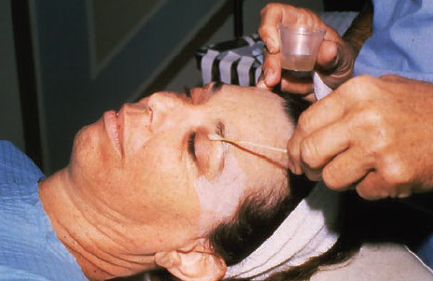
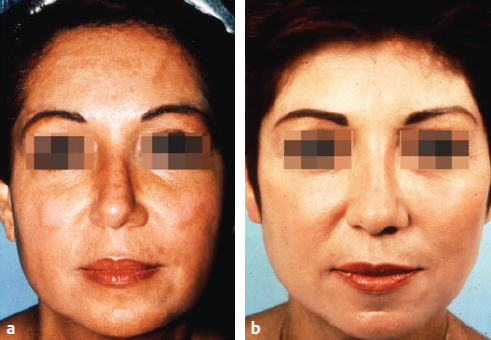
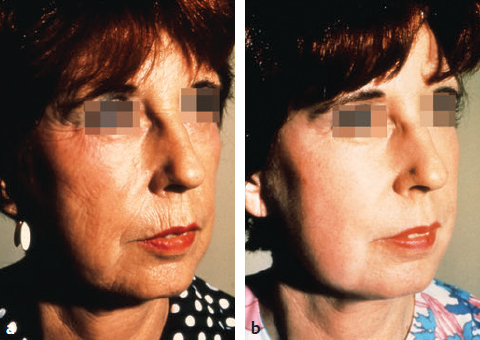
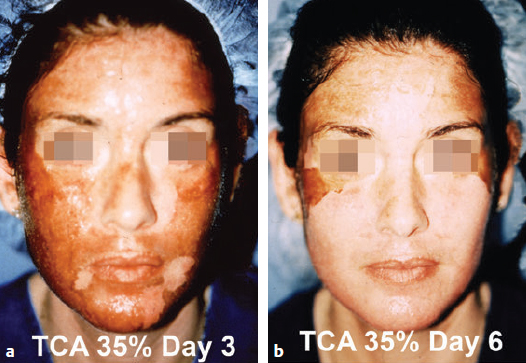
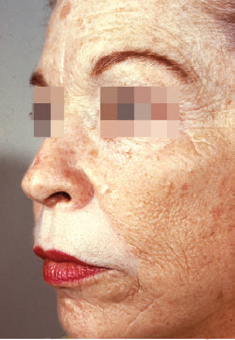
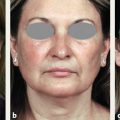
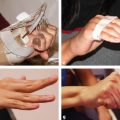
CHAPTER Chemical skin peeling remains among the most common cosmetic procedures performed by plastic surgeons and dermatologists. Chemical resurfacing has been safely and effectively used for decades to rejuvenate facial skin from the effects of aging and photodamage. Chemical peeling involves producing a controlled skin injury to promote epidermal regrowth with even melanin deposition to rejuvenate the facial skin. They are available in an ever-expanding variety of formulations that produce superficial, medium, and deep wounds. Deeper peels are the most effective in resurfacing skin but are also associated with more complications and are not appropriate for some patients. Physicians performing chemical peels should be knowledgeable about the properties, safety profile, and indications of the different available chemical peeling agents. Excellent results can be achieved in the treatment of photodamage, dyschromias, rhytides, and acne with chemical resurfacing in experienced hands. Chemical peels thus remain a mainstay modality of skin rejuvenation, even in the age of laser resurfacing. This chapter explores the most common complications associated with chemical peeling and offers recommendations for their prevention and management. Summary Box Complications of Chemical Peeling Major Minor • Contact dermatitis • Hyperpigmentation • Hypopigmentation • Hypertrophic scarring • Infections (bacterial, fungal, viral) • Cardiac arrhythmia (phenol peels) • Laryngeal edema • Milia • Persistent erythema • Pruritus • Ocular injury Achieving desired results and avoiding complications with chemical skin resurfacing begins with proper patient selection. Patients should undergo a thorough history and physical examination to identify risk factors for unfavorable results. The indications for chemical peeling should be carefully reviewed with the patient, and realistic expectations should be discussed in detail. Certain patient risk factors and contraindications exist for facial resurfacing with chemical peeling. A history of sun exposure and outdoor occupations should be reviewed with every patient. Patients must adhere to strict sun precautions starting 6 weeks before the peel and continuing postoperatively. Use of broad-spectrum sunscreens (SPF 30) and sun avoidance is critical to preventing undesirable effects such as postinflammatory hyperpigmentation, hypopigmentation, persistent erythema, and scarring.1 Patients who are not able or willing to adhere to this regimen will be at high risk for adverse outcomes and should not undergo facial chemical peeling. Active bacterial, fungal, or viral infections are an absolute contraindication to chemical peeling. Appropriate treatment should be completed before planning resurfacing procedures. In addition, patients with a history of herpes labialis are at increased risk of an outbreak postoperatively, which may result in abnormal wound healing and permanent scarring.2 All patients undergoing chemical peeling should be given antiviral prophylaxis. Patients with a history of herpes outbreaks should be followed closely and must adhere to antiviral prophylaxis. Patients with a history of atrophic dermatitis, psoriasis, or other dermatologic conditions should be screened carefully before chemical resurfacing is offered.3 These conditions can be exacerbated with chemical resurfacing. Patients with a history of abnormal wound healing, including hypertrophic scarring or keloid formation, are at increased risk for poor healing and scarring after chemical resurfacing. These patients should be screened carefully and a more conservative depth of peel should be considered to minimize the risk of postoperative scarring. A history of postinflammatory hyperpigmentation (PIH) puts patients at higher risk for this complication after chemical peeling.1,4,5 Patients should be counseled appropriately regarding this possible side effect. Patients with darker skin types (Fitzpatrick IV–VI) have a higher tendency to develop PIH.1,4 Medium and deep chemical peeling pose a higher risk for undesirable pigmentary changes than superficial peels. Patients at higher risk for PIH should undergo preoperative skin conditioning with bleaching agents, as described in the next section. Strict sun precautions must also be followed to decrease the risk of hyperpigmentation. Fitzpatrick skin types IV to VI are generally at increased risk for unfavorable results after chemical peeling.1,3 The most common side effects include pigmentary changes (transient hyperpigmentation or permanent hypopigmentation) and abnormal wound healing with potential scarring. These patients are best treated with superficial peels, or medium peels in carefully screened patients.3 Recent oral isotretinoin use before chemical resurfacing can result in severe hyperpigmentation and hypertrophic scarring because of isotretinoin’s inhibitory effects on sebaceous gland secretion, which is necessary for normal wound healing.6 The current recommendation is to stop taking oral isotretinoin at least 6 months before any skin resurfacing procedure.5 Skin should be examined for return of normal oil production before attempting chemical peeling.1 The medication history should be recorded with special attention to drugs known to have photosensitizing effects, such as estrogen-containing drugs (hormone replacement therapy, oral contraceptives) and nonsteroidal anti-inflammatory drugs (NSAIDs).1,3 These drugs should be stopped if clinically appropriate before chemical peeling to decrease the risk of postoperative hyperpigmentation. Details regarding the procedure, prepeel and postpeel care, signs and symptoms to expect postoperatively, and realistic expectations of results and possible complications should be thoroughly discussed with patients. Patients should have their questions and concerns addressed properly. Physicians must screen patients whom they suspect will not be compliant with the necessary regimen to ensure optimal results and prevent complications. Patients undergoing deep chemical peels with phenol should be evaluated for systemic diseases. Phenol has been associated with a risk of cardiac arrhythmias, and patients with cardiac, renal, or liver disease are at increased risk of systemic toxicity and should not undergo deep phenol peeling.7,8 Patients should be screened with appropriate preoperative laboratory tests and electrocardiogram during evaluation for deep phenol peeling.9 In general, elective cosmetic procedures including chemical peeling are delayed until after delivery in pregnant patients. Certain skin conditions such as melasma appear during pregnancy for which patients may seek treatment. In addition, patients may undergo chemical peeling during their first trimester before they are aware of their pregnancy. There is a lack of clinical trials investigating the safety of chemical peeling during pregnancy.10 Salicylic acid (also found in Jessner’s solution) is a U.S. Food and Drug Administration (FDA) pregnancy category C drug. Aspirin is safe during pregnancy and topical salicylic acid is available over the counter without any adverse events reported. However, its safety as a chemical peel during pregnancy has not been investigated. Topical trichloroacetic acid (TCA) (85%) has been safely used during pregnancy for the treatment of genital condylomas.11 TCA has minimal systemic absorption and is theoretically safe as a chemical peel during pregnancy; however, there is a lack of studies examining its safety. Moreover, high doses of TCA (1,000 mg/kg/day) have been reported to cause intrauterine growth retardation.12 Deep chemical peeling requires intravenous sedation with cardiac monitoring (phenol-based formulations) and should be avoided during pregnancy because of the lack of evidence-based recommendations. Given the paucity of safety trials for chemical peels, these elective cosmetic procedures should be delayed until after pregnancy. Preoperative planning with careful individualized selection of the peeling agent and concentration are critical to ensuring optimal outcomes. Patients should be given detailed instructions regarding their prepeel regimen. “Priming” the skin with bleaching agents such as hydroquinone 2% or tretinoin 0.0025% is started 2 to 4 weeks before the procedure to promote uniform peel penetration and reduce the risk of undesired pigmentary changes.3,13,14 Patients should exercise sun precautions, including daily use of broad-spectrum sunscreen. Antiviral prophylaxis should be started 1 to 2 days before peeling and be continued until reepithelialization is complete in 7 to 10 days.3,15 Patients are instructed to wash their face with mild soap and water on the night before and on the morning of the procedure. Cosmetics should not be applied on the day of the procedure. Fig. 18.1 Careful chemical peel application with a cotton-tipped applicator. Peels are applied in cosmetic units, usually beginning with the forehead. The most common superficial peeling agents include glycolic acid (30 to 70%), Jessner’s solution, resorcinol (40 to 50%), and lower concentrations of TCA (10 to 30%). The face should be cleansed with soap and water, followed by degreasing with acetone.3,16 Petroleum gel is applied to the inner canthi and nasolabial folds to protect from accidental spillage. Peels are applied in cosmetic units using a cotton-tipped applicator, usually starting with the forehead (Fig. 18.1). The peel is “feathered” at edges such as the mandibular border to prevent lines of demarcation with surrounding skin.1 Glycolic acid peels require neutralization with 10 to 15% sodium bicarbonate solution after the desired length of application.3 TCA peels are applied until an even frost appears and can be neutralized with cold water. Cool saline compresses can be applied afterward to reduce inflammation and discomfort. Products such as 30 to 50% TCA, 70% glycolic acid, and several combination peels (e.g., 35% TCA with Jessner’s solution) can achieve penetration into the upper reticular dermis. Medium-depth peels are typically used to treat Glogau category I to III skin damage16 (Fig. 18.2). Care should be taken when mixing and handling peel solutions to ensure that the desired concentration is achieved. Other precautions include having saline flushes available in case of accidental spillage into the eyes and avoiding passing any solutions over the face. Fig. 18.2 A patient with mild photoaging (Glogau I) before (a) and 1 year after (b) medium-depth chemical peel (trichloroacetic acid 35%). Fig. 18.3 Optimal results with deep chemical peels are achieved with lighter skin types (Fitzpatrick types I–II). This patient with deep facial rhytids preoperatively (a) is shown 1 year after (b) full-face resurfacing with a phenol peel. Deep chemical peels provide the best results for treating severely photodamaged skin (Glogau categories III–IV) in appropriate patients (Fig. 18.3). Formulations that achieve deep peels include classic Baker-Gordon’s formula, 50% TCA, and combination peels with lower concentrations of phenol or TCA, including Hetter’s croton oil–phenol formulation.16–18 The risk of adverse outcomes is greatest in this category of chemical peels. Monitored anesthesia care with continuous vital sign and cardiac monitoring provides the safest setting for deep chemical peeling. Phenol peels should be applied slowly, with a 10- to 15-minute pause between cosmetic units to minimize systemic toxicity.19 Operating suites are adequately ventilated to remove phenol fumes. Intravenous hydration is also recommended to prevent increased systemic levels of phenols and prevent toxicity, including cardiac arrhythmias. Patients should be monitored for at least 1 to 2 hours postoperatively. Proper wound care after chemical peeling is essential to ensure adequate healing and optimal results. In the immediate postpeel period, patients should only use mild soap to clean their face. It is imperative that patients avoid scratching, peeling, or picking at their skin to prevent mechanical trauma and poor results. Cold compresses, calamine solution, and antihistamines may help reduce itching and burning.16 During the reepithelialization period (7–10 days), topical agents should be withheld because of the risk of irritant dermatitis. After reepithelialization, sunscreen and mild moisturizers are restarted. Continued sun precautions are critical at this stage to prevent uneven pigmentary changes. Patients should be followed closely to ensure proper wound care and detect complications early to implement appropriate treatment. Patients may experience pruritus during the reepithelialization process after facial chemical peeling. Patients should be closely monitored to detect any warning signs associated with pruritus, such as persistent erythema or vesiculopustules, which may indicate a dermatitis or skin infection requiring prompt treatment to prevent postinflammatory hyperpigmentation or scarring. Cold compresses and calamine lotion can be helpful in reducing pruritus.3 A short course of topical steroids and oral antihistamines may be required to reduce itching. Patients should be counseled not to scratch their skin after peeling to prevent mechanical trauma that may take weeks to resolve. Erythema is a normal characteristic of the healing phase after chemical peeling (Fig. 18.4). The duration of the erythema depends on the depth of the peel. Superficial and medium-depth peels usually produce erythema for 7 to 14 days, but erythema can last up to 3 months after deep peels. Erythema that persists beyond the normal course can be a sign of delayed healing and potential scarring.1,4 Topical tretinoin and oral isotretinoin use in the perioperative period has been implicated as a cause of persistent erythema and scarring, particularly in medium and deep peels.1,6 Certain individuals may have genetic susceptibility to persistent erythema. If the erythema is consistent throughout the resurfaced area without skin thickening, then patients may be reassured that it will resolve spontaneously. Skin induration in the setting of persistent postpeel erythema is a warning sign for the development of permanent scarring. Patients should be treated with topical corticosteroids; systemic and intralesional steroids may be required in certain cases.1 Pain, burning, or pruritus along with erythema may indicate the development of contact dermatitis, irritant dermatitis, or infections. Exposure to allergens and irritants from commonly used topical agents may result in contact dermatitis after chemical peeling. Type IV delayed hypersensitivity reactions after chemical peeling are uncommon and result from loss of the epidermal protective barrier, exposing the dermis to antigens.5 Although rare, they typically develop during the reepithelialization process but may present up to 4 weeks after peeling. Topical antibiotics, cleansers, moisturizers, and even sunscreens have been implicated in allergic reactions after skin resurfacing with chemical peeling.1,5 Irritant contact dermatitis is more common than allergic reactions and may occur during reepithelialization or in newly epithelialized skin. Irritants may include a variety of topical agents such as tretinoin, glycolic acid, bleaching agents, fragrances, and other common ointments.1,5 It is thus important to counsel patients not to start any topical products other than the prescribed regimen. Patients may experience burning, pruritus, prolonged erythema, swelling, and skin eruptions. The presentation of contact dermatitis may be indistinguishable from infections and thus cultures are necessary to differentiate the two. Discontinuation of all possible offending agents is the first and most important step in management. Cool compresses are helpful in relieving symptoms of pain and itching. Oral antihistamines are used if allergic dermatitis is suspected. Judicious use of topical corticosteroids is indicated to decrease inflammation and prevent severe irritation that can lead to delayed wound healing and hypertrophic scarring.5 Fig. 18.4 Normal desquamation and erythema observed during reepithelialization 3 days (a) and 6 days (b) after a medium-depth chemical facial peel (35% trichloroacetic acid). Hyperpigmentation is among the most common side effects of facial chemical peeling, occurring in up to 30% of patients.1,4,5 It may become apparent as early as the first week after peeling, or up to 2 to 3 months later.4 In most cases the hyperpigmentation is transient, but long-term sequelae can result if not treated appropriately. Patients should be made aware that nevi and solar lentigines might become more prominent in the initial postpeel period.1 Patients with darker skin types (Fitzpatrick types IV–VI) and those with baseline dyschromia, melasma, or a history of PIH are at an increased risk of this complication.1,4,5,9,20 Postoperative hyperpigmentation may also occur in lighter skin types (Fitzpatrick types I–III) with excessive sun exposure before or after chemical peeling.1,5 Certain photosensitizing drugs including NSAIDs and oral contraception may increase the risk of PIH. As with most chemical peel complications, deeper peels are associated with a higher risk for undesirable pigmentary side effects. Careful patient selection and medical history is important in identifying those patients with increased risk for hyperpigmentation. Prevention includes a strict regimen of sun exposure avoidance and use of broad-spectrum sunscreen 6 weeks (or longer in high-risk patients) before the peel.5 Preconditioning with bleaching agents such as hydroquinone or tretinoin starting 2 to 4 weeks before the peel is recommended in higher risk groups, especially patients with darker skin types.1,5,13,14,20 In two separate small trials, 2% hydroquinone was more effective in preventing postpeel hyperpigmentation than 0.0025% tretinoin.13,14 After chemical peeling, strict sun precautions are necessary to prevent and treat hyperpigmentation. Patients can be reassured that the hyperpigmentation is typically transient even without treatment. Topical bleaching agents can be used after reepithelialization, but excessive or prolonged use can cause irritant dermatitis.1,5 These agents include hydroquinone, kojic acid, retinoic acid, azelaic acid, and ascorbic acid.1,20 In some resistant cases, superficial peeling with glycolic acid (30 to 40%) or salicylic acid (30%) is indicated to hasten resolution.1,5 Hypopigmentation is a normal phenomenon of chemical skin resurfacing as old photodamaged skin is replaced by lighter skin. Darker-skinned patients (Fitzpatrick types III–VI) may also experience transient hypopigmentation while their skin regains its pigment.5 True permanent hypopigmentation is a major complication seen with deep chemical peels.1,4 It is usually a late complication that can become apparent months after skin resurfacing5 (Fig. 18.5). Permanent hypopigmentation is believed to be secondary to loss of melanocyte function or dermal fibrosis with deep peels that reach the reticular dermis.1,4,5 Darker-skinned patients undergoing medium or deep chemical peeling are at risk for this complication. Typically this is most apparent at the jaw border where a line of demarcation is seen between the resurfaced facial skin and the neck (Fig. 18.6). Strategies to minimize the risk of permanent hypopigmentation are limited. Use of broad-spectrum sunscreen and sun avoidance before and after the peel are important in preventing any undesired pigmentary changes. Careful selection in both the patient and depth of peel are the most important determinants in mitigating the risk of permanent hypopigmentation. For patients with darker skin types (Fitzpatrick III or greater), a more conservative depth of peel should be strongly considered. “Feathering” the edges of the facial peel with a lower concentration peel has been advocated to minimize these lines of demarcation.1 Subsequent superficial chemical peeling can be used to attempt to even out the pigmentary changes at the edges from a prior deep peel.5 In most cases, treatment is difficult. Continued use of sunscreen and makeup may be the only options for patients.
18
Chemical Peels
Avoiding Unfavorable Results and Complications of Chemical Peels
Preoperative Planning and Patient Selection
Patient Selection
Sun Exposure and Occupation
Infections
Inflammatory Dermatoses
Abnormal Wound Healing
Postinflammatory Hyperpigmentation
Darker Skin Types
Isotretinoin Use
Photosensitizing Drugs
Unrealistic Expectations or Noncompliant Patients
Systemic Disease
Pregnancy
Technique
Preparation
Application of Chemical Peel
Superficial Peels
Medium-Depth Peels
Deep Peels
Postoperative Care
Postoperative Complications
Pruritus
Persistent Erythema
Contact Dermatitis
Hyperpigmentation
Hypopigmentation
Plastic Surgery Key
Fastest Plastic Surgery & Dermatology Insight Engine