Chapter 11 THE EVOLUTION OF ADIPOSE BIOLOGY AND REGENERATIVE MEDICINE
Historically, the functions of fat have been myopically assigned by investigators to static energy storage and protective structural support for organs and neurovascular bundles. In recent decades, however, adipose tissue has been revealed as a dynamic, biologically active organ involved in an array of physiologic and pathologic processes. Studies within the field of adipose tissue biology have most recently shown it to be a premier source of cells for evolving tissue engineering and regenerative therapies.
The dissociation and separation of adipose tissue into a single cell suspension was first described by Rodbell in 1964, 1 , 2 allowing characterization and study of its different cellular components. Studies subsequently revealed the adipocyte precursor cell (preadipocyte) and its differentiation program. 3 – 5
Although the cellular constituents of adipose tissue became a central topic of study within the contexts of obesity and fat grafting, 6 – 8 it was not until the past 15 years that the preadipocyte became a focal point for the emerging field of tissue engineering. 9 – 13
After the potential therapeutic benefit of bone marrow stromal cells was tempered because of their limited supply, interest in adipose as a cell source escalated, concurrent with the delineation of adipose cellular components and resulting appreciation of cellular diversity.
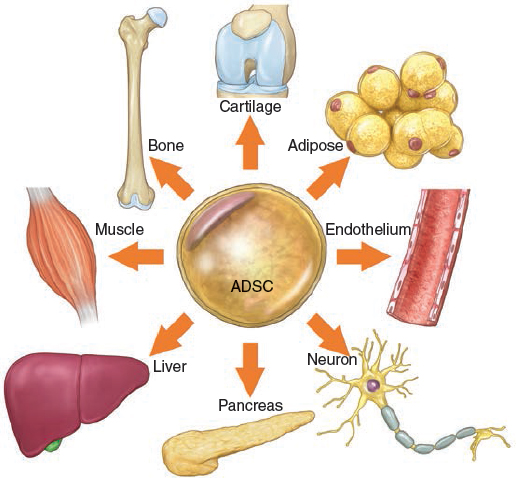
In 1986 Jarrell et al 14 published a paper on the isolation and use of adipose-derived microvascular endothelial cells for lining synthetic vascular grafts. 15 By the turn of the century, the idea emerged that adipose tissue contained progenitor/stem cells with the ability to differentiate into lineages other than adipocytes. In 2000 Halvorsen et al 16 described the osteogenic differentiation of adipose-derived stromal cells, in 2001 Zuk et al 17 published findings demonstrating that adipose tissue was a novel source of mesenchymal stem cells (MSCs) with multilineage differentiation potential. There is now a large body of literature to suggest that a wide variety of tissues may contain an MSC fraction. To this end, Crisan et al 18 and several other independent groups have proposed a “unifying theory” of sorts that MSCs from various organs or tissues are located in the perivascular space or niche. 19 , 20 Indeed, the concept that pericytes (also called mural cells or Rouget cells) have multilineage potential is not new; it dates back at least to the early 1980s. 21 – 25 Furthermore, the unique microvascular composition of adipose tissue has been appreciated for at least a quarter century or longer. 26 – 28
Cell Identity and Terminology
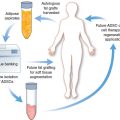
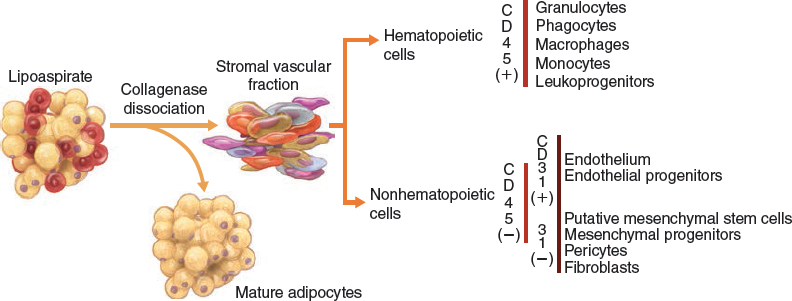
Subcutaneous human adipose tissue is an accessible and abundant cell source for multiple applications in regenerative medicine. Researchers and biotech companies have explored different methods and point-of-care devices to isolate regenerative cells from human adipose aspirates obtained from liposuction procedures. Upon collagenase dissociation of these lipoaspirates and subsequent exclusion of mature adipocytes, an array of cells collectively known as the stromal vascular fraction (SVF) is obtained. The SVF is a primary, uncultured, heterogeneous population including fibroblasts, pericytes, vascular progenitors, and multipotent mesenchymal stem cells (MSCs). 14 , 16 , 17 , 29
Within the SVF, the “traditional” cells of interest express the following cluster of differentiation markers: CD45–, CD235a–, CD31–, CD34+, and the following surface antigens: CD13, CD73, CD90, CD105. ADSCs, in contrast, have been plated into culture (that is, selected by plastic adherence) and possess the multipotential capability of differentiation into a variety of mesenchymal lineages. ADSCs express clusters of differentiation markers (CD90, CD105, CD73, CD44, CD36, and so on), but are negative for CD45, CD31, CD 106. 29 ADSCs are therefore a distinct entity, as defined by immunophenotypic expression and plastic adherence, and were previously assumed to have different developmental plasticity from other stromal cells. In fact, several studies have validated these assumptions and have described preferential lineage models for ADSCs (which have a greater propensity of differentiating into muscle tissue) and MSCs (which more readily differentiate into chondrogenic and osteogenic lineages). 29 , 30
Immunomodulation
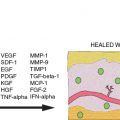
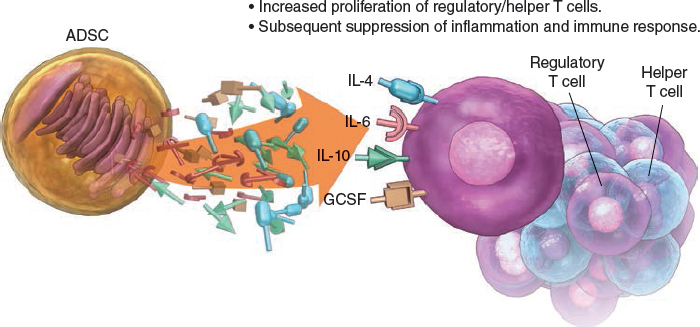
The scope of adipose tissue and its uses was further broadened when the immunomodulatory effects of ADSCs were discovered. Adipose stem cells increase the secretion of cytokines and growth factors (specifically IL-6, IL-10, IL-4, granulocyte colony–stimulating factor [G-CSF]) and modify cell-mediated immunity (that is, they stimulate proliferation of regulatory and helper T cell phenotypes). The ability of these cells to suppress an immune response has obvious implications for autoimmune disorders, these antiinflammatory effects have been exploited in studies of several pathologic processes, such as hemorrhagic stroke, reactive airway disease, multiple sclerosis, and rheumatoid arthritis. 31 – 36 Studies in transplant-related immunosuppressive therapy are currently being undertaken and expanded. 31 , 37
Secretome
The ability of ADSCs to modify their environment is not restricted to direct differentiation into target tissue cells. A secretome is the compilation of the secretory organelles and their secreted protein products. The adipose secretome generates adipokines and cytokines with both local and systemic effects—abilities that extend to isolated ADSCs. ADSC-conditioned media, a direct product of the ADSC secretome, has been demonstrated in several studies to exert these endocrine and paracrine effects. For example, ADSC-conditioned media generated under proinflammatory stimuli improve wound healing rates in rodent wound-healing models by means of the attraction of monocytes to sites of inflammation and tissue damage. Tissue regeneration through extracellular matrix (ECM) remodeling, keratinocyte migration, an increase in capillary density has also been seen with the application of ADSC-secreted cytokines (connective tissue growth factor [CTGF], SERPINE1, plasminogen activator inhibitor [PAI], hepatocyte growth factor [HGF], FGF-1). 38 – 47 The aforementioned immunomodulation effects of ADSCs, which are of increasing importance in the creation of novel treatment modalities for autoimmune-mediated disease, have been demonstrated by ADSC-conditioned media suppression of peripheral blood mononuclear cells through modulation of interferon-gamma (IFN-gamma), HGF, prostaglandin E2 (PGE- 2), transforming growth factor-beta 1 (TGFB-1), indoleamine 2,3-dioxygenase (IDO), interleukin-10 (IL-10). 32 , 35 , 40 – 43 The effects of ADSC-conditioned media have experimental efficacy in other pathologies, including angiogenesis in ischemic limb tissue and acute myocardial injuries. 38 , 39
Interestingly, the dimensional configuration of ADSCs used to generate conditioned media modifies the ADSC-secretome, and subsequently the intensity of media-induced endocrine and paracrine effects. Three-dimensional scaffold-free spheroid preparations of ADSCs generate media that exerts more potent effects on wound-healing rates than media generated by ADSCs grown in two-dimensional monolayers, perhaps because of reported synergistic effects that can occur between ECM and growth factors. 48 – 50
Utilizing SVF in Cell-Based Therapies
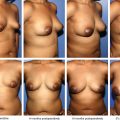
It is of no surprise that depending on the outcome of interest, clinical applications and cell therapies might be better served by particular derivations of lipoaspirates (that is, the stromal vascular fraction versus adipose stem cells). Although there are many factors affecting decision-making, including regulatory and facility requirements, cell dose requirements, associated costs, one factor of particular importance is the processing time. The SVF can be isolated without selection by plastic adherence, therefore, processing times from fresh lipoaspirate to fractionated product is on the order of minutes as opposed to the ADSC requirement of multiple incubation days. This allows SVF cells to be implemented in a real time, “point-of-care” manner that also provides unique advantages, including less likelihood of contamination, lower associated costs, rapid therapeutic application. Several studies have demonstrated improved results from SVF when compared with ADSCs, including more intense inhibition of encephalomyelitic progression, improved range of motion and decreased pain in canine hip dysplasia models, augmented cartilage and subchondral bone regeneration in caprine models. 51 – 53
Some disadvantages of using the SVF that have been previously reported are insufficient cell dose if high cell numbers are needed, lack of efficacy. It is of note that the majority of research performed to date has focused on the characterization, biology, therapeutic potential of ADSCs, as opposed to that of SVF cells. 54 – 57 There are few studies in the literature directly comparing (using different experimental models) the efficacy and potency of SVF cells with that of ADSCs. Furthermore, a guided definition of adipose-derived stem cells as a means to progress toward consensus in the field is a relatively new endeavor. 29 It should be emphasized that many published reports inaccurately exchange the terms ADSCs and SVF, using them interchangeably despite their recent characterization as two distinct cellular populations. In summary, it remains to be determined whether putative functional differences between SVF cells and ADSCs have relevance for specific clinical applications. Additional preclinical data and clinical trials comparing both cell types may help to clarify these questions.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree