 1960s. This imaging modality has set the stage for improved technologies leading to better imaging. There are several types of lymphedema including postinflammatory (postinfective), post-traumatic, and postsurgical oncologic. The indications and outcome of surgical procedures may differ with variable types of lymphedema.
Lymph Nodo- and/or Lymphatico-Venous Microsurgical Shunts
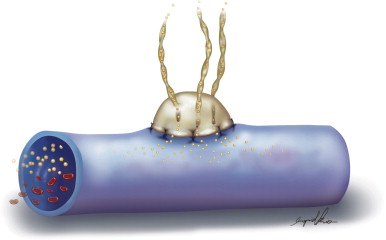
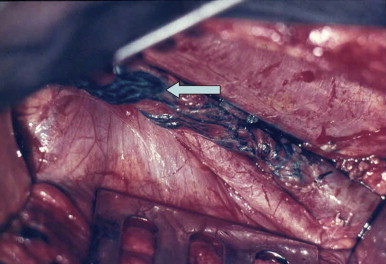
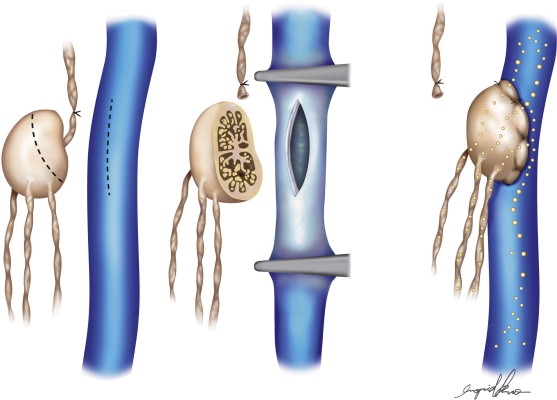
The development of vascular microsurgery in the 1960s created a basis for designing lymphovenous shunts (LVS), mimicking the natural communications between lymphatics and veins. The physiological principles of the operation were based on observations of natural anatomical lymphovenous communications in the retroperitoneal space in animals and in humans in cases of obstruction of the thoracic duct. We observed that when the lymph node was cut transversely, lymph started oozing from the cortical sinuses ( Figure 2.3 ). Such a node was implanted end-to-side into a window in the neighboring vein. The first operations were performed on dogs ( Figure 2.3 ). The mesenteric lymph node was transected and its distal part with afferent lymphatics was implanted into the inferior vena cava. Lymph flowed freely into the vein because blood pressure in the vena cava was slightly negative at inspiration. These shunts, created in dogs, remained patent throughout their life.
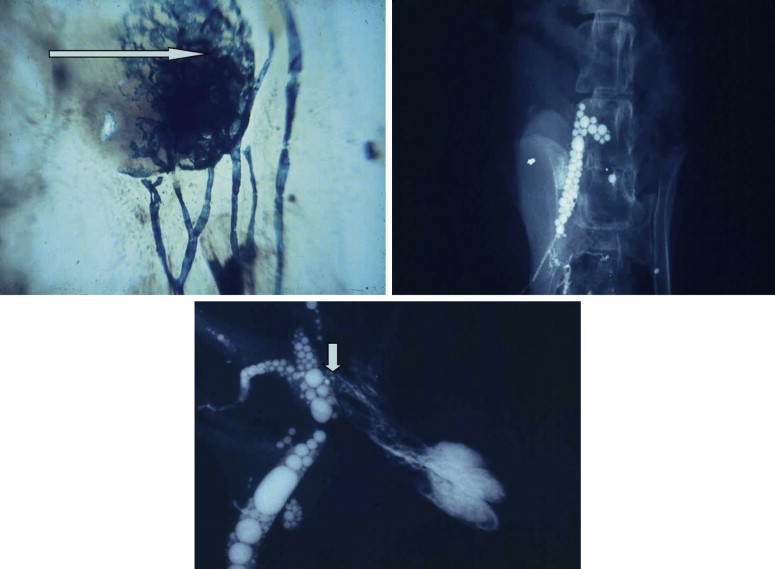
In 1966, we carried out the first five operations of microsurgical lymphovenous shunts in humans, directing the stream of stagnant lymph of the lymphedematous lower limbs to the femoral vein. The patients were women who developed obstructive lymphedema of the lower limbs after iliac dissection and radiotherapy of the pelvic region for cervical cancer. Surprisingly good results prompted us to carry out this type of operation in patients with other types of lymphedema of the lower limbs such as postinflammatory, post-traumatic and other “idiopathic” cases. Over time, various modifications of the lymphovenous shunt operations have been introduced and tried by us and other authors. Although difficult to accurately estimate, the numbers of these types of procedures are many thousands around the world. The worldwide experience in indications, technique, and results has been described abundantly in the literature.
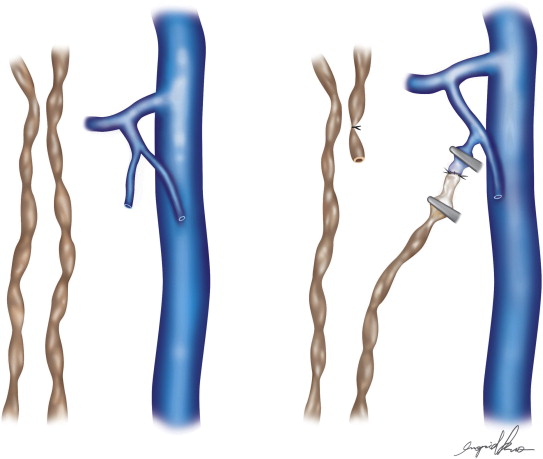
Microsurgical Inguinal Lymphovenous Shunts
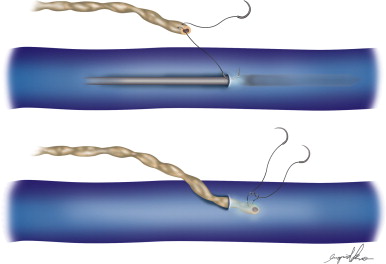
Two types of shunts were primarily performed, the lymph node–saphenous vein (LNSV) and afferent lymphatics–saphenous vein (i.e., lymph vessel–saphenous vein, LVSV) ( Figures 2.4–2.10 ). The technique has been described previously in detail. The most important factor remains the objective evaluation of results following these procedures.
Postoperative Evaluation and Results
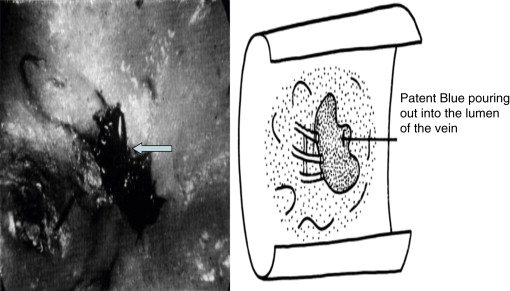
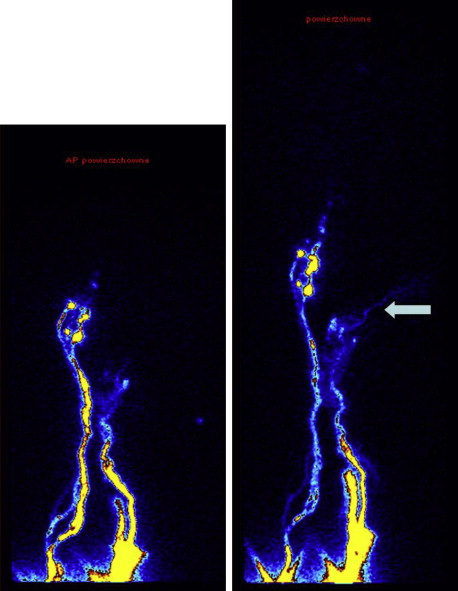
In some patients, the following tests were done as a part of a research protocol: (a) time of appearance of radioactivity in the liver after Nanocoll webspace injection (less than 30 mins in a horizontal position), (b) decreased tissue fluid pressure in leg subcutaneous tissue measured with the use of the wick-in-needle method, (c) decreased volume of the interstitial space (postoperative intrasubcutaneous fluid volume infusion test), and (d) magnetic resonance measurement of tissue water content. Evaluation was done in both limbs as temporary postoperative immobilization also brings about volume changes in a normal limb. Postoperative lymphoscintigraphic imaging of lymphatics with venous occlusion above the shunt was done in some cases. In a few, the radioactive tracer could be visualized in the draining vein ( Figure 2.11 ).
Clinical evaluation parameters were also used to assess outcomes. These measurements included: (a) decrease in leg circumference, (b) improved flexing at the ankle (to 80°) and knee joints (minimum 90°), (c) increase in the softness (compliance) of the tissues, (d) subsidence of limb pain during long-lasting upright positioning.
When evaluating five-year outcomes following these procedures, certain observations were made. Postinflammatory lymphedema was found to have improvement in 29–42% of patients with stage I and II disease. Results were lower in stage III, with no improvement seen in stage IV patients. There were no differences between the types of LNSV and LVSV shunts. In the setting of postsurgical lymphedema secondary to hysterectomy and radiotherapy due to uterine cancer and after inguinal and iliac lymph node removal for melanoma or seminoma, improvement was observed in 49–80% of stage I and II patients. In idiopathic lymphedema, improvement was seen in 0–41% of stages I and II patients. There were no investigated patients in stage III and IV as this form of lymphedema only occasionally proceeds to advanced stages. In the setting of hyperplastic lymphedema, improvement was observed in 82–100%. These patients had large lymphatics of a diameter of greater than 5 mm and did not have DLA attacks prior to surgery.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree