CHAPTER 46 Animal Models of Lymphedema
KEY POINTS
Various animal models of lymphedema have been described.
Most animal models are secondary (injury-induced) lymphedema.
There is no perfect model, but available models can mimic the various stages of the disease process.
Large animal models are most useful for anatomic and histologic studies but are limited by costs and the availability of molecular reagents.
Small animal models are cost efficient and have widely available molecular reagents but are limited by the potential for surgical interventions.
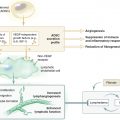
Animal models of lymphedema have long been sought to increase our understanding of this disabling disease process. Historically, the goal of most models was to mimic secondary lymphedema, in which the lymphatic system was injured and lymphedema subsequently developed. However, recent advances in molecular biology have identified some genetic models of lymphedema that mimic primary (or congenital) forms of this disease.
Validity of Animal Models
In general the currently available models of lymphedema are useful to study the pathologic process of this disease (Table 46-1). However, there are no perfect models that accurately match the clinical phenomena. All the available models have various deficiencies, including short-term and spontaneously resolving lymphedema, the need for extensive surgical manipulation, inherent animal morbidity, and additional manipulations such as radiotherapy or sclerosing agents to cause lymphatic injury, and the length of time required for the development of sustained lymphedema. These deficiencies occur because the lymphatic system has a significant built-in redundancy and excess capacity, the potential to regenerate and develop collateral vessels after injury, and the development of spontaneous lymphatic bypass channels (for example, spontaneous lymphaticovenous anastomoses) after injury.
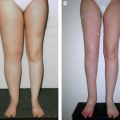
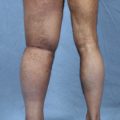
Understanding the natural history of clinical lymphedema also illuminates why it is difficult to develop animal models of lymphedema. In developed countries, secondary lymphedema occurs most commonly after lymph node dissection for cancer treatment. In these procedures the lymph nodes and deep collecting system of the limb are damaged; however, the remaining soft tissues and superficial lymphatic network remain intact. As a result, the mild edema that develops in most patients immediately after surgery almost always resolves in a few weeks, and the affected limb is clinically indistinguishable from the normal limb. In a subset of patients, approximately 10% to 30%, chronic lymphedema develops months (and sometimes years) later either spontaneously or precipitated by a secondary insult, such as radiotherapy, infection, or trauma. 1 In these patients, lymphedema is typically progressive and requires continuous management.
The delayed onset of lymphedema (in a subset of patients) is critical to the inherent difficulties in the development of animal models, because the vast majority of researchers have tried to develop models in which chronic lymphedema forms immediately after surgery in 100% of animals. This goal does not match the clinical natural history of lymphedema (that is, delayed development in a subset of patients) and as a result has complicated the development of suitable models. Many of these models necessitate extensive surgery or the addition of other interventions, thereby increasing their complexity and morbidity. Nevertheless, various models have been described, which have been useful to study the pathologic findings of lymphedema. As such, the goal of researchers who study the pathologic processes of lymphedema is to select an animal model that balances animal morbidity, husbandry costs, and the availability of molecular reagents.
Surgical Models of Lymphedema
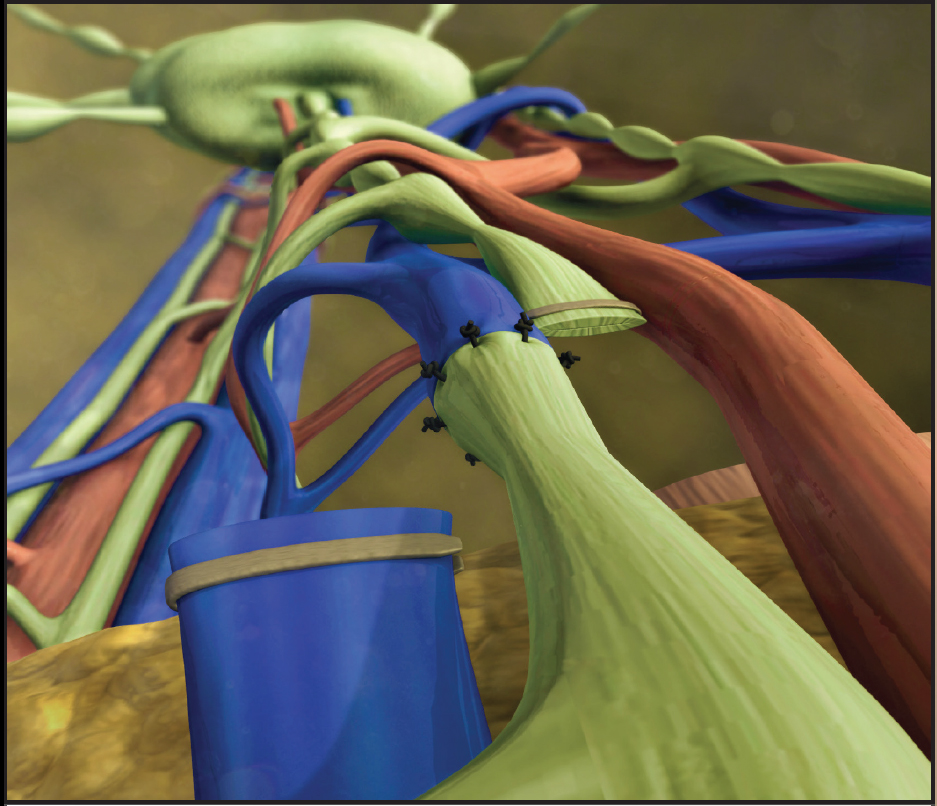
Several reports have detailed the surgical techniques required to develop various animal models of surgically induced lymphedema in a large number of species, including dogs, 2 – 6 sheep, 7 rabbits, 8 rats, 9 – 11 and mice. 11 – 13 In general, these models involved injury to the lymphatic system (usually both the superficial and deep lymphatics) and can be used for the histologic and radiologic study of the pathologic process of lymphedema (larger animal models) or with molecular techniques (small animal models).
CANINE LIMB MODELS
The use of dogs to develop models of chronic lymphedema has a long history and dates back to Halsted, 14 who was interested in this disease after he popularized the radical mastectomy. However, despite multiple attempts, Halsted was unable to develop a chronic model of lymphedema that was sustained over the long-term despite extreme surgical resections. Reichert 15 and Drinker et al 2 expanded on these efforts and removed the lymph nodes and deep soft tissues. Drinker et al repeatedly cannulated the deep lymphatics with sclerosing agents. Eventually, chronic lymphedema developed in a subset of animals at the cost of severe surgical morbidity and mortality and in most cases, multiple surgical interventions. Olszewski et al 3 understood the importance of the superficial lymphatic network and described a canine model in which the deep lymphatics were ligated at the midthigh, and a circumferential strip of skin, subcutaneous tissues, fascia, and periosteum was excised (to remove the superficial lymphatics), leaving a gap for healing by secondary intention. They followed 23 dogs over 1 year and found that all animals had initial edema that resolved in 6 to 8 weeks. However, in 8 of 23 animals (that is, 35%), chronic lymphedema developed 7 to 10 months later, which was confirmed by limb volume measurements, histologic findings, and radiologic analysis.
Danese et al 4 reported a canine hindlimb model that combined surgery and radiation treatment. In this model, the animals were initially treated with 1500 rad from the midthigh to the right lower quadrant, followed by surgical excision 2 to 4 weeks later. During surgery, the skin flaps were elevated from the abdomen to knee and muscle connective tissues, and the periosteum was removed, leaving only the skin bridge and neurovascular bundle intact. The deep lymphatics were visualized and removed for a length of at least 5 cm. These animals had initial edema that resolved, and approximately 50% developed chronic lymphedema at a later stage. Chen et al 5 also described a radiation and surgical model in which the animals for the most part developed modest lymphedema (15% increase in volume). However, the combination of surgery and radiation was morbid, with postoperative infection and mortality rates of about 30%.
Suami et al 6 reported on lymph node dissection of a canine forelimb model. In this model, which used detailed radiologic methods and indocyanine green (ICG) lymphangiography, they described the lymphatic anatomy of normal dogs and also the anatomy 6 months after axillary lymph node dissection. Although axillary lymphadenectomy in this model did not result in long-term differences in limb volume or overt lymphedema, they noted a rerouting of the lymphatic system in the axilla, with bypass of the damaged lymphatic vessels through the superficial network.
In general, the main reason for the use of dog models is the relative size of the lymphatic vessels and supporting structures, enabling the identification, cannulation, and radiologic analysis of the lymphatic tree with or without injury. The limitations of the described dog models include the morbid procedures necessary to produce lymphedema, the time required for lymphedema to develop, the development of lymphedema in only a subset of animals, the high costs of canine experiments (for example, animal husbandry and surgical procedures), and the lack of molecular reagents for canines.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree