Introduction
The vulva is a poorly described and understood part of a woman’s body. Part of this may reflect historical social construct: the dictionary definition of male genital anatomy describes the function and action of the organs while the corresponding definition for female genital anatomy refers to the location only.
This chapter describes the anatomy, physiology and function of the vulva and then considers where this is placed within the general understanding of women and the medical community.
Anatomy
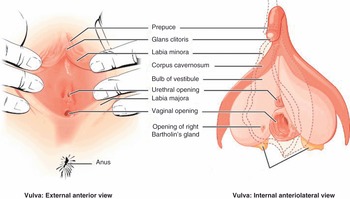
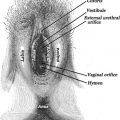
The vulva can be considered to be the area bordered by the mons pubis at the base of the abdomen, the anus and the folds of the thighs on either side. Contained within this area are the labia majora, and labia minora which borders the central vaginal opening, known as the introitus, and the urethral opening or meatus. The clitoris sits in the midline above the urethral meatus. The area between the vaginal introitus and anus is known as the perineum and is the meeting point of several pelvic floor muscles.
To understand the anatomy and function of the vulva in adulthood it is useful to revisit the embryological development of this area. While genetic sex is determined at conception, development of the genital area into a typical female or male system does not take place until around 8 weeks of embryonic life. This is triggered by the sex-determining region of the Y chromosome (SRY) gene, which leads a cascade of events resulting in genital development.
Internal Genital Development
The genital duct system, which in adult females comprises the uterus and Fallopian tubes, and the vagina, consists of the Müllerian duct (MD) and the Woolfian duct (WD) systems. These are each a set of paired tubes and are precursors of the female and male internal genital anatomy respectively. Both coexist initially in all fetuses, until the action of the SRY gene on the developing testes leads to expression of anti-Müllerian hormone (AMH), resulting in repression of the MD with only the WD remaining. This then allows the male genital internal anatomy to develop, with regression of the MD and any potential female structures. This usually takes place between 7 and 8 weeks of fetal life. However, in the absence of AMH the MD will persist and proliferate in the female fetus, proceeding to form the internal structures typically seen in females, at around 8–9 weeks.
The two MD tubes join in the midline, with the internal walls at the points of contact dissolving, forming the uterus, cervix and upper two-thirds of the vagina. The free ends at the top of the tubes proceed to form the Fallopian tubes. The ovaries form separately from the MD, on the gonadal ridge near the diaphragm. They descend towards the pelvis, finally lying adjacent to the Fallopian tubes on the pelvic side wall.
The External Genitalia and Urinary Opening
The vulva develops from a membrane and fold between the legs known as the cloacal membrane and cloacal fold. These proceed to form a bud in the midline, known as the central genital tubercle with paired folds and swellings lying on either side of the midline. These differentiate into the clitoris, the labia majora and the labia minora respectively. The labia majora appear flattened and the labia minora are tiny folds of skin on either side of the midline. The clitoris itself develops two paired corporal bodies which meet at the clitoral glans, at the tip of the clitoris. The glans has a covering of skin, the clitoral hood, which runs down to meet the labia minora on either side. The clitoral hood is derived from a layer of cells which develops at the top of the clitoris. Further genital and anal folds also develop, resulting in a separation between the anus and combined urethral and vaginal opening, which further divides into the urethra and the vagina. This sequence of events results in the usual three separate openings onto the female genital area (see Figure 2.1).
Figure 2.1 The vulva showing the urethra, vagina and anus, and the internal view with the clitoris and accessory glands.
The lower third of the vagina moves upwards to join the upper vagina, followed by dissolution of the meeting point, allowing the vagina to become a continuous tube leaving just the circumference behind, which is the hymen.
By 20 weeks of gestation, the female genitalia are developed [1].
Variations of Fetal Vulval Development
Male external genital development is mediated by androgen hormones, including testosterone expressed from the developing testes, which act upon the androgen receptor (AR), resulting in development of the central genital tubercle into a penis enclosing the urethra, and changing the genital folds into the scrotum. The testes pass through from the abdomen down into the scrotum.
A female fetus therefore can develop a more typically male appearance if higher than usual levels of androgen hormone are expressed. This leads to enlargement of the clitoris and lack of complete separation of the urethra and vagina, resulting in only one opening to the outside rather than the more usual two. Conversely, a lack of typical male development may occur if androgen hormones are not expressed or recognised by the developing genetic male fetus. Interestingly, there appears to be a continuum in anatomical development between the level of androgen hormone and the distance between the base of the clitoris and the female urethra. Females who are least able to recognise androgen hormones develop a longer distance between the clitoris and the urethra, whereas those with a higher level of hormones will have a urethra which opens very close to the clitoris [2].
This therefore illustrates how females with increased androgen hormones during pregnancy will develop a uterus and vagina, as determined by the absence of the SRY gene, but also enlargement of the clitoris due to the action of androgens on the central genital tubercle. Where this is very marked, babies may be mistakenly assigned male sex, with the clitoris appearing to look like a penis, and only later presenting with a salt-wasting crisis characteristic of congenital adrenal hyperplasia.
For those with a Y chromosome, if the gonads (future ovaries or testes) have not developed in the usual way and are non-functional, no response can be mounted to the SRY gene and therefore AMH will not be expressed. This allows the development of the MD, regression of the WD and results in internal typically female structures. Similarly, such gonads will be unable to express androgens and therefore also unable to promote a typical male external genital appearance, leading to a female appearance externally. This would result in the birth of a baby girl who has XY chromosomes.
Development during Childhood and Adolescence
The genital tract remains essentially quiescent from birth and during early childhood. The effect of maternal oestrogen slowly decreases for approximately 6 months after birth. Occasionally an infant may have a small uterine bleed as a withdrawal bleed while the effects of maternal oestrogen wear off. For those who are born preterm, an isolated clitoromegaly may be observed and will persist throughout childhood [3]. Once any other causes for this have been excluded, parents can be reassured that this is a variant of normality and no treatment is indicated.
During childhood the labia minora remain very thin and small, and the vulva generally has a flattened appearance. With the onset of the first hormonal signals coming from the brain to the ovaries gradual changes to the genital tissues occur. These hormonal pulses start from age 8 onwards, and slowly increase in size and frequency. The action of androgens on the tissues results in pubic hair developing over the mons and labia majora. The labia minora elongate and darken in colour, and usually protrude beyond the labia majora. The mons pubis and the labia majora slowly develop fat pads. The clitoris enlarges with further development of the paired corpora, which run for several centimetres inside the pelvic bone, at the pubic rami. The clitoral glans enlarges and is covered by the developing clitoral hood, which becomes more prominent while running on to meet the labia minora.
The vulva takes many years to develop fully. Changes in the labia minora are often observed first, along with pubic hair development, as a result of the effect of androgen hormone. The labia majora fat pads may take many years to develop fully, leading to a relative prominence of labia minora as part of the appearance of the vulva. Tanner described pubic hair development, which is still used today as a means to gauge pubertal development [4]. However, labial anatomy has never been described in detail throughout puberty in the manner of breast development, limiting knowledge regarding normal pubertal development [5].
One study assessed genital dimensions in prepubertal children from 1 month to 10 years, and developed algorithms to predict expected measurements according to age, weight and height [6]. For older children, significant variation continues to be seen in the external anatomy [7] (see Figure 2.2). A study assessed 58 girls and observed a linear relationship between age and genital dimensions. The clitoral hood was noted to be a separate structure from the labia minora in all age groups, reflecting its embryological origins. A variety of shapes were noted, being variously described as a horseshoe, trumpet, coffee bean or tent. All of these papers illustrated wide diversity in genital dimensions, yet the information is promoted as having applicability for those seeking to alter the vulval area for cosmetic reasons [6].
The Adult Vulva
Once fully developed, the adult vulva consists of the mons pubis, which leads to the labia majora on either side of the vaginal introitus. Situated laterally on each side of the introitus are the labia minora. The labia majora consists of hair-bearing skin, and androgens will promote growth of darker and coarser pubic hair. This will grow up to the labia minora, which have a mucosal surface on both sides. Pubic hair also extends over the mons pubis and often onto the top of the thighs. The labia minora vary in appearance but tend to have a rugose texture and are darker than the surrounding tissue. A wide variety of dimensions have been described in the literature, but it is not unusual for labial width to measure up to 5 cm at the widest part, and sometimes beyond [8]. It is also common and usual for the labia minora to protrude beyond the labia majora, which is not necessarily understood by girls and women [5].
Surrounding the vaginal introitus and urethra are various glands, often known as Skene’s glands, Bartholin’s glands and vestibular glands. Skene’s glands are situated just laterally to the urethral meatus, with Bartholin’s glands being placed at 5 and 7 o’clock on the vaginal introitus. The glands of the vestibule occur at various positions around the introitus, just up to the level of the hymen. The clitoris is situated approximately 3 cm above the urethral meatus, which is tucked just on the anterior vaginal wall [8]. The blood supply to the vulva is derived mainly from the pudendal artery, with the inferior epigastric artery supplying the mons pubis. The innervation to the vulva and its component parts is largely supplied by the pudendal nerve, arising from S2–4, although the ilioinguinal nerve (L1) and the genital branch of the genitofemoral nerve (L1–2) provide sensation to medial and lateral vulval skin.
The adult clitoris can reasonably be considered as an iceberg, with only the tip being visible. The external portion consists of paired corpora which contain erectile tissue, capped at the end by the clitoral glans. The clitoral hood covers the glans, and is more retractile than in childhood. It proceeds to meet the labia minora at the clitoral frenulum, at an angle clearly defining the start of the labia. The external clitoral body measures 2–4 cm in length and divides into two internal crura just under the pubic arch. Internally these extend laterally and inferiorly, running along the inferior pubic rami, with the crura extending for up to 9 cm (see Figure 2.1). Coursing underneath, two suspensory ligaments extend to join the mons pubis. A deeper ligament complex extends from the symphysis pubis to join the body and bulbs of the clitoris. When described, these ligaments were found to be considerably larger than had previously been documented, and differed in shape, extent and orientation from any analogous structures in the penis [9]. The nerve supply to the clitoris is derived from the pelvic, pudendal and hypogastric nerves, with the clitoral nerve running along the inferior pubic rami accompanying the crura of the clitoris. The nerves fan out around the clitoral glans, and perforate the covering tunica of the corpora.
Postmenopausal Changes
The average age at menopause in the United Kingdom is 51, with a range of 45–55 years. Vulval changes after this time reflect waning oestrogen levels, and include a loss of labial fat pads and a gradual sparseness of pubic hair. The labia minora will experience thinning and will decrease in size with loss of architecture and detail [10]. The vagina is shortened and appears pale due to a reduction in vaginal epithelial layers. The vaginal mucosa can become dry and have a reduced response in producing lubrication during sexual activity. Many of these changes can be mediated by the use of topical oestrogen.
Physiology
The development of the whole genital area during fetal life, with an upper uterus and vaginal section descending to meet the external developing vulva, is reflected in the subsequent adult functioning of the female genital tract. Generally speaking, the upper genital tract, including the upper two-thirds of the vagina, is more concerned with reproductive function, whereas the lower and external parts are associated with sensation and sexual pleasure.
The clitoris and labia minora are rich in innervation and blood supply and contain erectile tissue, all of which are of prime importance in sexual sensation and contribution to orgasm.
The clitoris itself is a dynamic organ which becomes engorged with blood during sexual arousal. Paired clitoral arteries supply blood to the glans, with cavernosa vessels supplying the corporal system. During arousal the cavernosa fill with blood, with venous valves closing, thereby preventing drainage and facilitating erection, contributing to sexual pleasure and achievement of orgasm. After orgasm the valves open, allowing the venous flow to return to the internal pudendal vein.
The labia minora contain erectile tissue which also becomes engorged with blood during sexual arousal. Oestrogen receptors have been demonstrated on the labia minora along the free edges [10]. One study assessed 62 women and asked them to rate various parts of the vulval area on the basis of anatomy and sexual sensitivity [11]. The majority of women rated the labia minora and majora as average-sized, with 13% rating the labia as large and 5% as small. Sensitivity for the labia increased towards the vagina, with the medial edge of the labia minora being the most sensitive area. Sensitivity did not vary according to self-reported size, although those with larger labia reported higher ratings for sexual pleasure. It may be that as more erectile tissue was present, more stimulation resulted in higher levels of sexual pleasure.
In contrast to the clitoris and labia minora, the labia majora become thinner during sexual arousal, which opens the introital area and thereby promotes vaginal penetration. The accessory glands of the vulva contribute to sexual function by producing fluid during arousal and aiding lubrication for penetrative intercourse.
During sexual activity and arousal, the vagina lengthens and expands around the cervix to provide a receptacle for sperm, facilitating passage through the cervical canal and onto the Fallopian tubes with anticipated reproductive consequences. Magnetic resonance imaging (MRI) has shown that the vaginal walls enlarge with fluid during penetrative intercourse, with the anterior vaginal wall lengthening and the uterus being elevated [12]. This may contribute to positioning the cervix more closely to the posterior fornix where sperm may be deposited.
There is no evidence for the existence of the Grafenberg spot (G-spot), which was thought to be a distinct structure intimately related to sexual pleasure and situated on the anterior vaginal wall. It is more likely that any increased sexual sensation is related to clitoral fibres around the urethra. Female ejaculation is also not consistently described; where it occurs it may be related to fluid being expressed from paraurethral (Skene’s) glands on either side of the urethra.
Clitoral surgery is known to risk sexual function and achievement of orgasm. Work assessing long-term outcome of childhood feminising surgery for those born with disorders/difference of sex development (intersex) showed a 1:4 anorgasmia rate for women who had undergone surgery [13]. Clitoral sensation is impaired in those who have undergone surgery [14]. Given the anatomical distribution of nerves, and the inevitability of surgical incisions causing cutting of nerve fibres in a densely innervated structure, it is likely that repeated surgery would risk function further.
Recent work assessed women who had undergone female genital cutting of the clitoris and compared them with unaffected women matched for age and parity [15]. Those who had undergone cutting had a significantly smaller clitoral volume and also reported significantly lower scores on sexual function and desire. Interestingly, there was no difference on the subscores of orgasm desire and satisfaction, but those who had undergone cutting did report significantly more dyspareunia.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree